Article Categories:
- All
- AI
- Articles
- Clinical Application
- Clinical Case
- Cone Beam CT
- Conference
- CubeVue Software
- Curvebeam AI Connect
- CurveBeam Events
- Customer Spotlight
- Evidence & Exchange
- HiRise
- InReach
- InReach HR-pQCT
- LineUP
- News
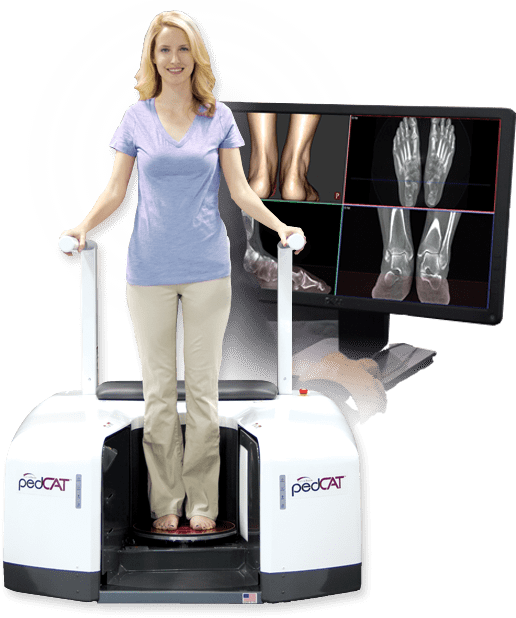
- pedCAT
- Podcasts
- Poster Presentation
- Product Cubevue Software
- Scientific Literature
- SkyRise
- Testimonial
- Uncategorized
- Videos
- Webinars
- Weight Bearing CT
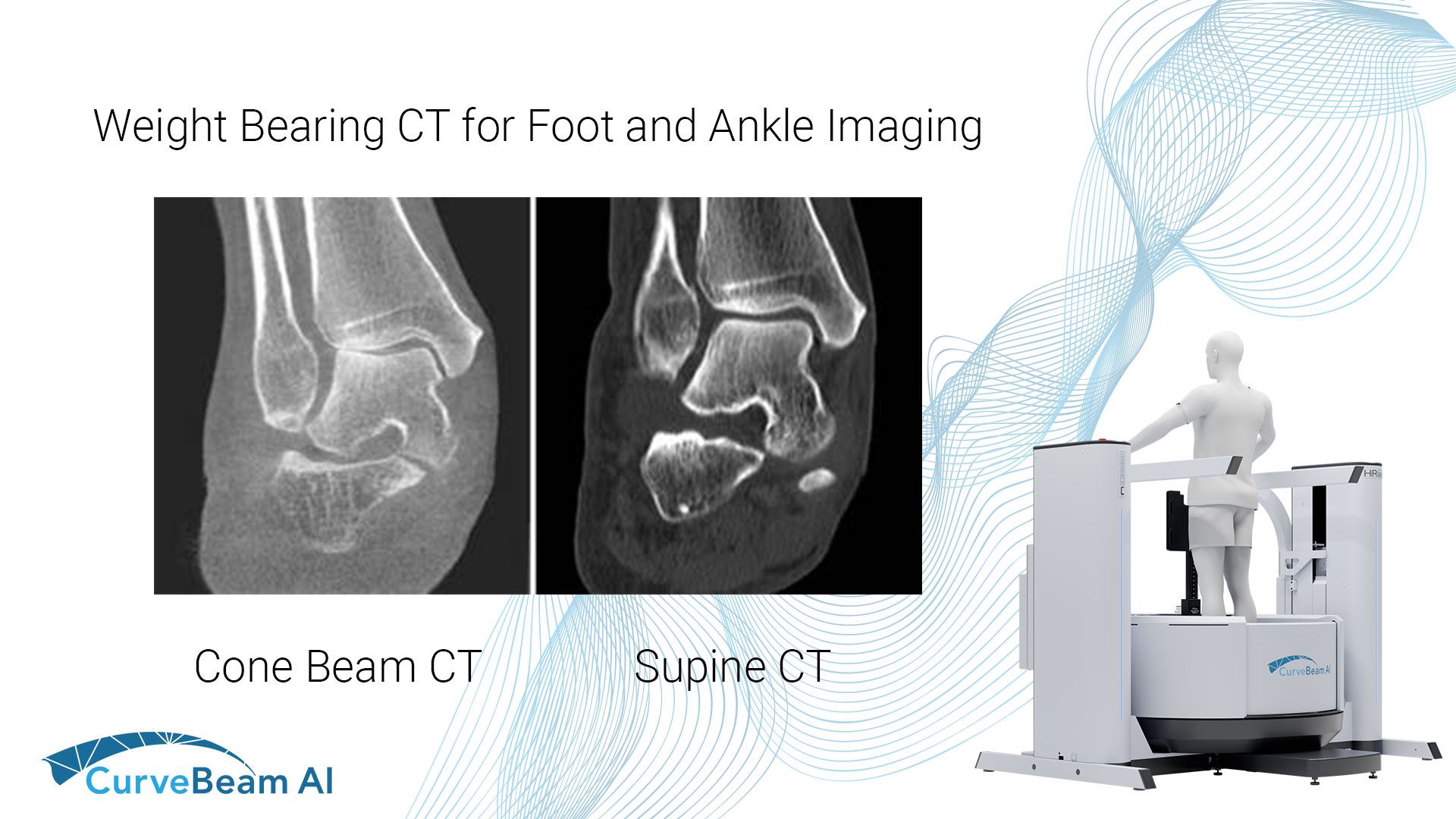
Advancing Accuracy in Orthopedic Imaging
AI-Driven Segmentation on Weight Bearing CT Introduction Accurate segmentation is critical for orthopedic workflows, including…
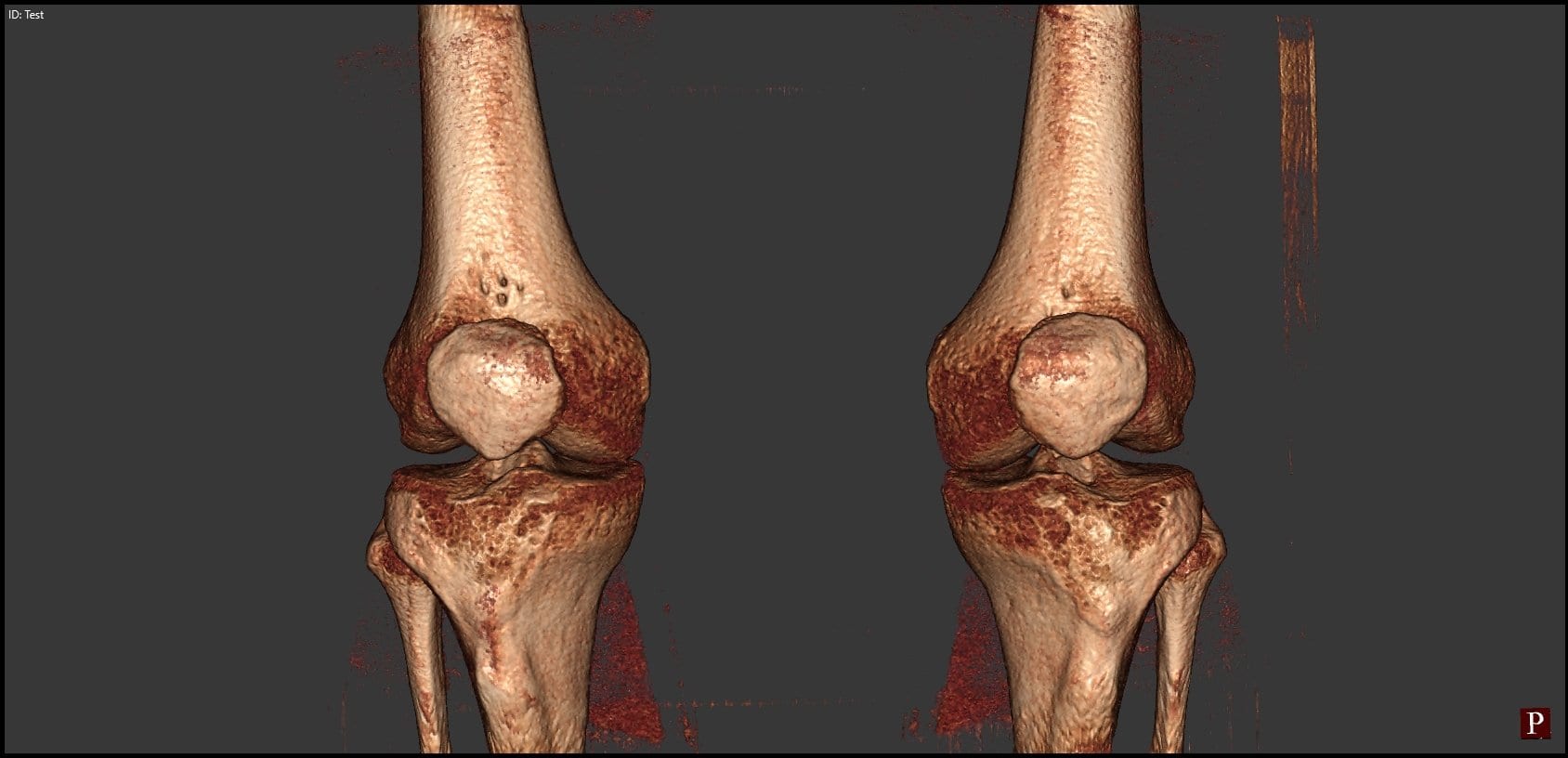
Sub-Millimeter Accuracy in Weight-Bearing Orthopedic Imaging
Orthopedic decision-making depends on accurate representation of anatomy—particularly when joint alignment and bone relationships change…
Imaging, Innovation, and Independence: A Conversation with Dr. H. Kurtis Biggs
In a recent episode of CurveBeam AI Connect, Vice President of Marketing Vinti Singh spoke…
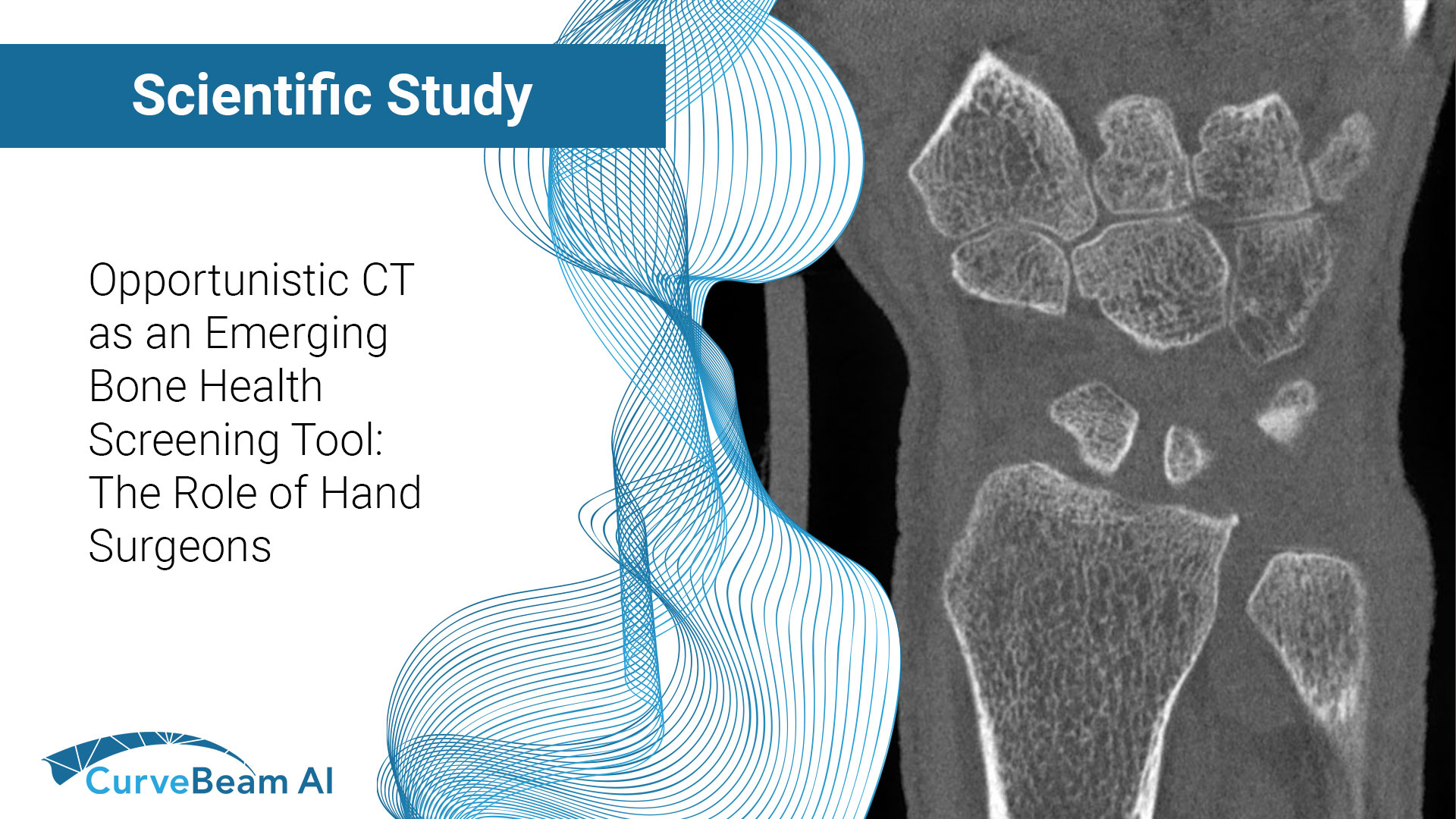
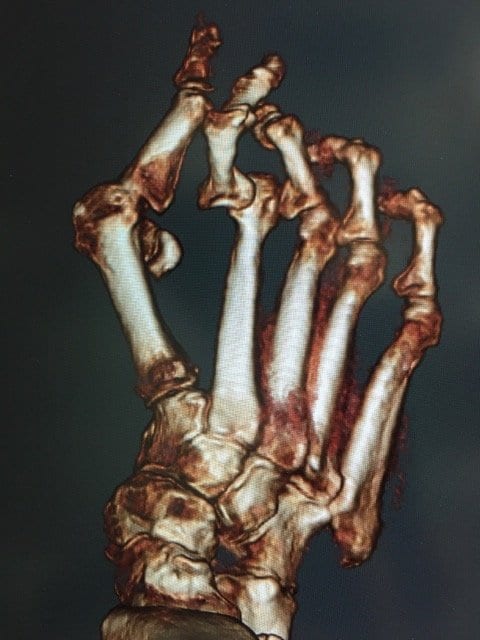
CT as an Emerging Bone Health Screening Tool: The Role of Hand Surgeons
Fragility fractures are often the first visible sign of underlying osteoporosis but too often, they…
Enhancing Precision in Total Ankle Arthroplasty with PSI and Weight Bearing CT
In recent years, patient-specific instrumentation (PSI) for total ankle arthroplasty (TAA) has gained momentum, driven…
New Study Confirms WBCT Delivers Equivalent 3D Bone Models to MDCT for Joint Replacement Planning
A groundbreaking study published in Scientific Reports confirms that weight-bearing cone beam CT (WBCT) produces…
CurveBeam AI’s Enhanced HiRise System Receives Health Canada Approval and CE Marking
Hatfield, PA — July 1, 2025 — CurveBeam AI, a global leader in weight bearing…
Smarter Surgical Planning Starts with Weight Bearing CT: Highlights from CurveBeam AI at the Robotic Knee Conference
For Scientific Exchange Only – Not for Promotional Use. This communication includes information about an…
Orthopedic Leaders Share Real-World Impact of Weight Bearing CT at AAOE 2025
At the 2025 American Alliance of Orthopedic Executives (AAOE) Annual Conference in Atlanta, CurveBeam AI…
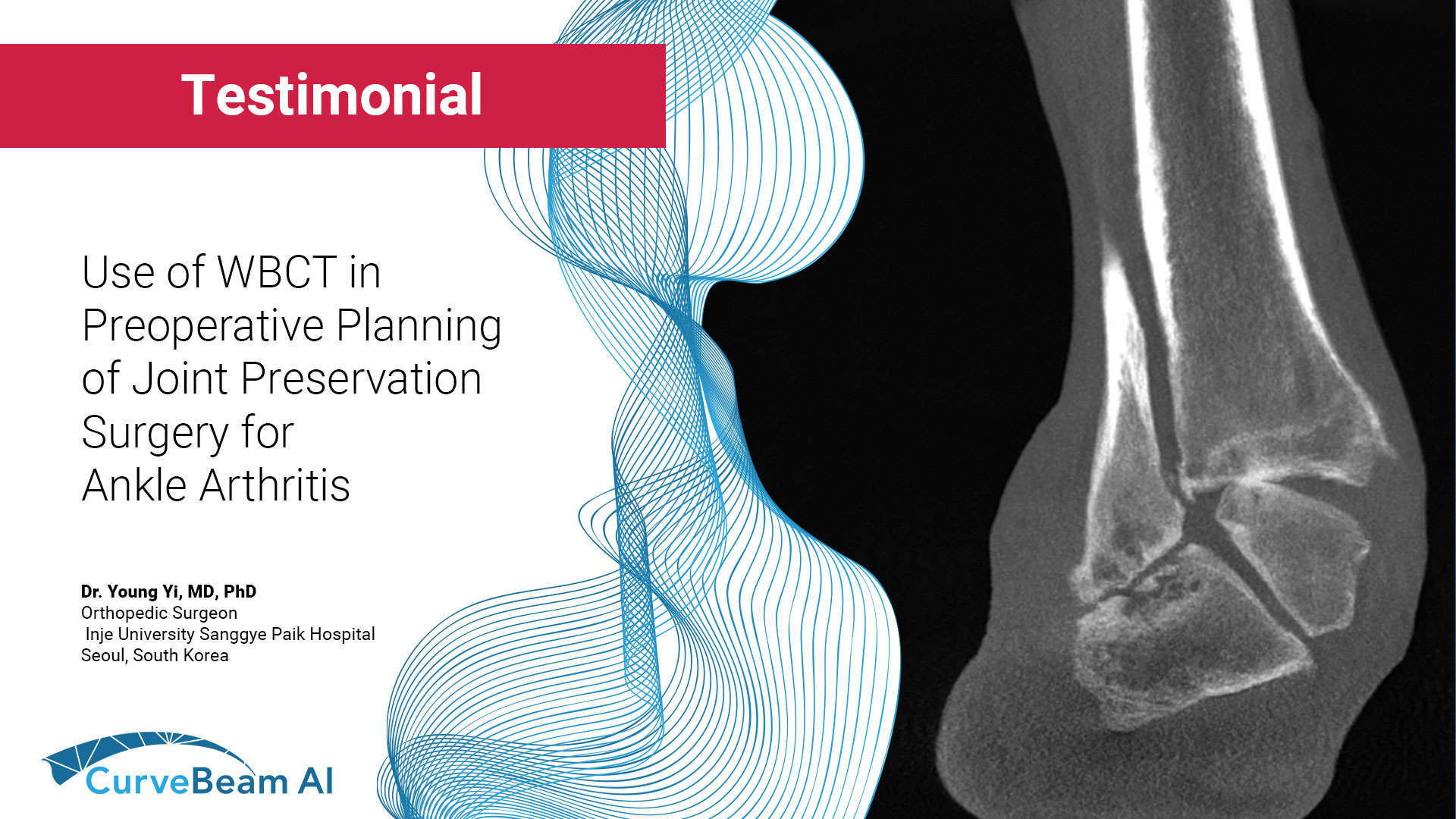
Use of WBCT in Preoperative Planning of Joint Preservation Surgery for Ankle Arthritis
In a recent AOFAS WBCTS video, Young Yi, MD, PhD, orthopedic surgeon, Inje University Sanggye…
Not Just for Complex Cases: The Everyday Impact of Weight Bearing CT
Scott Shawen, MD, a foot and ankle surgeon from OrthoCarolina, Charlotte, North Carolina shared how…
Practical Benefits of Using a Cone Beam Instead of X-Rays for Pre-op and Post-op Imaging
Dr. Marie-Aude Munoz, Orthopedic Surgeon, Montpellier, France, shares her personal journey of integrating weight bearing…
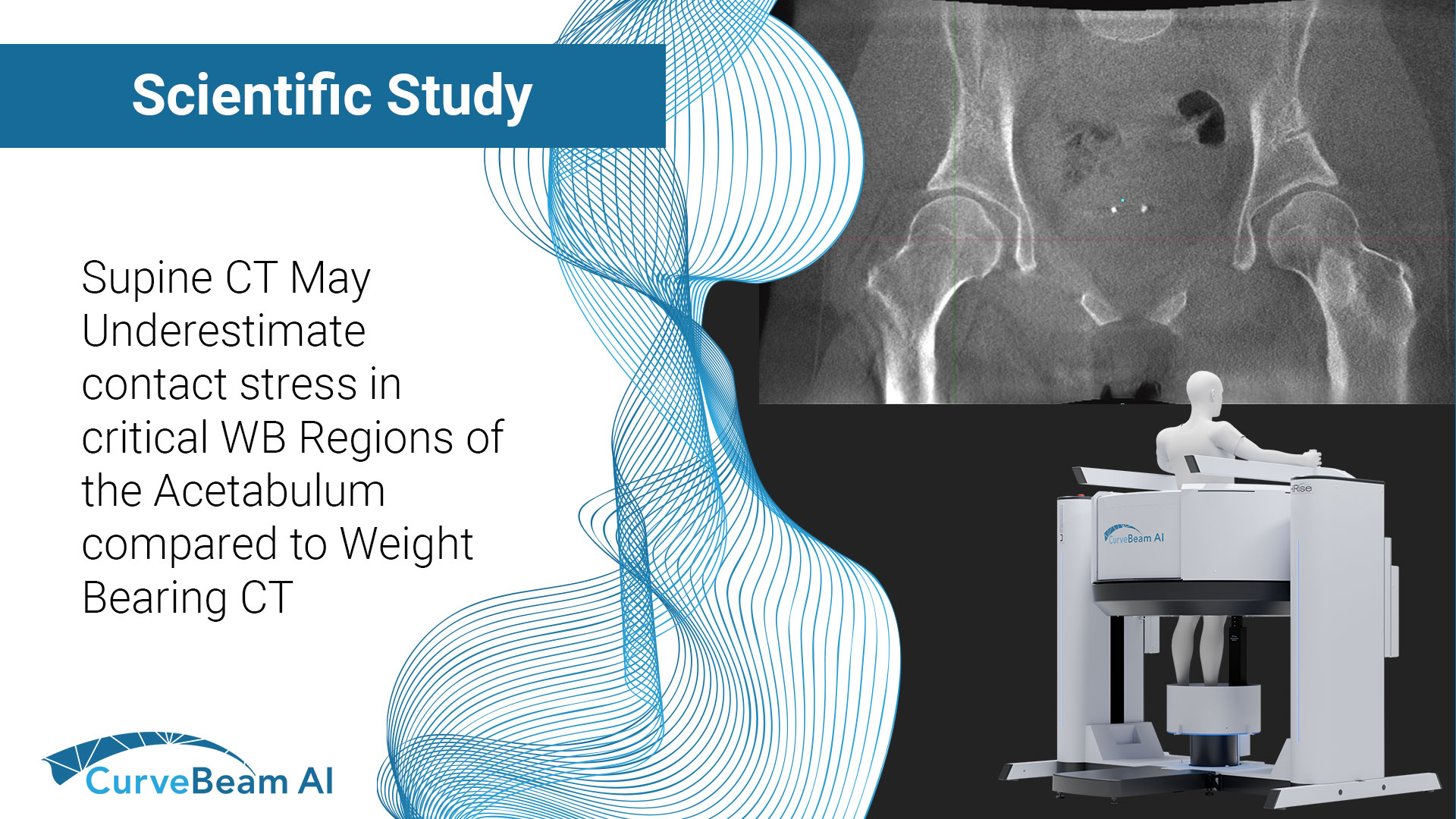
Supine CT May Underestimate Contact Stress in Critical WB Regions of the Acetabulum Compared to Weight Bearing CT
Key Points: Computational models of the hip often omit patient-specific functional orientation when placing imaging-derived…
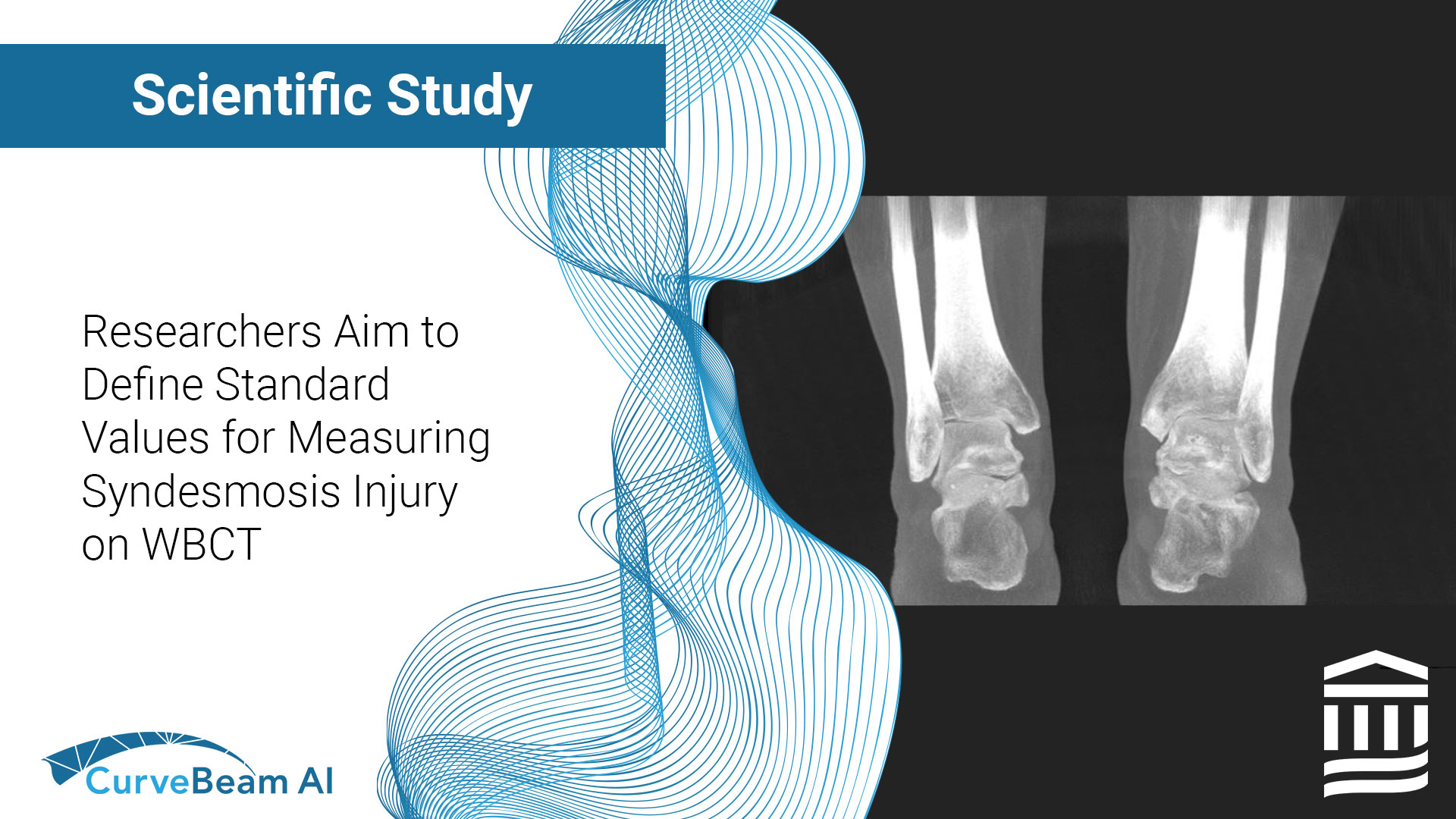
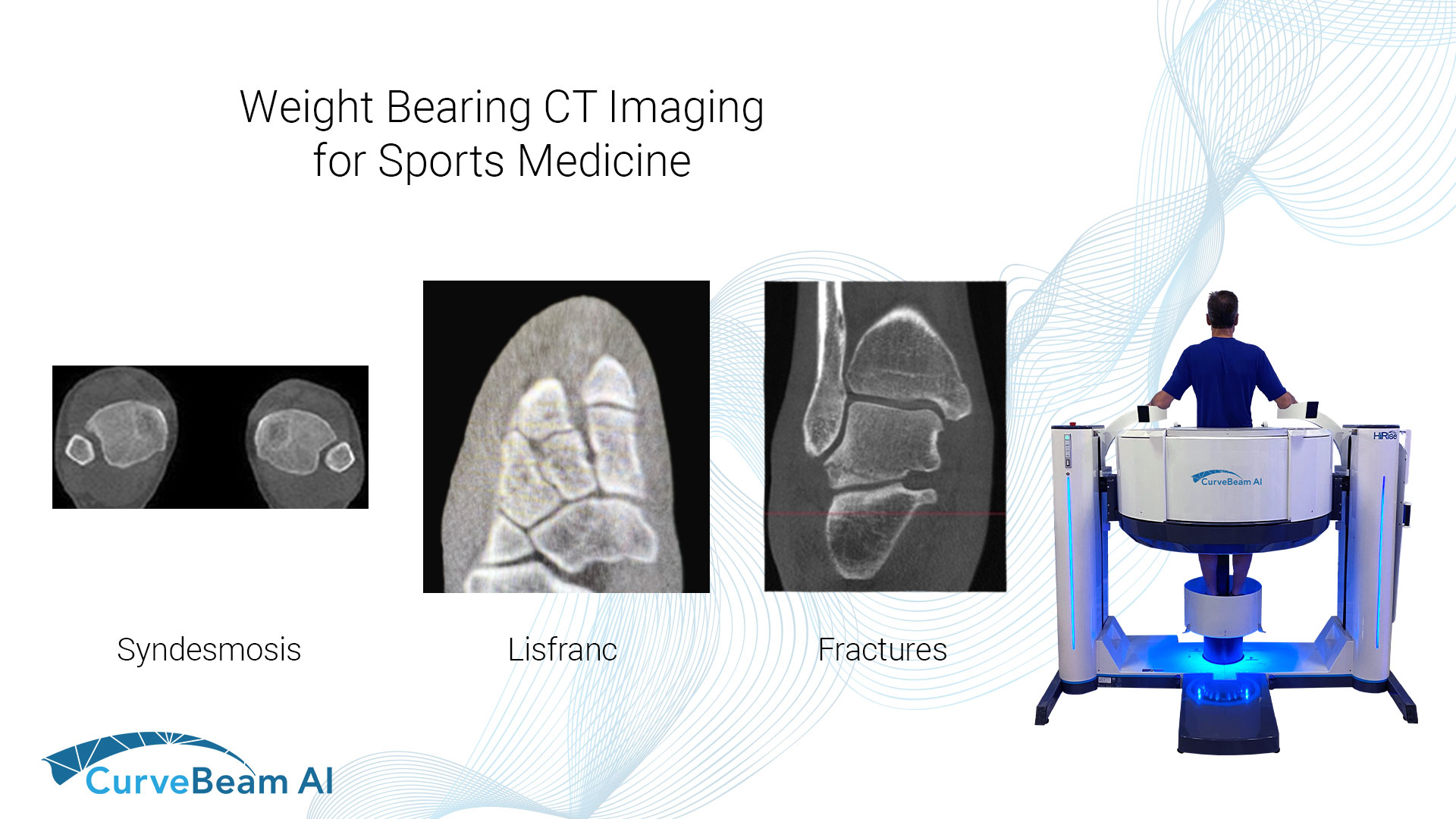
Researchers Aim to Define Standard Values for Measuring Syndesmosis Injury on WBCT
Key Points: Weight Bearing CT (WBCT) allows for bilateral comparative data that is imperative when…
Fewer Missed Diagnoses: The Power of Point-of-Care Weight Bearing CT Imaging
Weight bearing CT is more than just a modality for functional imaging — "it’s a…
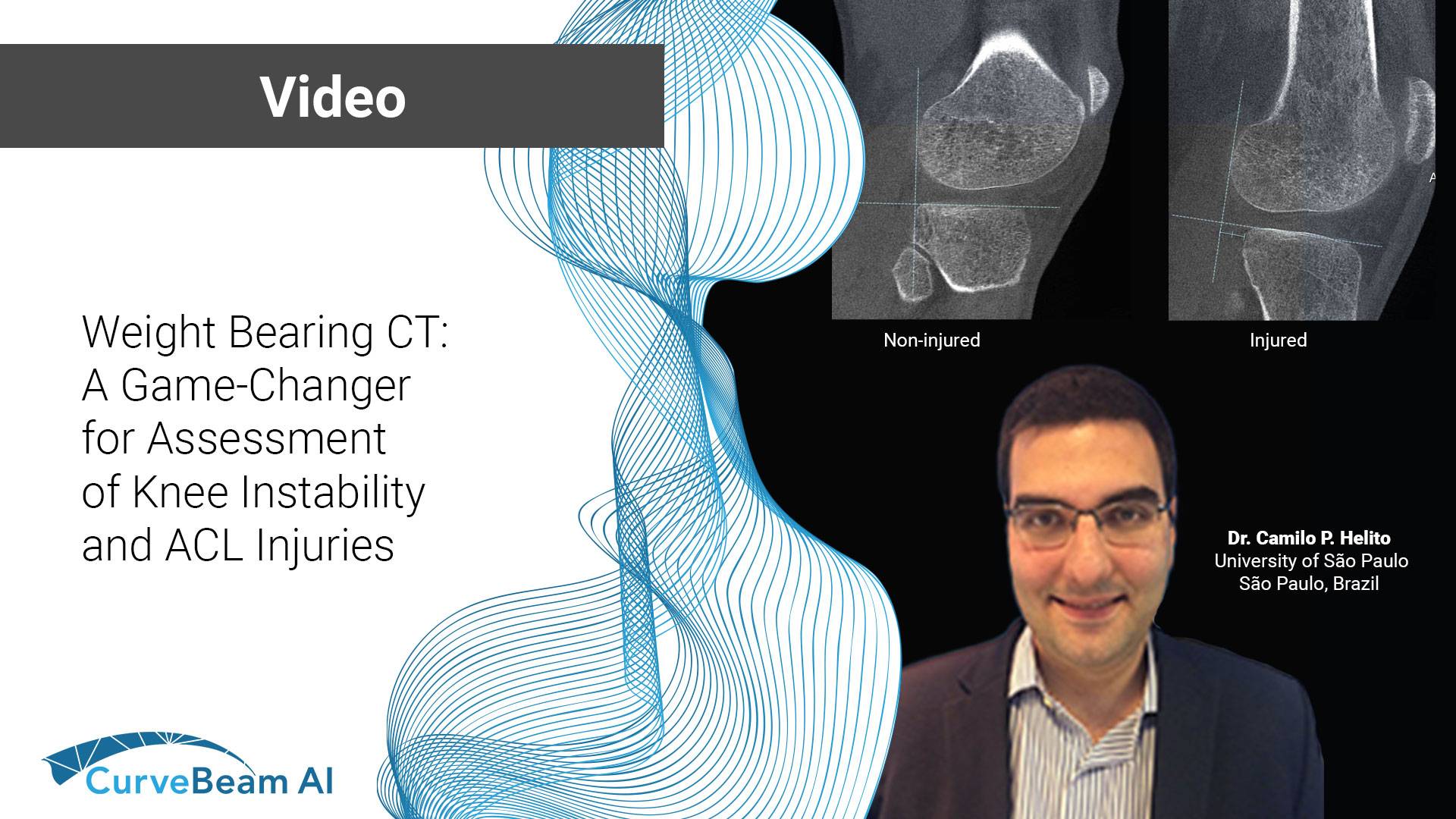
Weight Bearing CT: A Game-Changer for Assessment of Knee Instability and ACL Injuries
Weight bearing CT offers a new way to evaluate knee instability and ACL injuries.- a…
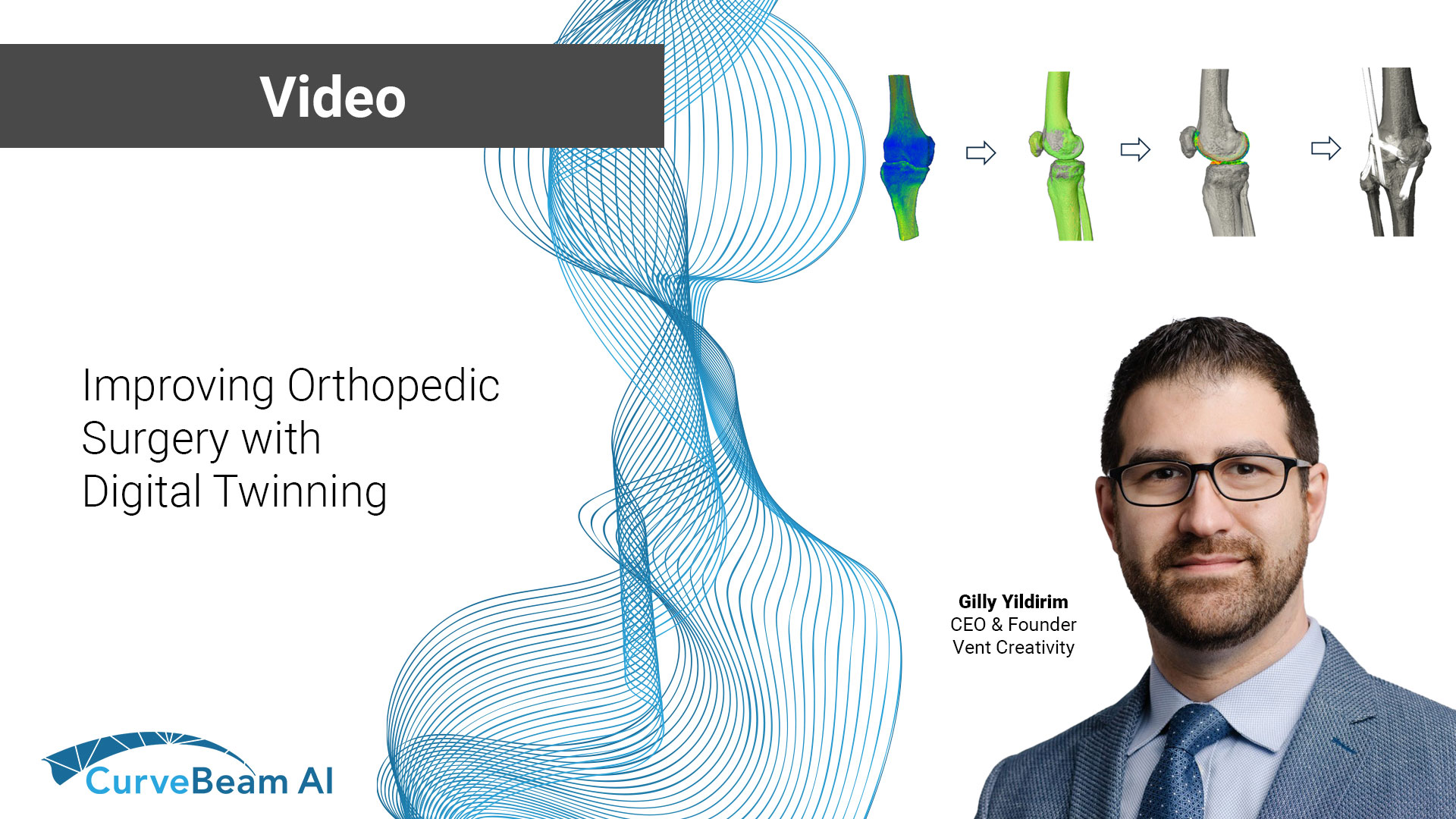
Improving Orthopedic Surgery with Digital Twinning
Advancements in CT imaging are transforming orthopedic surgery, reducing surprises and improving surgical outcomes. In…
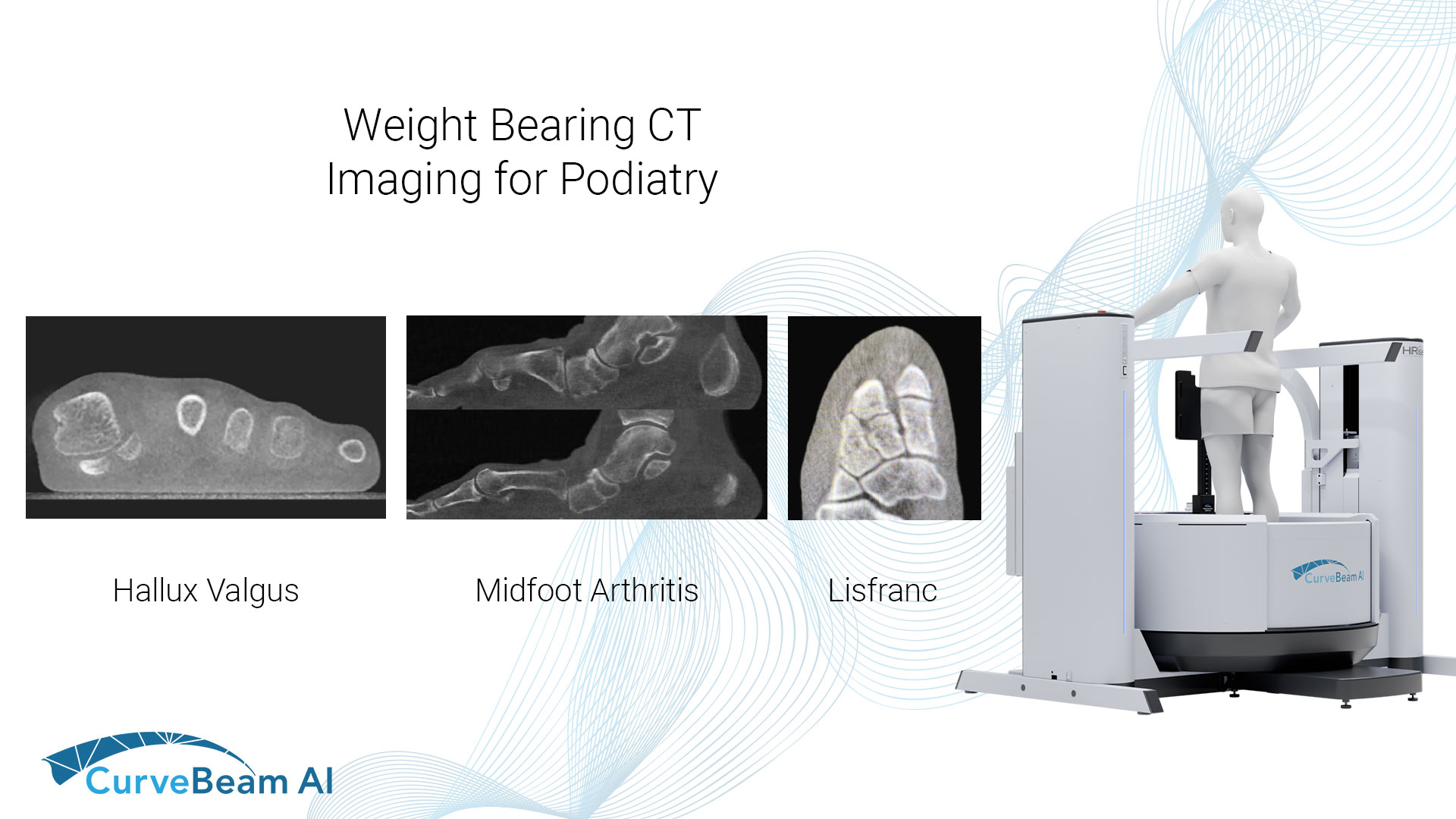
Advantages of WBCT Measurement Parameters in Hallux Valgus
When it comes to hallux valgus (bunion) deformity, weight bearing CT provides significant advantages over…
WBCT-Based Method to Detect Implant Loosening Shows Promise
Key Points: Radiostereometric Analysis (RSA) uses X-Ray and markers inserted into the bone intraoperatively to…
Stay ahead of the Curve with Weight Bearing CT
In the world of orthopedic diagnostics, imaging plays a crucial role in identifying deformities and…
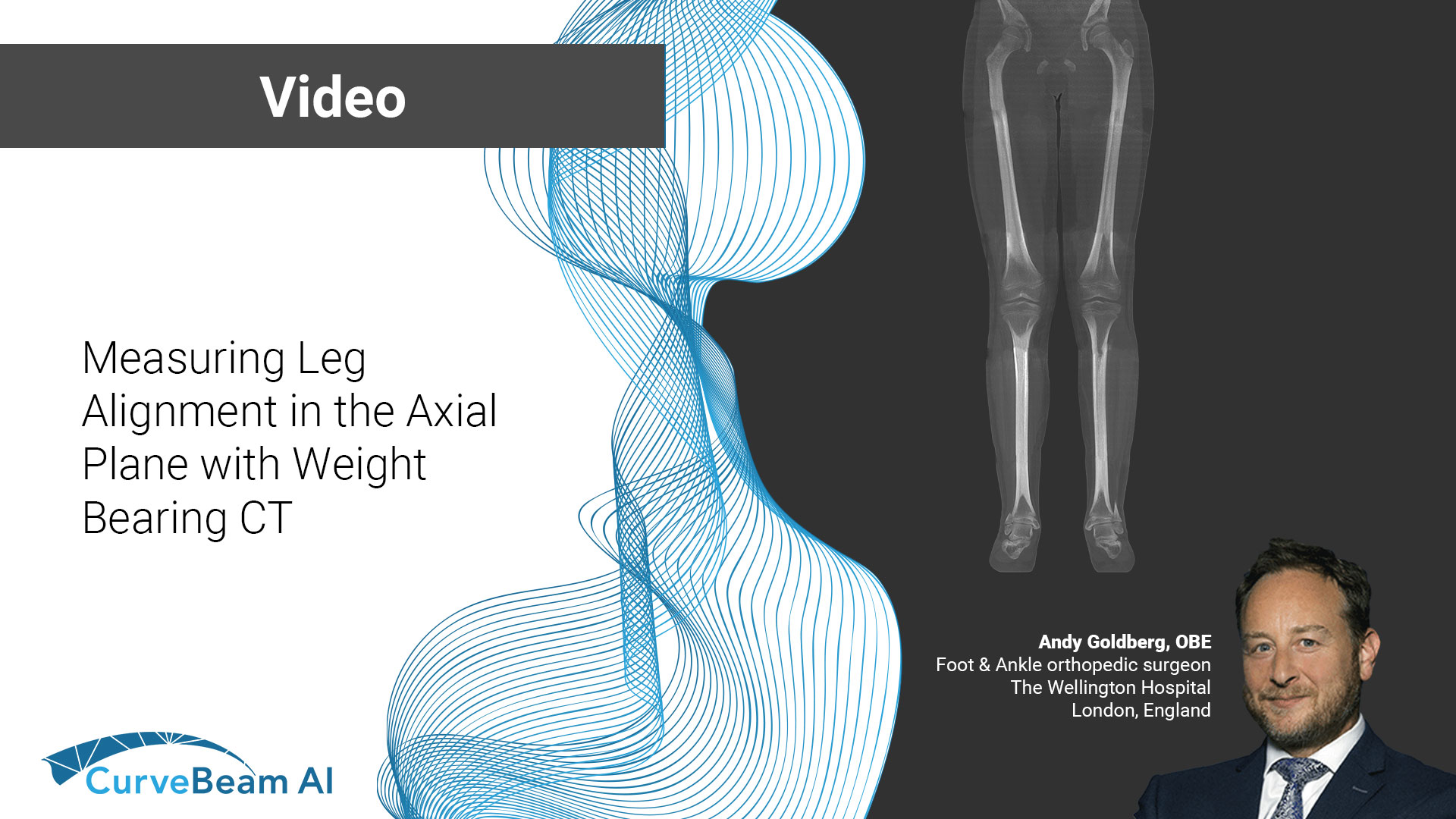
Measuring Leg Alignment in the Axial Plane with Weight Bearing CT
At the International Weight Bearing CT Society (WBCT) session during the 2024 European Foot &…
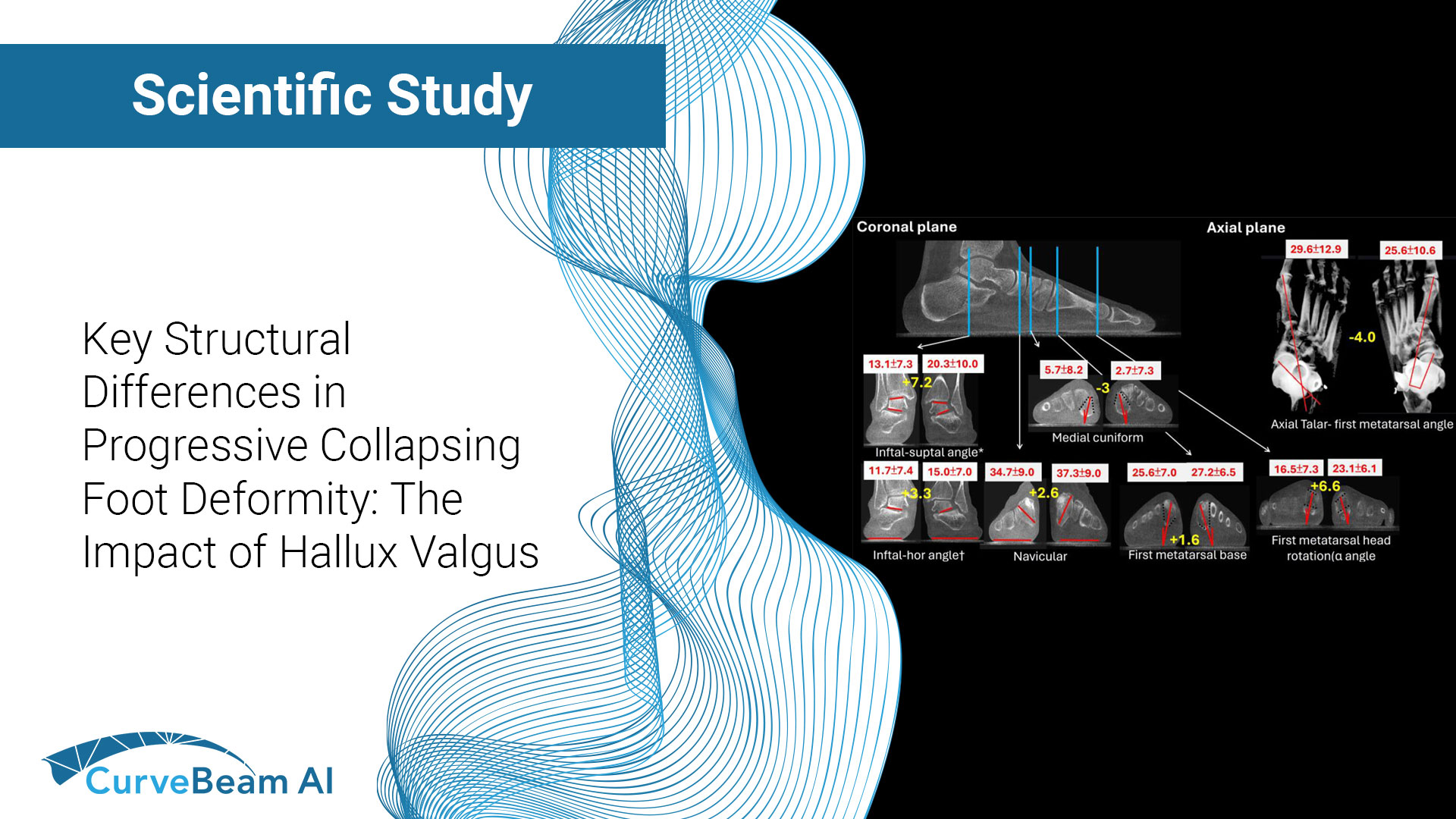
Comparative Analysis of Structural Differences in Progressive Collapsing Foot Deformity With and Without Hallux Valgus
Key Points: Weight Bearing CT (WBCT) helps visualize several structural and alignment differences between Progressive…
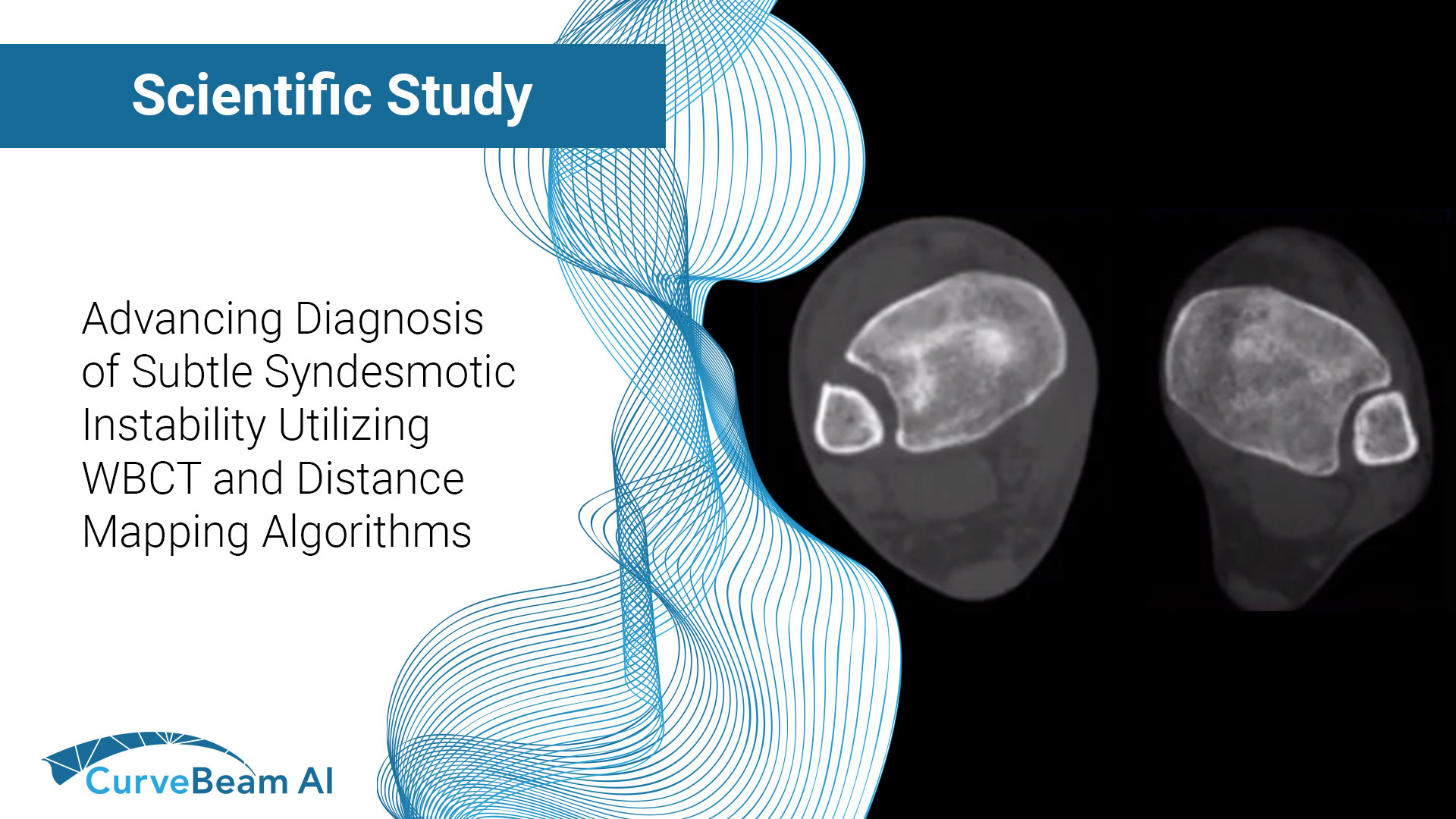
Cadaveric Diagnostic Study of Subtle Syndesmotic Instability Using a 3-Dimensional Weight Bearing CT Distance Mapping Algorithm
Key Points: Imaging modalities such as plain radiographs (X-Ray), computed tomography (CT), and magnetic resonance…
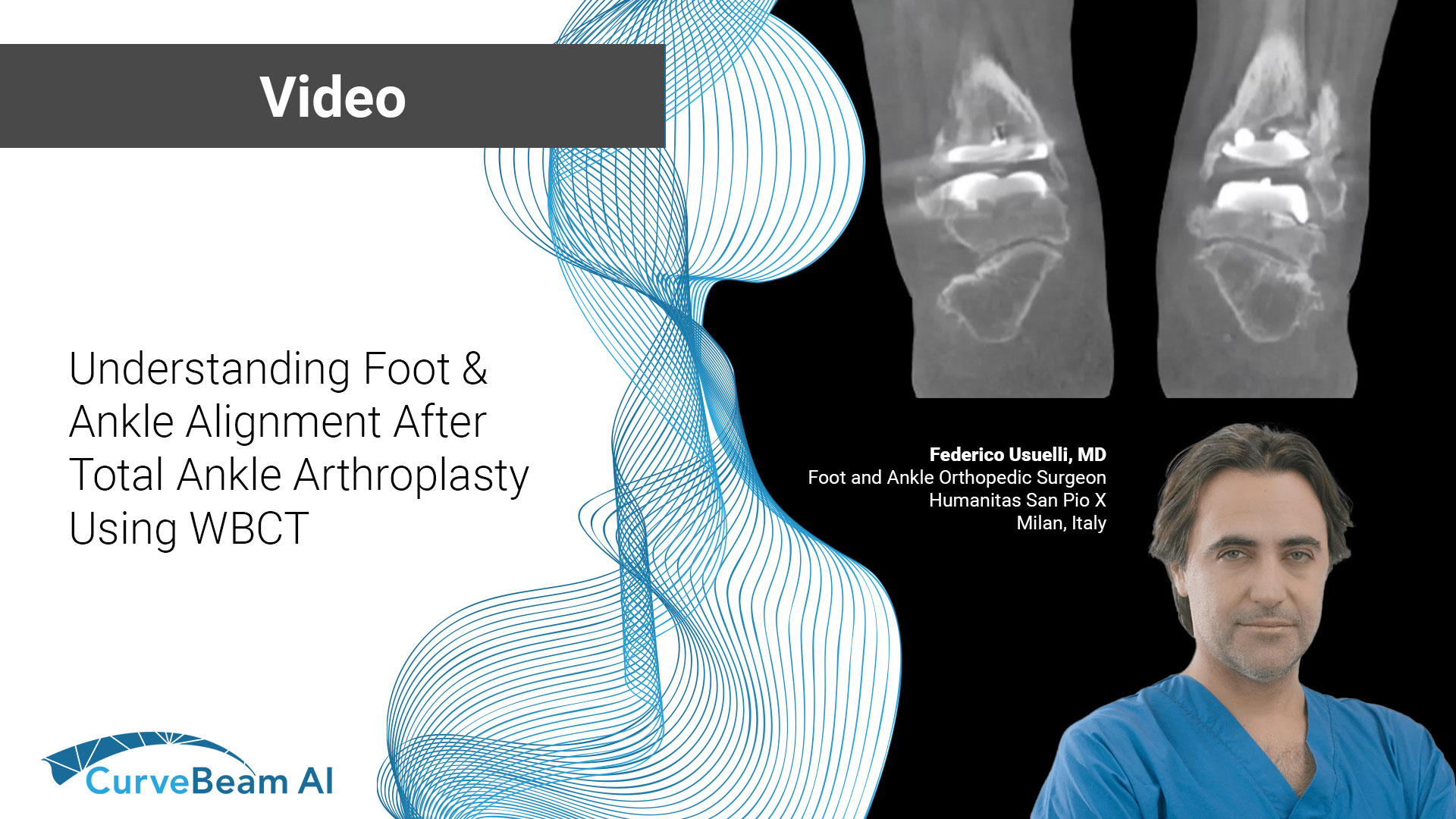
Understanding Foot & Ankle Alignment After Total Ankle Arthroplasty Using WBCT
At the International Weight Bearing CT Society (WBCT) session during the 2024 European Foot &…
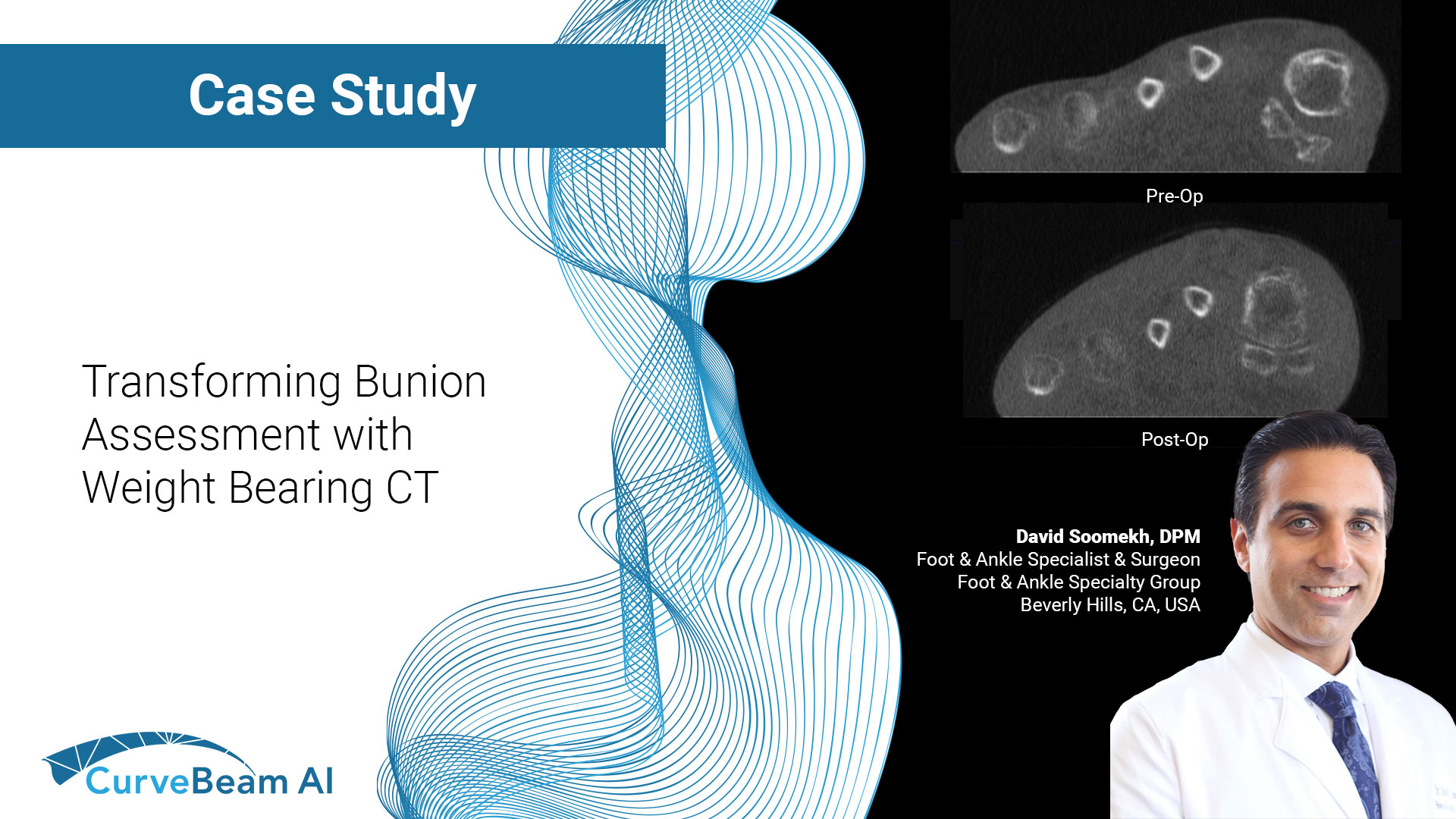
Transforming Bunion Assessment with Weight Bearing CT
By Dr. David Soomekh, DPM Foot and ankle specialist and surgeon, sports medicine & reconstructive…
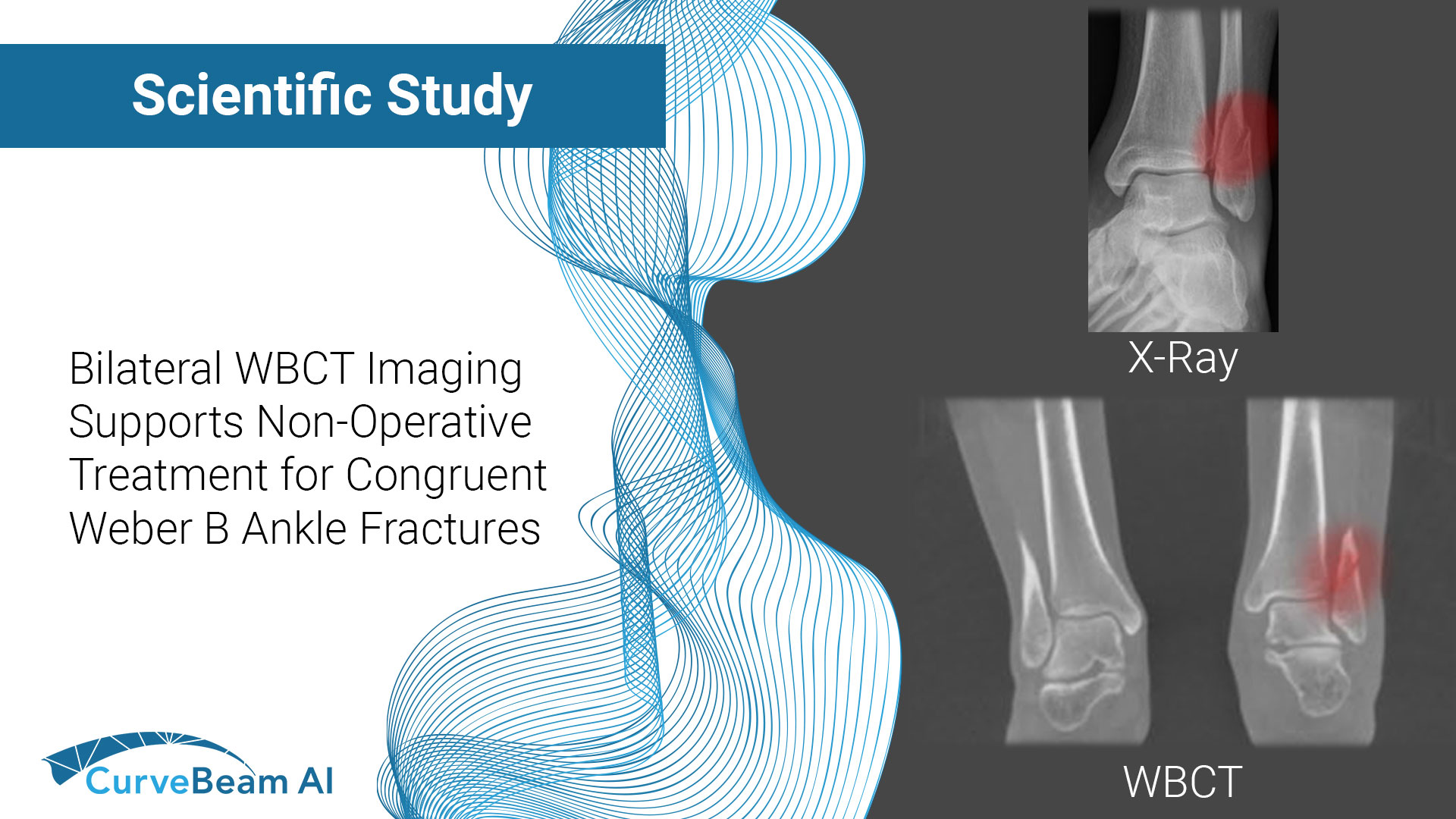
Congruent Weber-B Ankle Fractures Do Not Alter Tibiotalar Contact Mechanics
Key Points: Weight Bearing CT (WBCT) provides critical data otherwise missed in weight bearing radiographs…
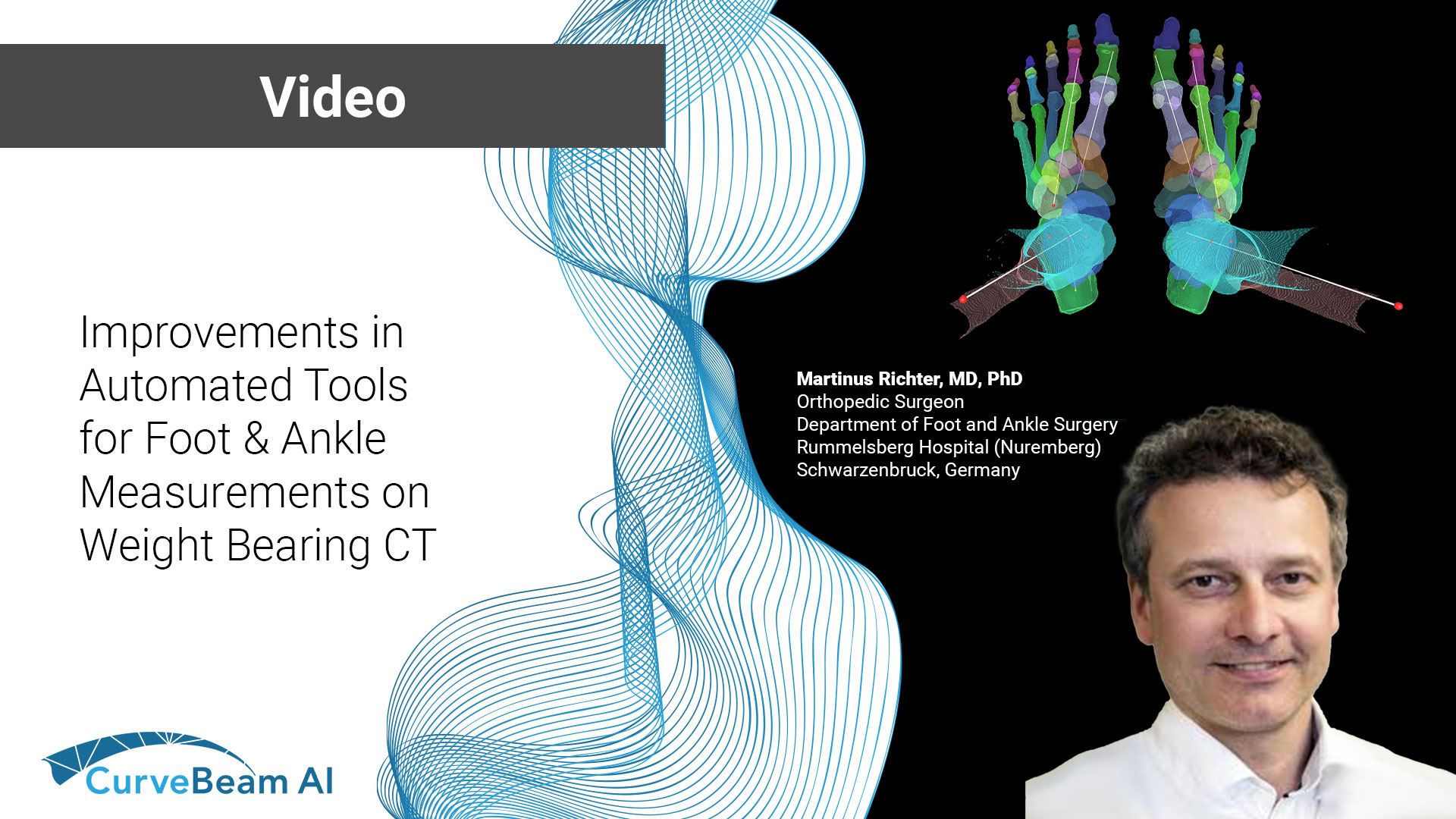
Improvements in Automated Tools for Foot & Ankle Measurements on Weight Bearing CT
At the International Weight Bearing CT Society (WBCT) session during the 2024 European Foot &…
Shaping the Future: CurveBeam AI’s Countdown to 2025
As 2024 draws to a close, we’re excited to highlight CurveBeam AI’s top five milestones…
Making Patient-Specific Solutions More Accessible Every Step of the Way
We all know the benefits of patient-specific planning, but how do we make it more…
Surgeons Weigh In: Top Indications for WBCT: Part 5
Weight bearing CT (WBCT) imaging has fundamentally changed the evaluation and management of foot and…
Surgeons Weigh In: Top Indications for WBCT: Part 3
Weight bearing CT (WBCT) imaging has fundamentally changed the evaluation and management of foot and…
Celebrating National Radiologic Technology Week® (NRTW).
Providing patients with safe medical imaging examinations and radiation therapy treatments is the primary goal…
Surgeons Weigh In: Top Indications for WBCT Part 2
Weight bearing CT (WBCT) imaging has fundamentally changed the evaluation and management of foot and…
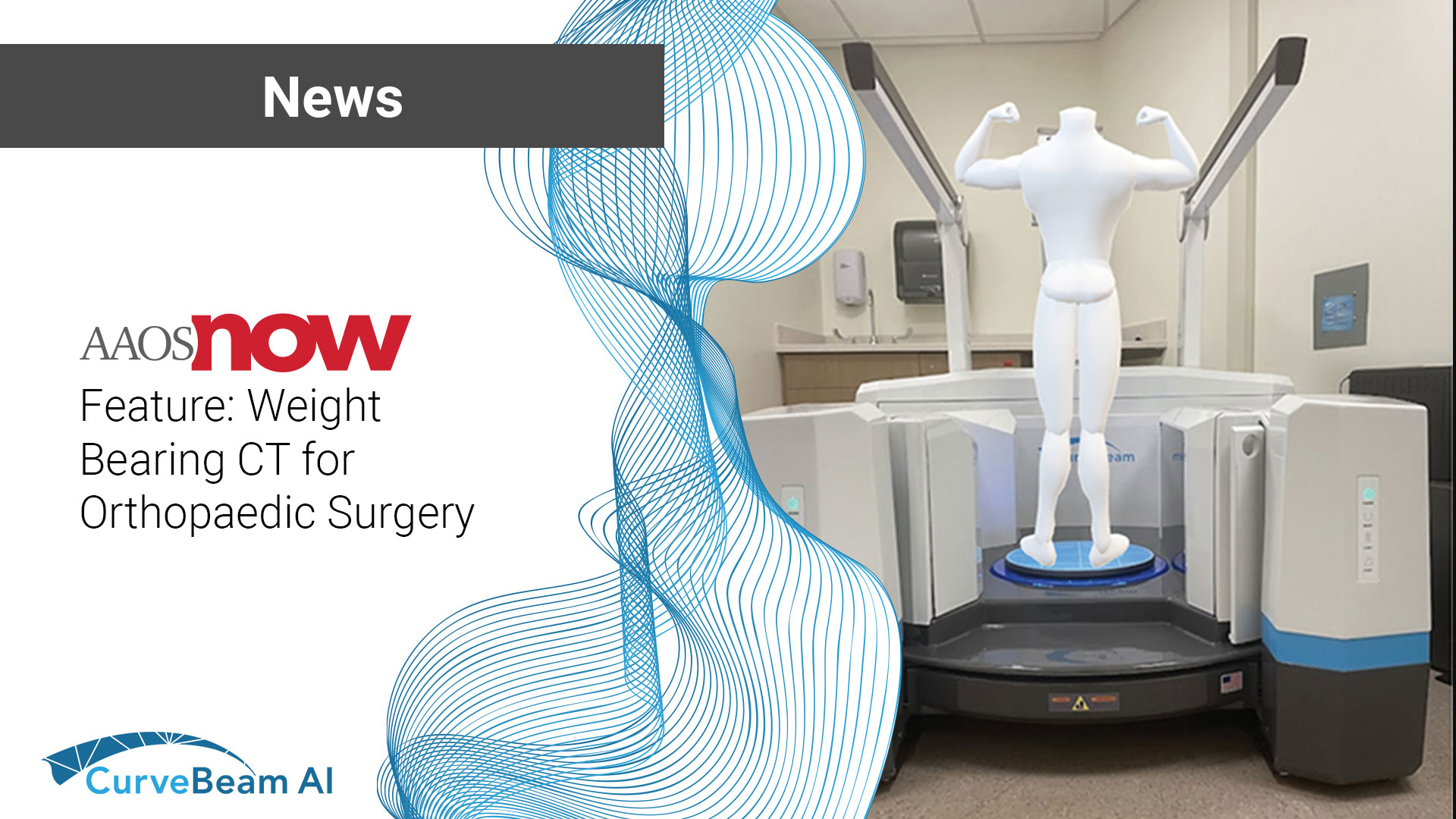
AAOS Now Feature: Weight Bearing CT for Orthopaedic Surgery
The August/September 2024 issue of AAOS Now includes the first of of a two-part series…
Surgeons Weigh In: Top Indications for WBCT
Weight bearing CT (WBCT) imaging has fundamentally changed the evaluation and management of foot and…
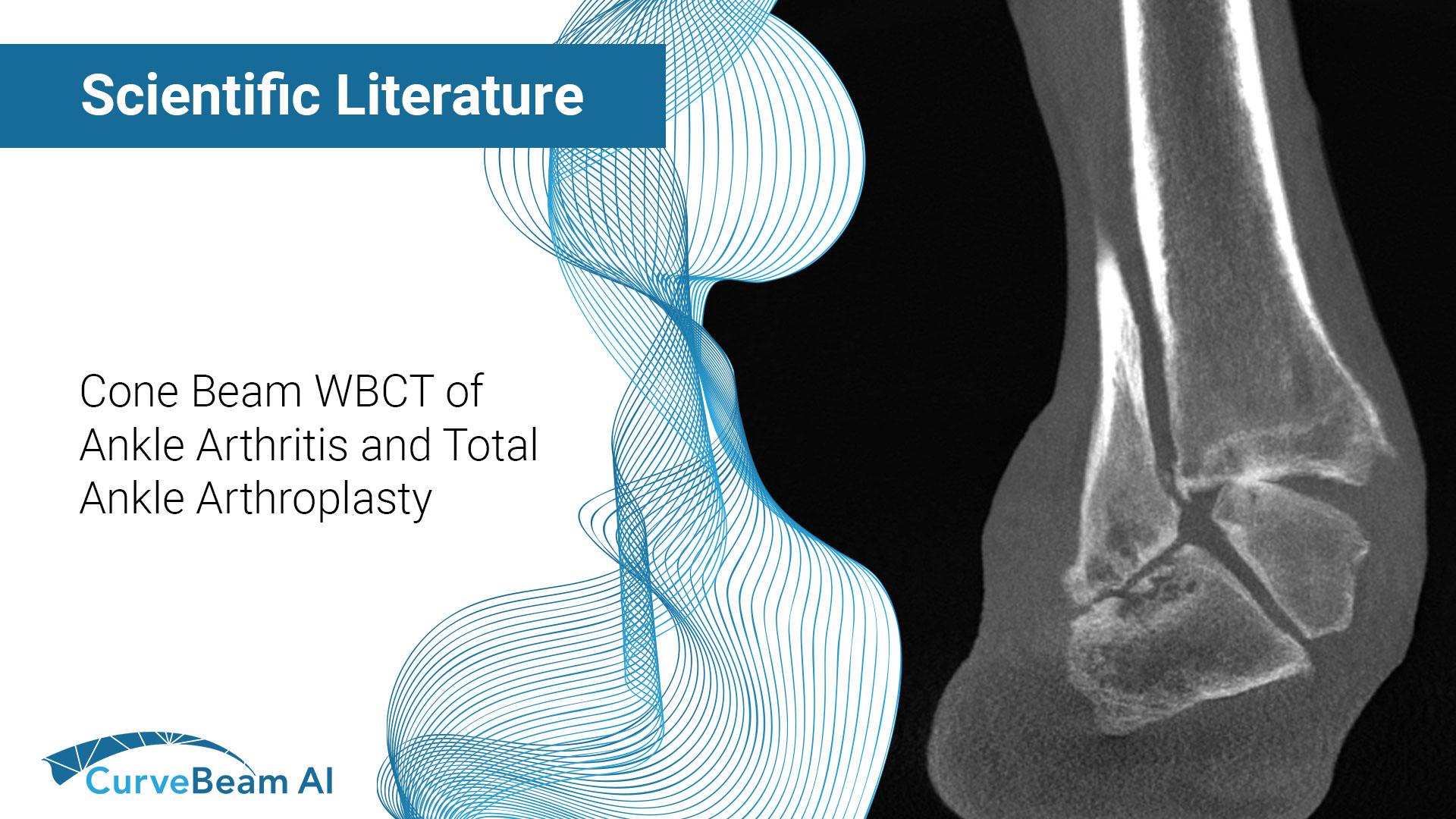
Cone Beam Weight Bearing Computed Tomography of Ankle Arthritis and Total Ankle Arthroplasty
Key Points: Cone Beam CT (CBCT), specifically WBCT three-dimensional (3D) visualization approach allows for a…
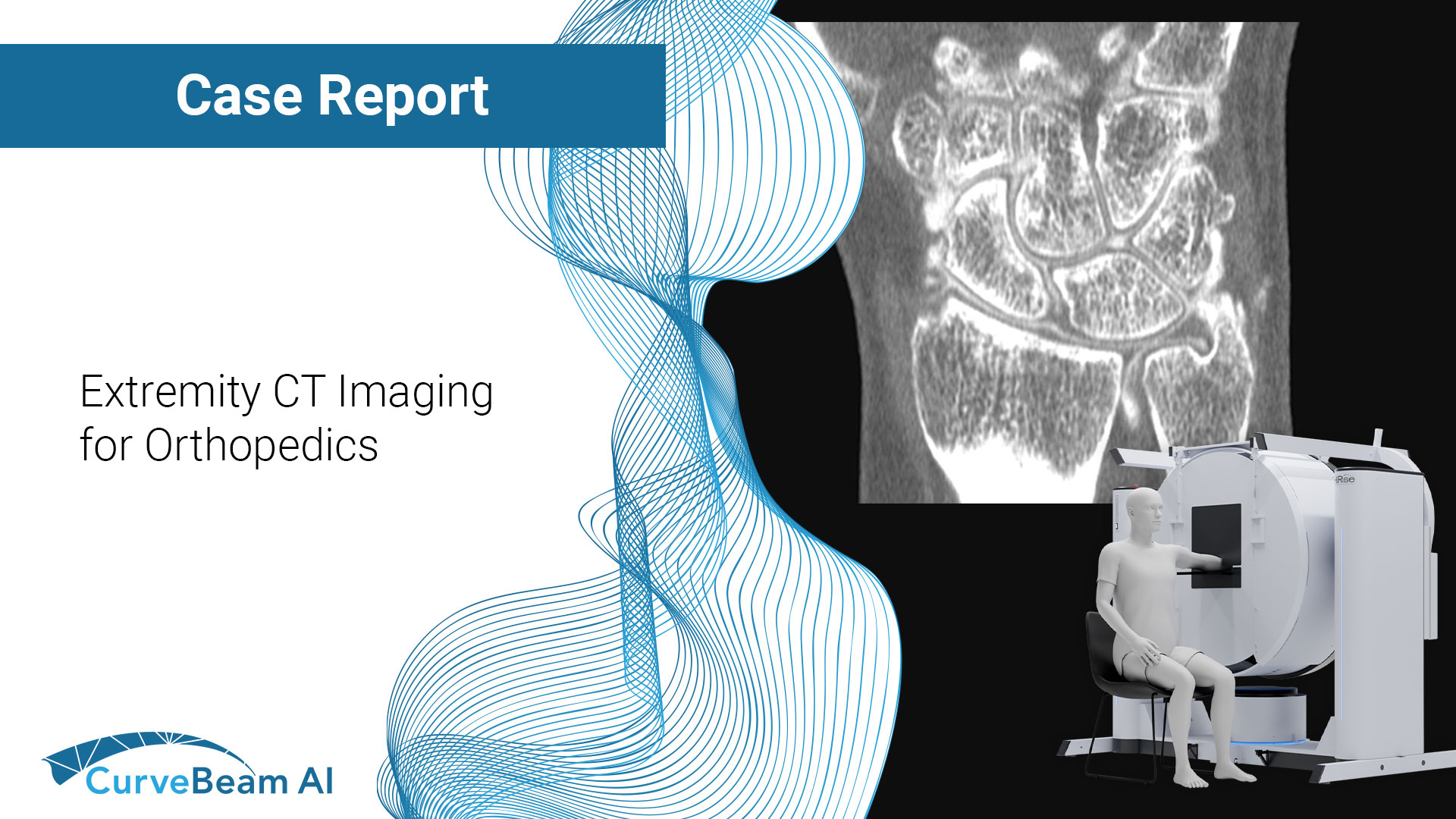
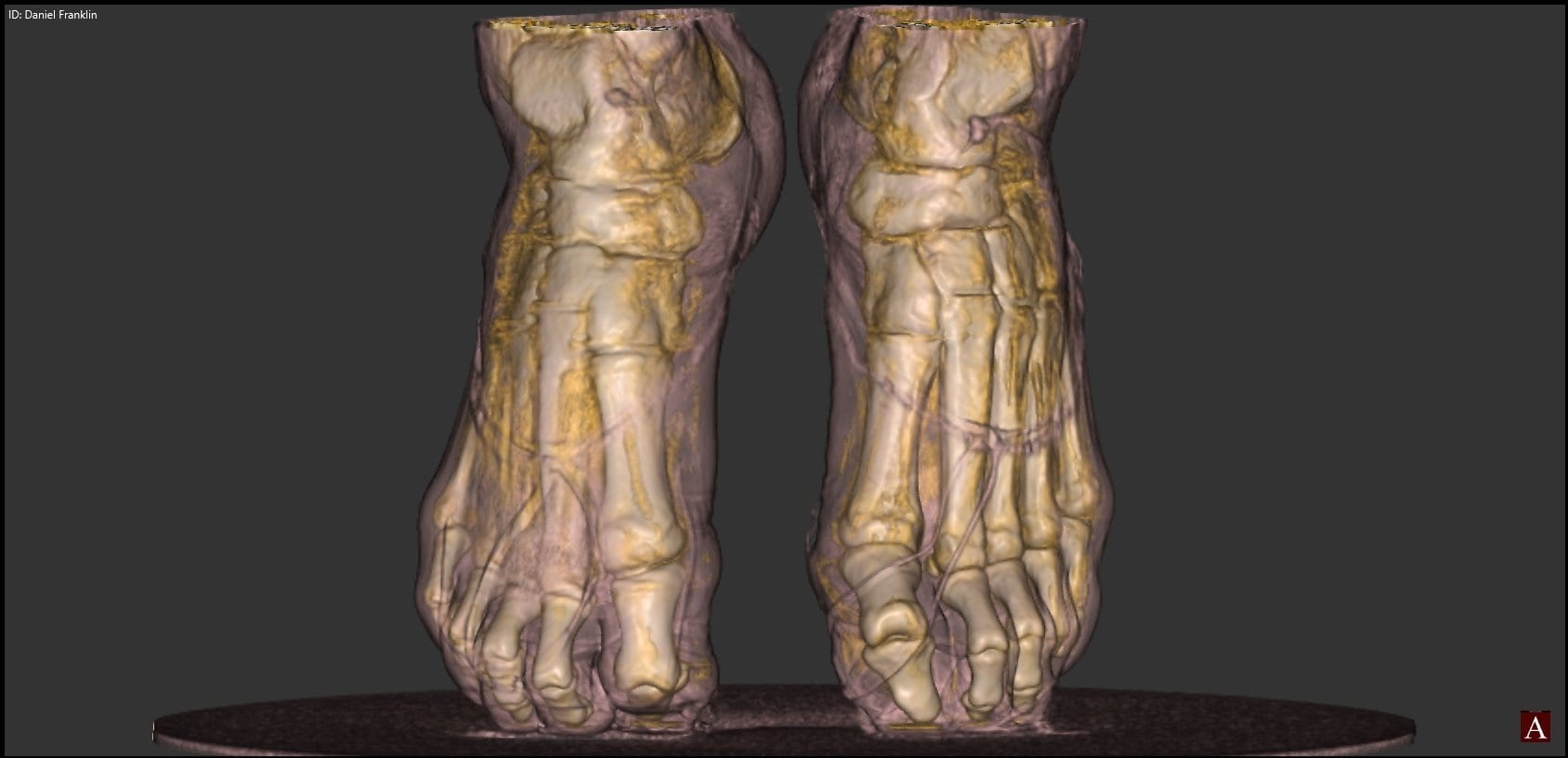
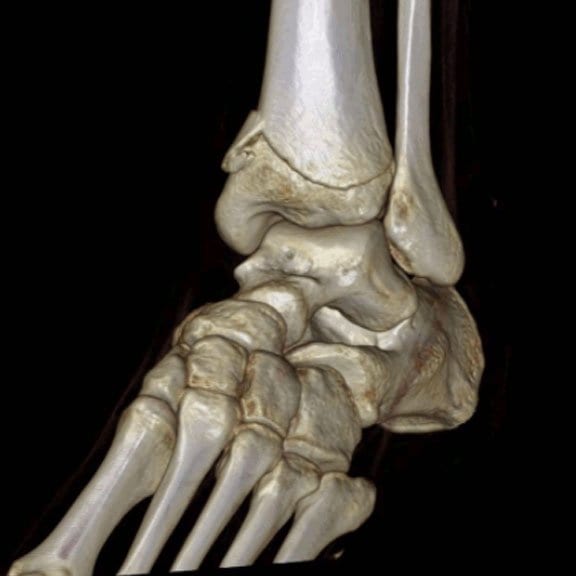
Extremity CT Imaging for Orthopedics
The HiRise is a low dose cone beam CT system. The upper extremity scans are…
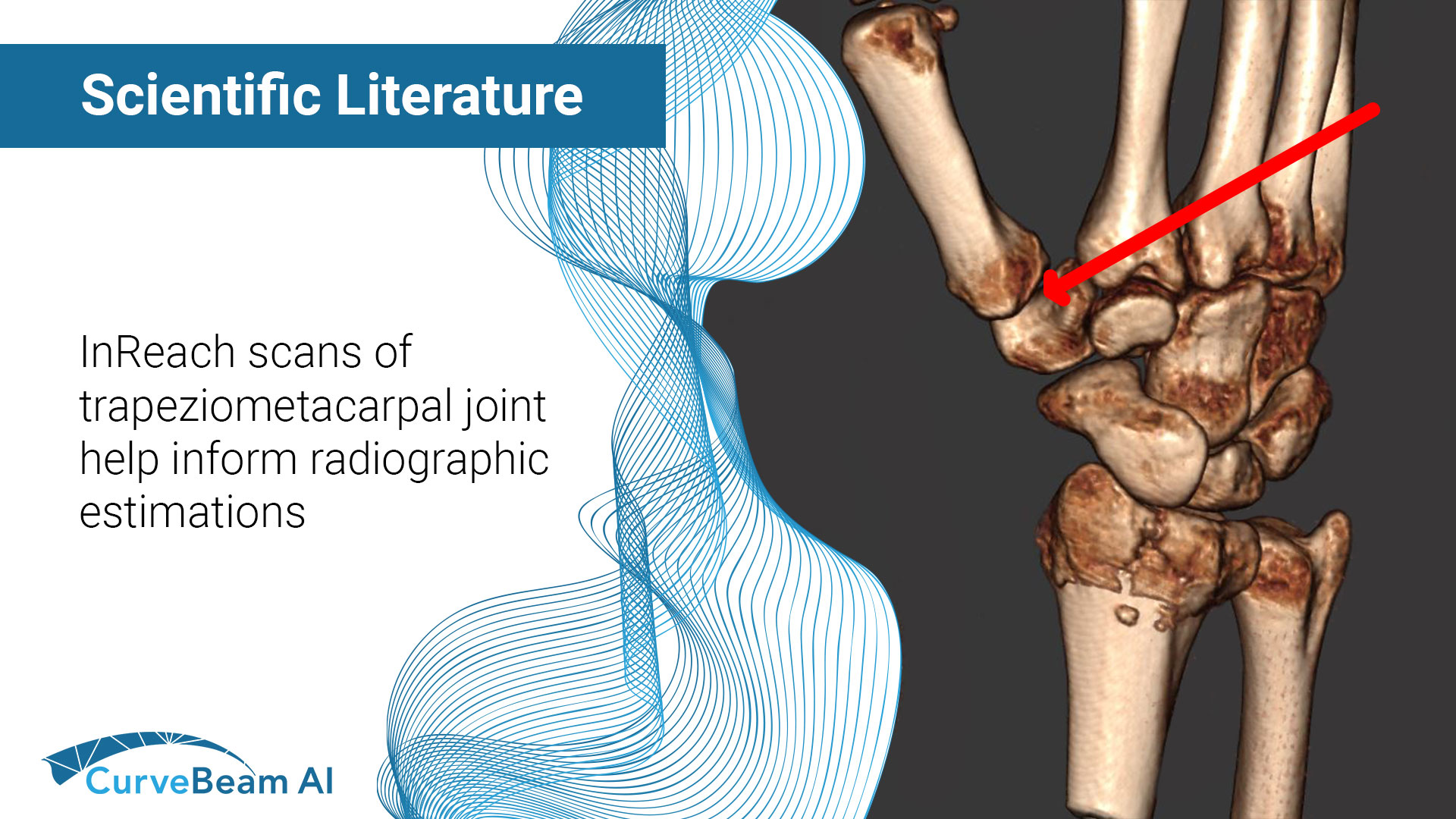
Estimation of Radiographic Joint Space of the Trapeziometacarpal Joint with Computed Tomographic Validation
Key Points: Cone Beam Computed Tomography (CBCT), a three-dimensional (3D) imaging modality, can readily evaluate…
CurveBeam AI Cast: How InReach CT has Improved Workflow at the University of Arizona
The integration of cone beam CT imaging in research labs is revolutionizing orthopedic investigations. Dr…
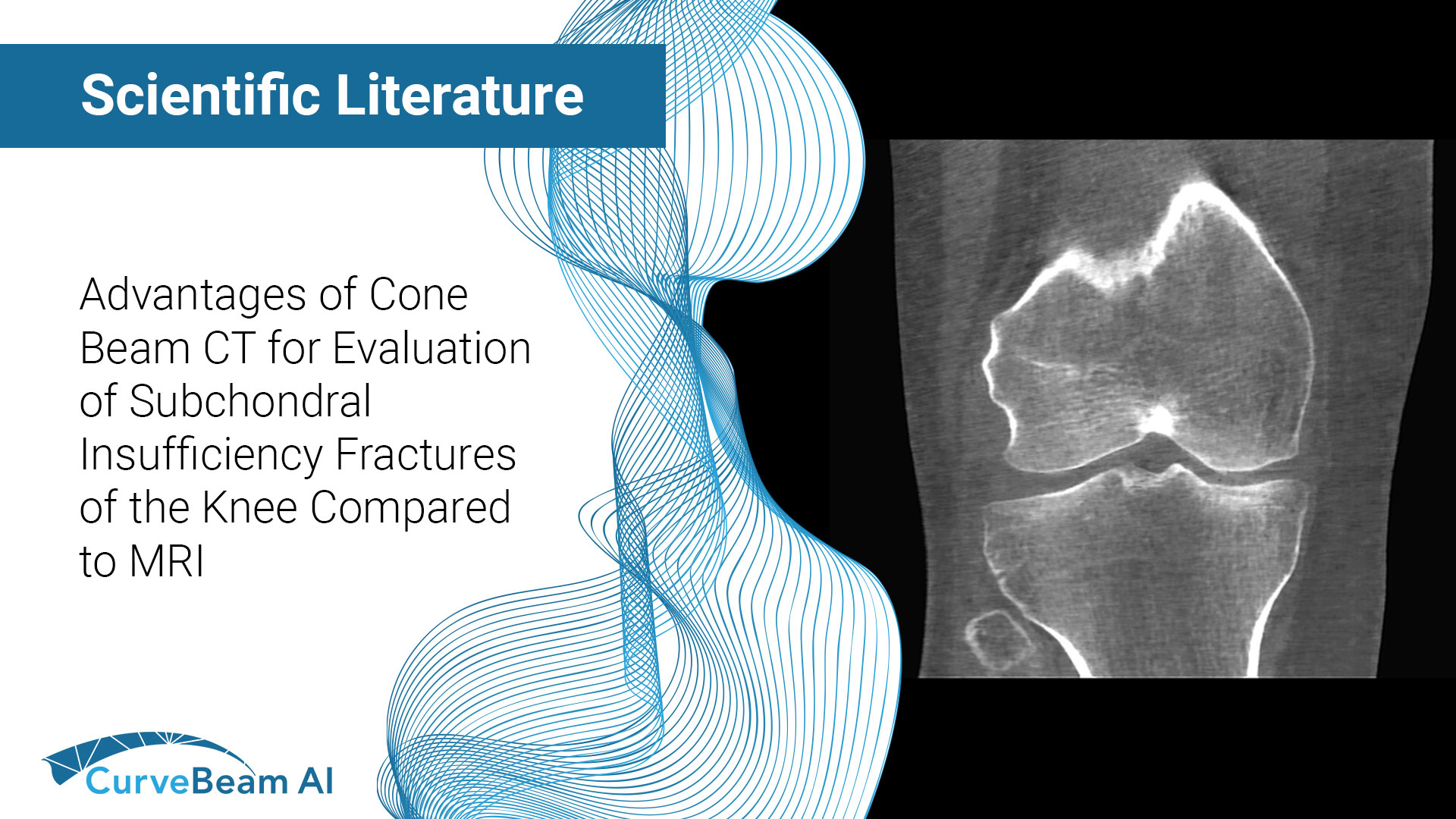
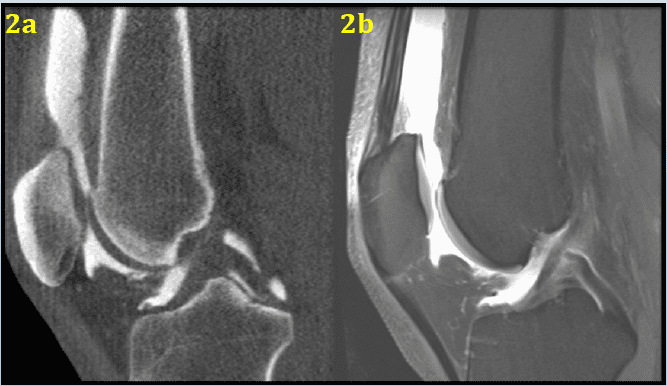
Advantages of Cone Beam Computed Tomography for Evaluation of Subchondral Insufficiency Fractures of the Knee Compared to MRI
Key Points: Cone Beam CT (CBCT) is superior in assessing bony structures compared to magnetic…
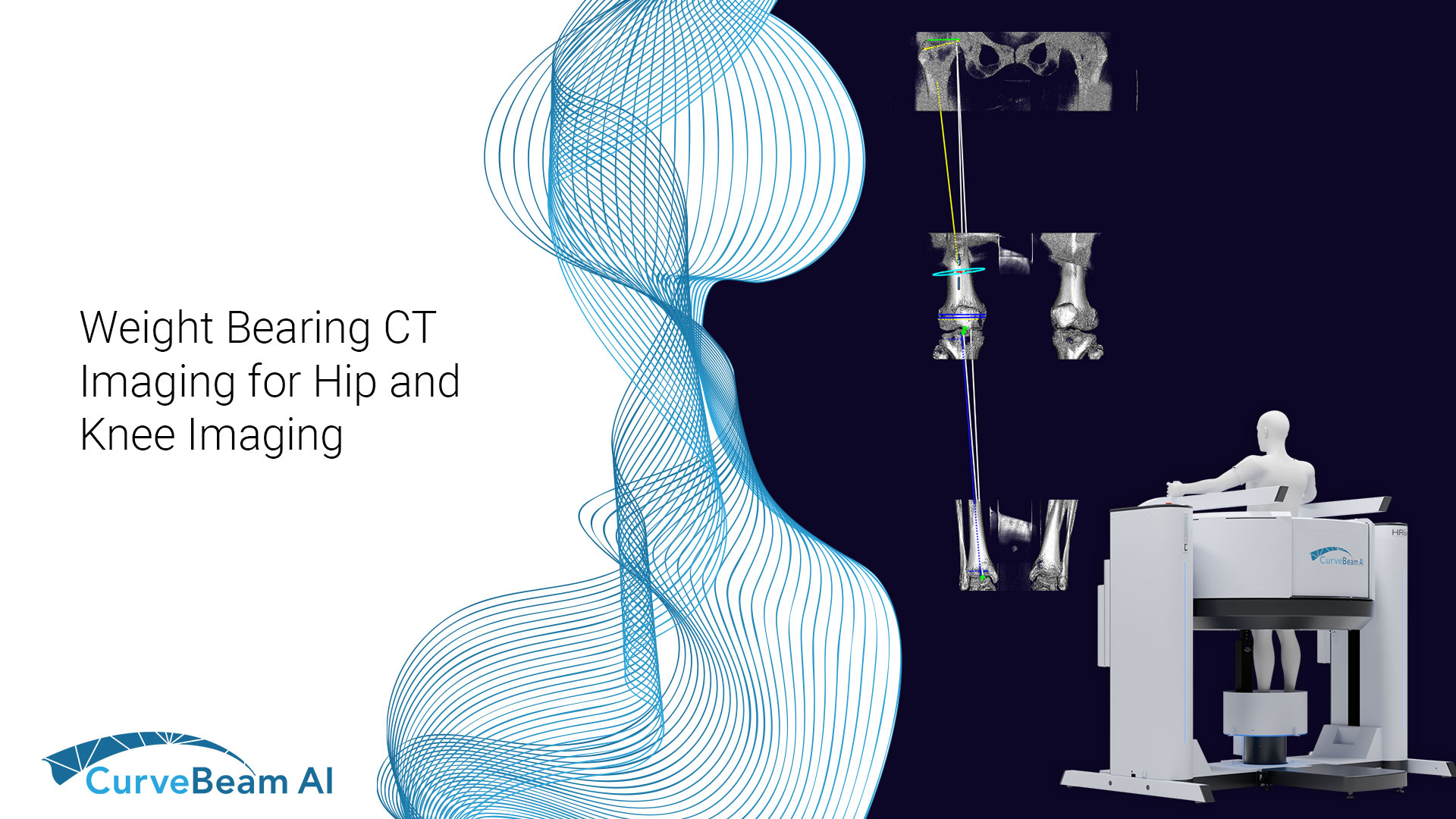
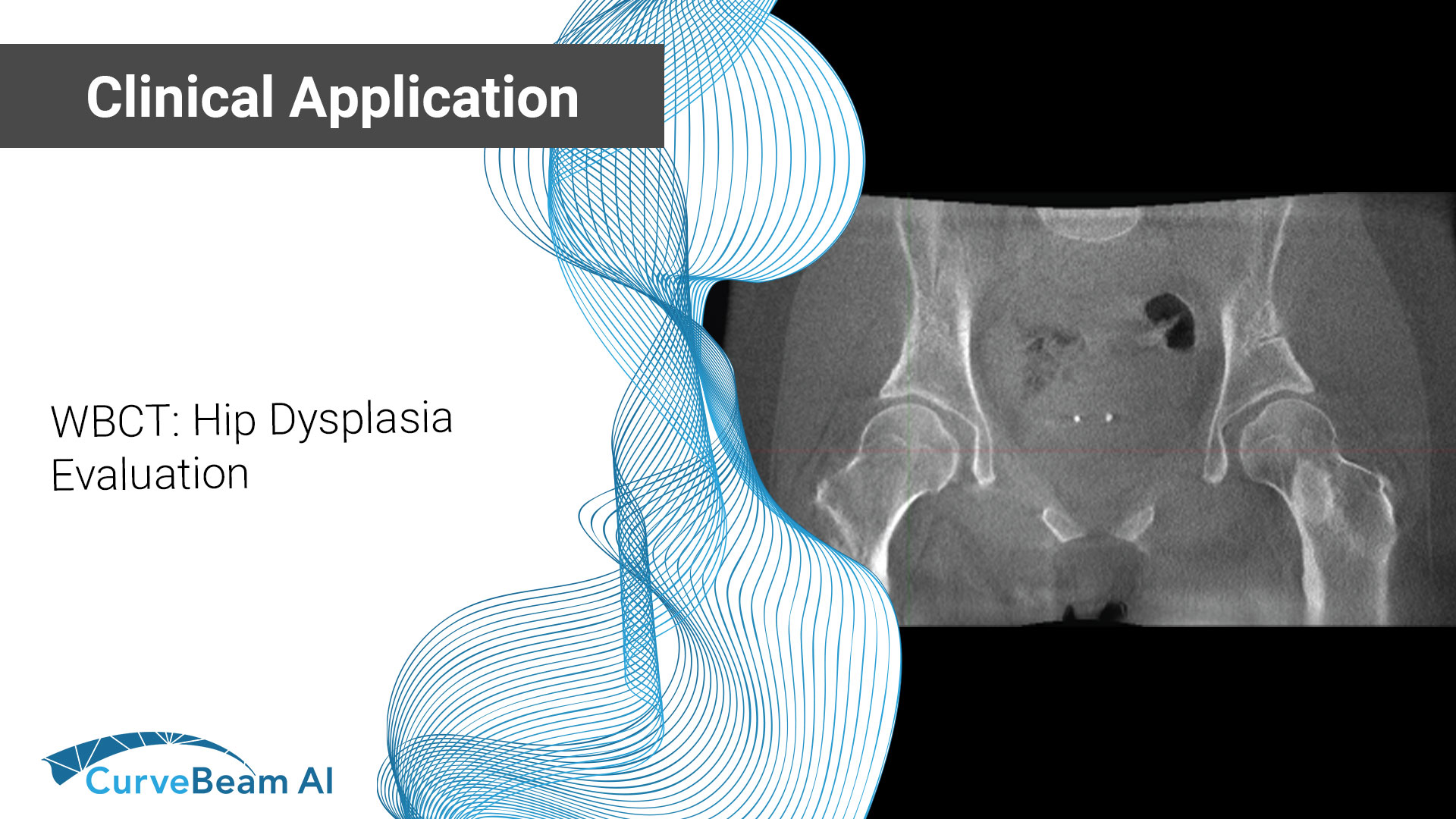
WBCT: Hip Dysplasia Evaluation
There is a new era in hip dysplasia evaluation when it comes to advanced imaging…
Automation Improves the Efficiency of Weight Bearing CT Scan 3D Volumetric Assessments of the Syndesmosis
Key Points: WBCT 3D Measurements volumetric measurements of the syndesmosis have shown to have a…
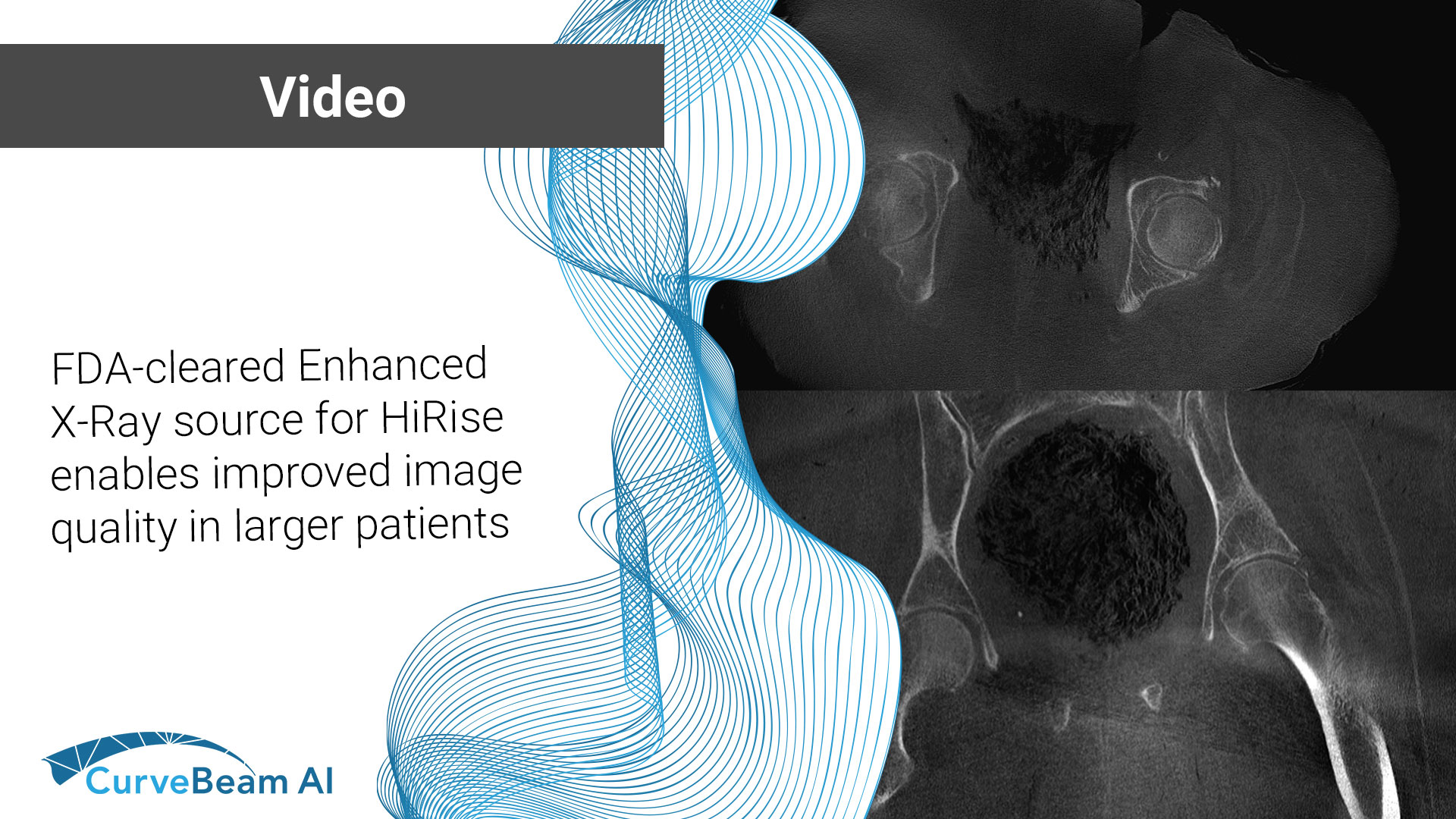
FDA-cleared Enhanced X-Ray source for HiRise enables improved image quality in larger patients
CurveBeam AI recently announced FDA clearance for an enhanced X-Ray source for the HiRise weight…
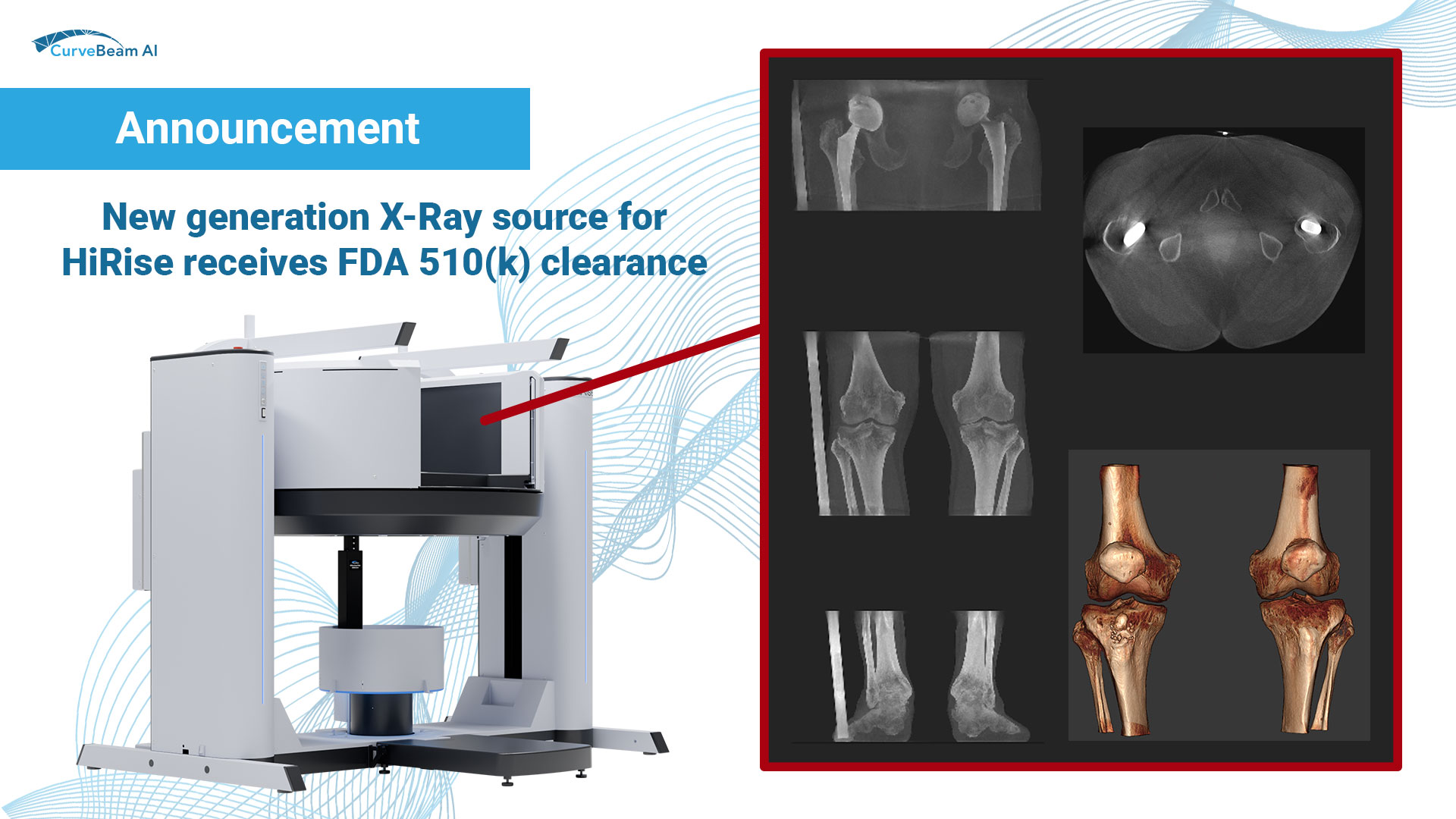
New generation X-Ray source for HiRise receives FDA 510(k) clearance
CBAI is proud to announce it has received FDA 510(k) clearance for enhancements to the…
on-site weight bearing CT system at Optim Orthopedics
Lisa Smith, MHA, OTR/L, CHT, CHC, Chief Operating Officer for Optim Orthopedics in Savannah, Georgia,…
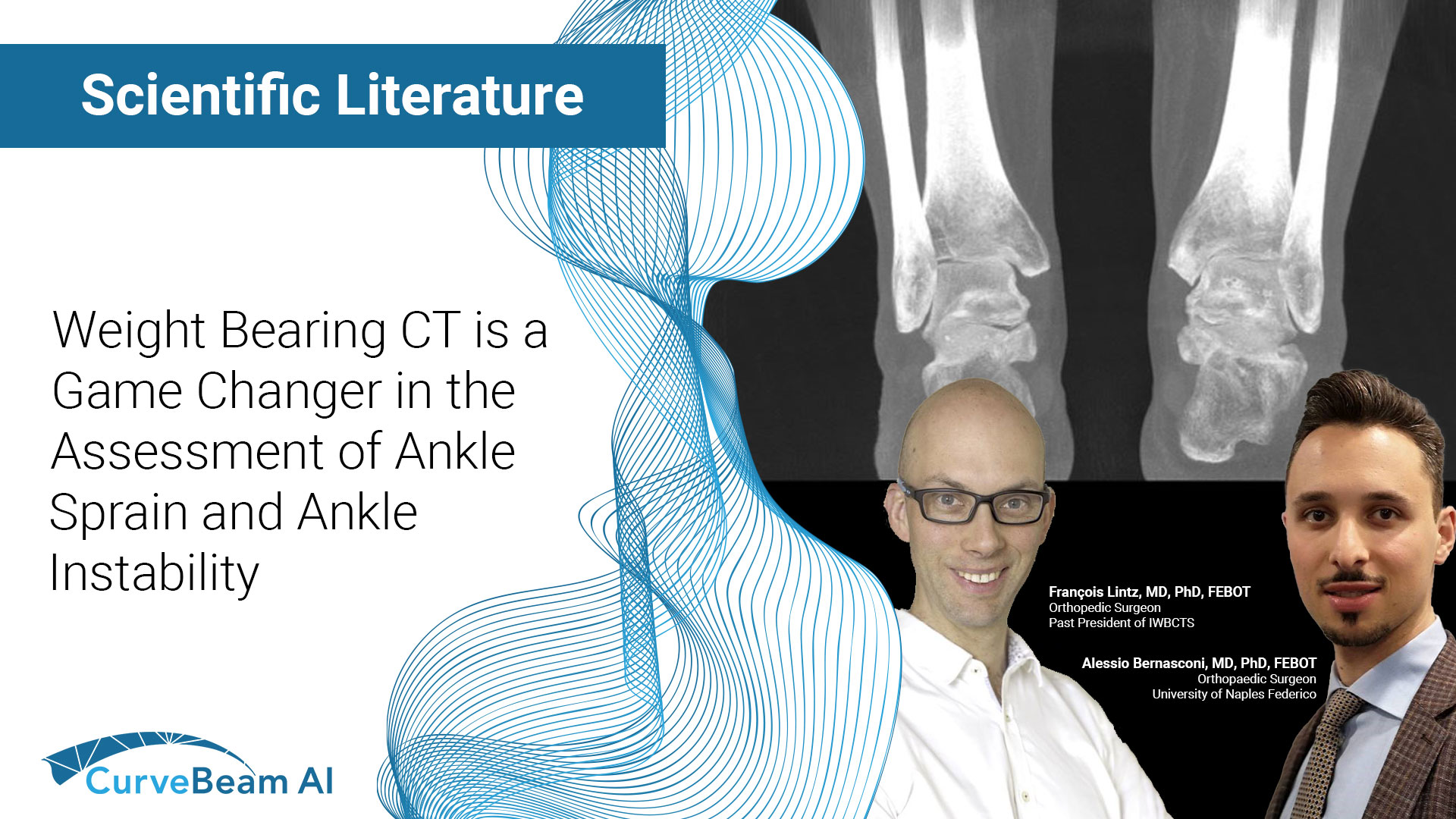
Can WBCT Be a Game-Changer in the Assessment of Ankle Sprain and Ankle Instability?
Key Points: The most important advantage of weight bearing CT (WBCT), which utilizes cone beam…
The Impact of On-Site Weight Bearing CT at Tennessee Orthopedic Alliance
Jeff McPherson, MBA, FACMPE, East Tennessee Market President, Tennessee Orthopedic Alliance, highlights says weight bearing…
Assessment of Knee Instability in ACL-Injured Knees Using Weight Bearing Computed Tomography
Key Points: Anterior tibial translation (ATT) and femorotibial rotation (FTR) are the two most common…
Dr. Sudheer Reddy Explores the Essential Uses of Weight Bearing CT in Orthopaedics
Weight Bearing CT can help to illuminate the patient’s injury and deformities in a better…
The Advantage of CBCT in Short Staffing Challenges for MSK Imaging
Clinic short staffing in medical imaging departments presents a challenge, impacting patient care, patient leakage,…
Duke Health’s HiRise Weight Bearing CT Sets the Standard in Orthopedic Care
Duke Health celebrates success in patient care after adding a HiRise weight bearing CT (WBCT)…
Case Study: Augmented Stress Weight Bearing CT for Evaluation of Subtle Lisfranc Injuries in Elite Athletes
Key Points: Subtle Lisfranc injuries often go undiagnosed which can lead to devastating consequences and…
Seeing Beyond X-Rays: The Power of Weight Bearing CT Scans in Patient Care
In patient evaluations, doctors typically rely on physical exams and weight bearing x-rays to assess…
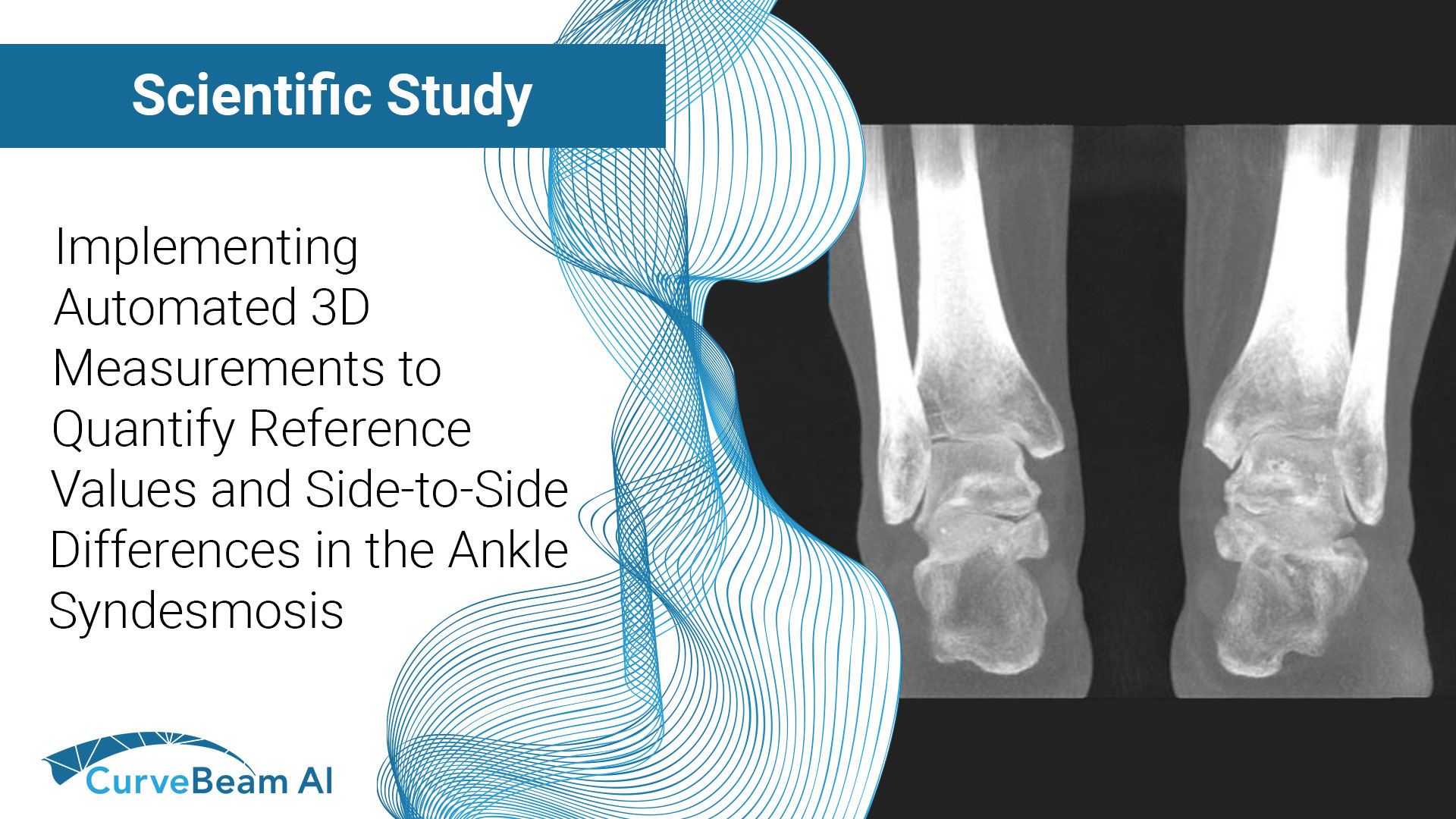
Implementing Automated 3D Measurements to Quantify Reference Values and Side-to-Side Differences in the Ankle Syndesmosis
Key Points: Currently plain radiographs are the standard method in diagnosing syndesmotic ankle injuries even…
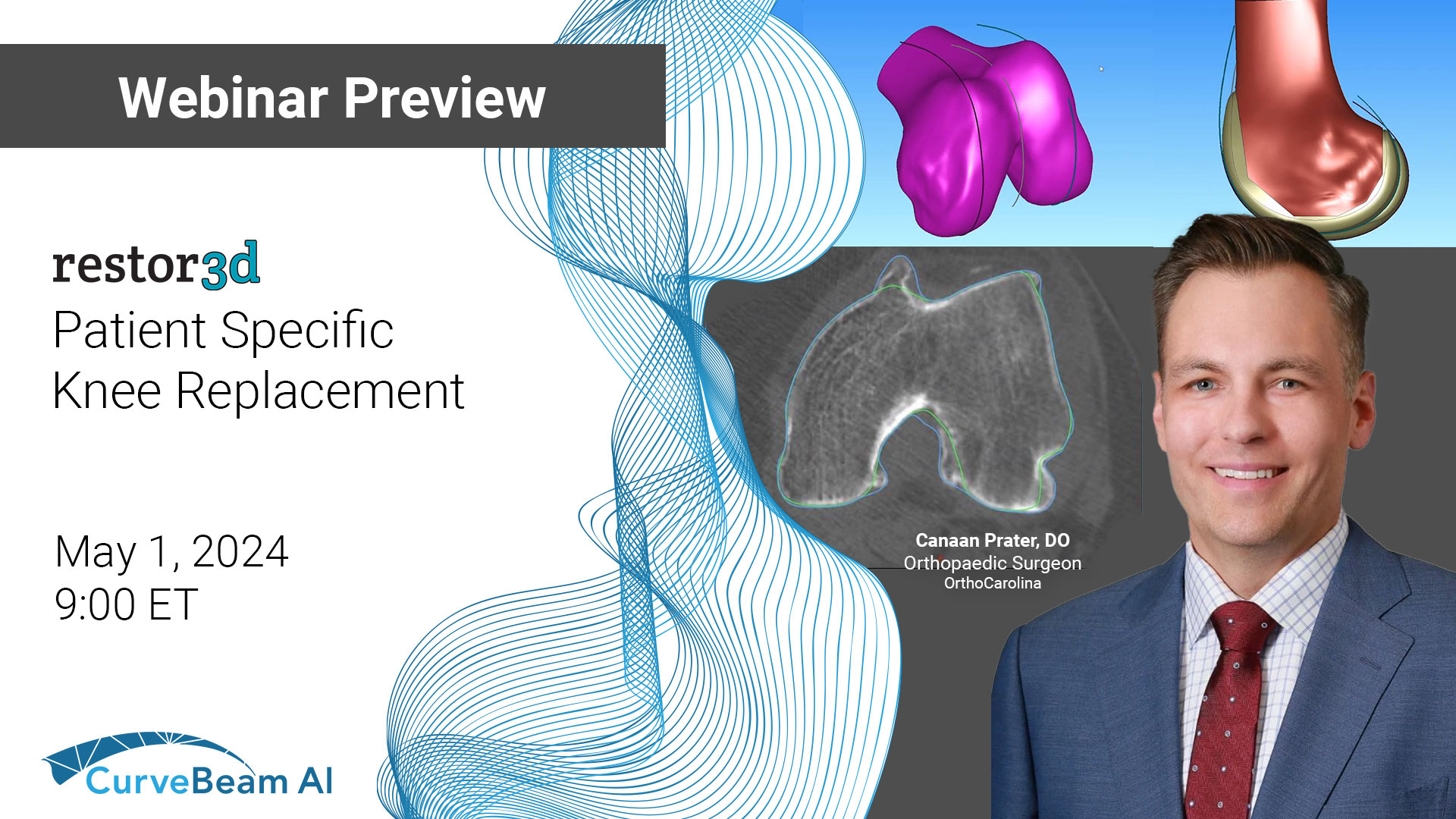
Webinar Preview: Using HiRise WBCT to Inform Surgical Planning of Patient-Specific Total Knee Replacement
Join us to hear firsthand from Dr. Prater as he shares insights on executing the…
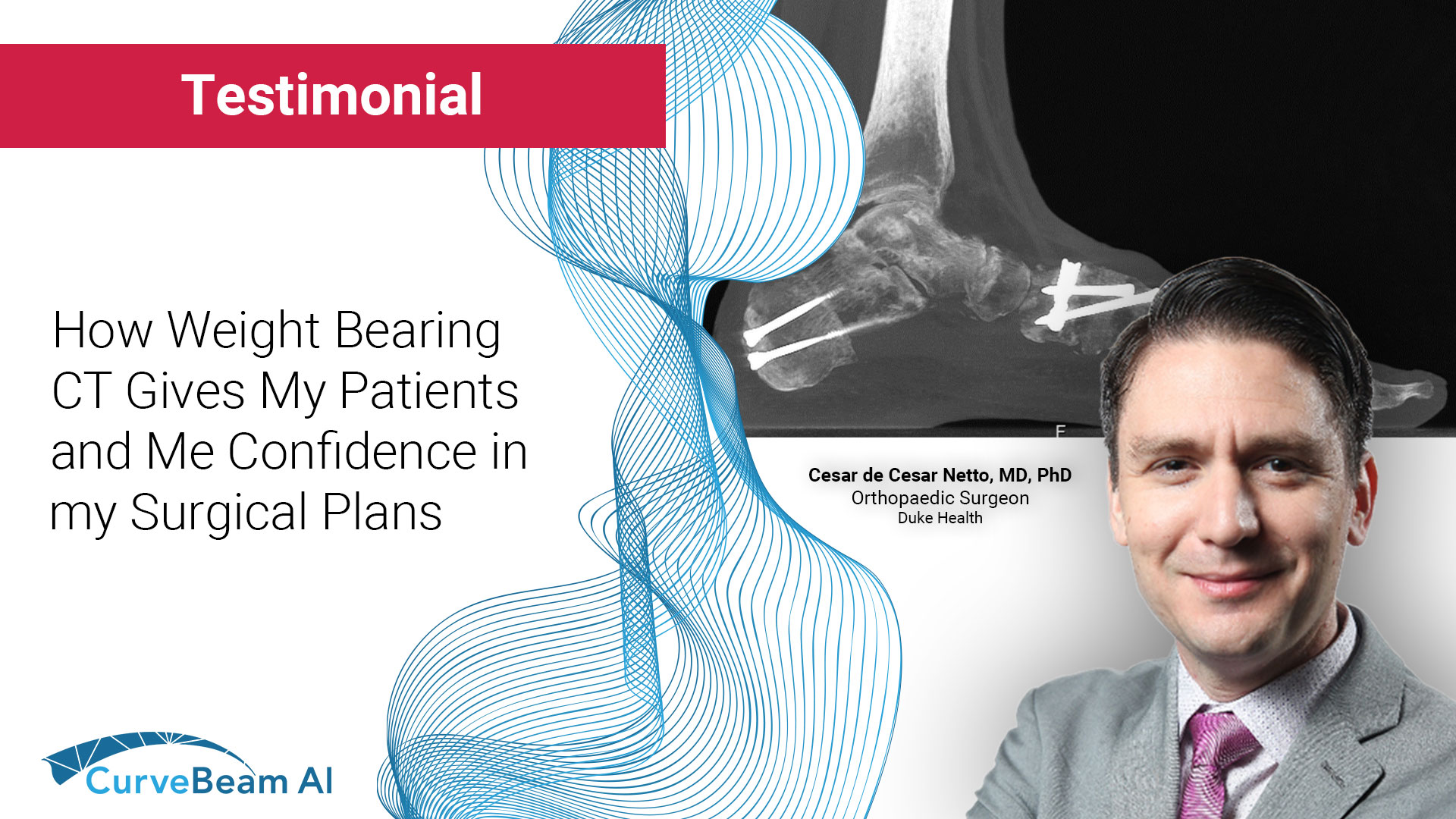
How WBCT Gives Dr. Netto and His Patients Confidence in His Surgical Plans
Weight bearing CT (WBCT) allows the patient to stand upright while a gantry rotates around…
New Cone Beam CT Imaging Orthopedics Course Approved for ASRT Continuing Education Credits
Radvant Education, a continuing education platform for healthcare professionals, announced it has launched a new…
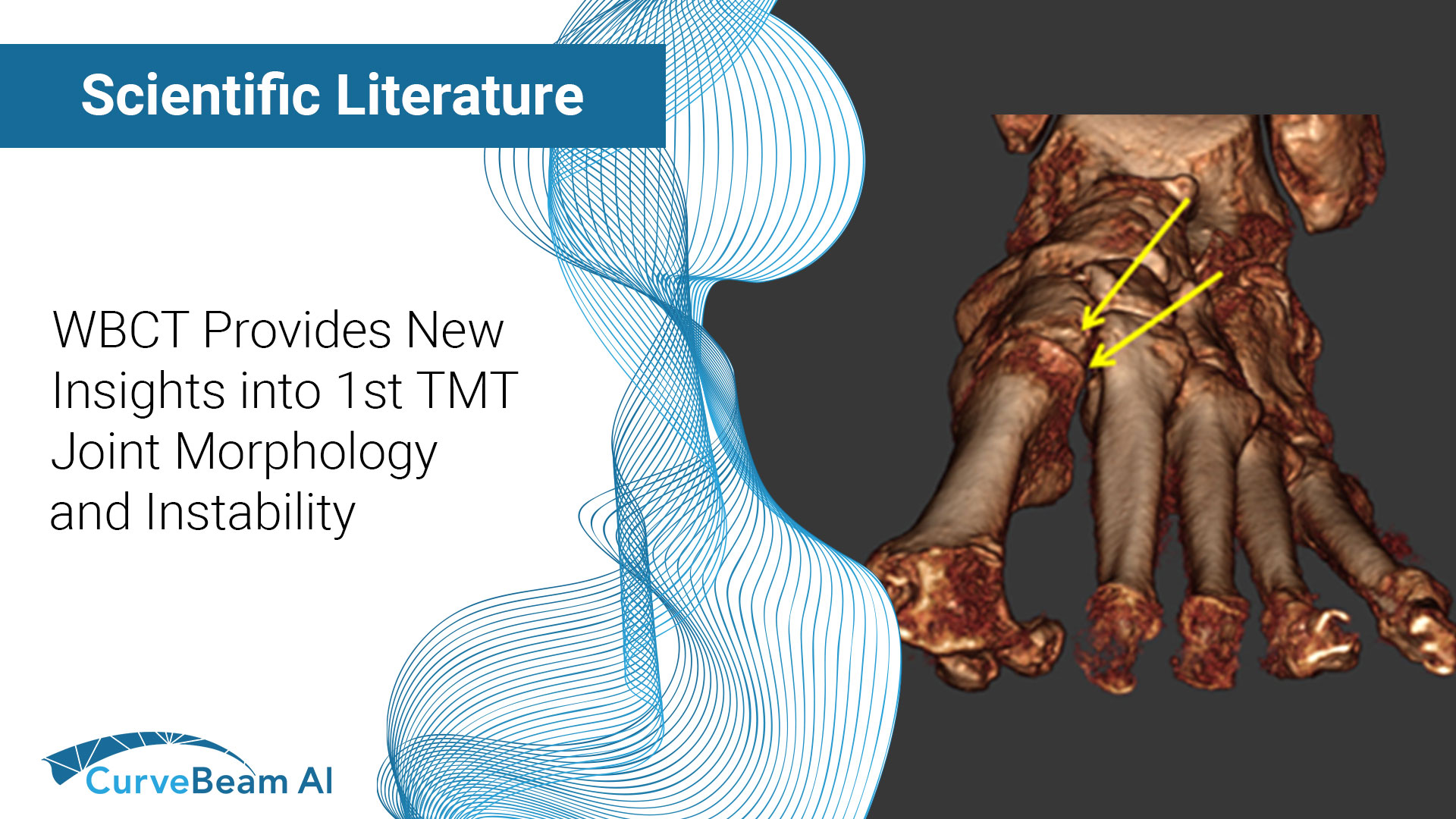
Case-Control Study: The Role of First Tarsometatarsal Joint Morphology and Instability in the Etiology of Hallux Valgus
Key Points: First tarsometatarsal joint (TMT1) hypermobility is associated with hallux valgus (HV) which is…
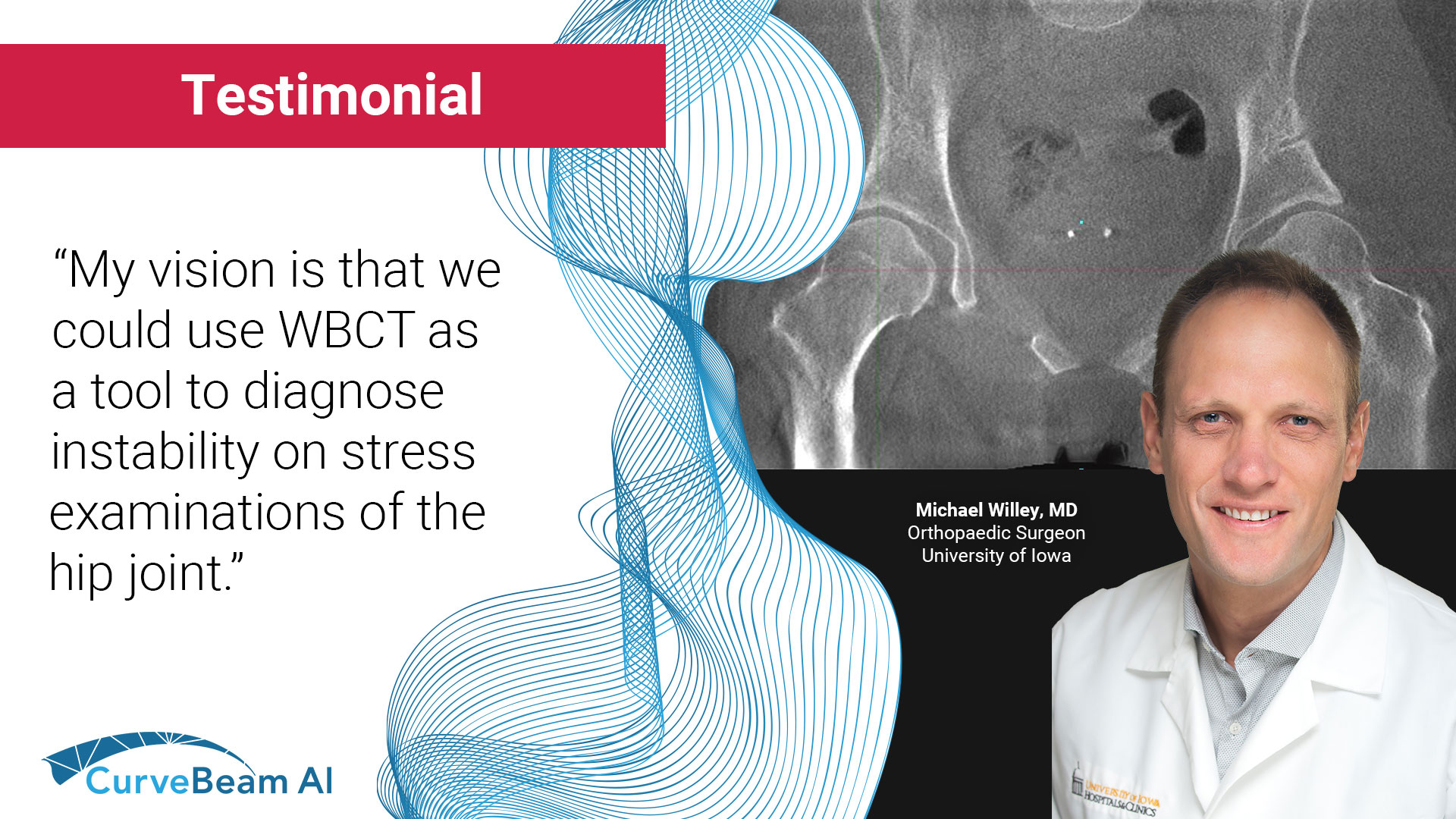
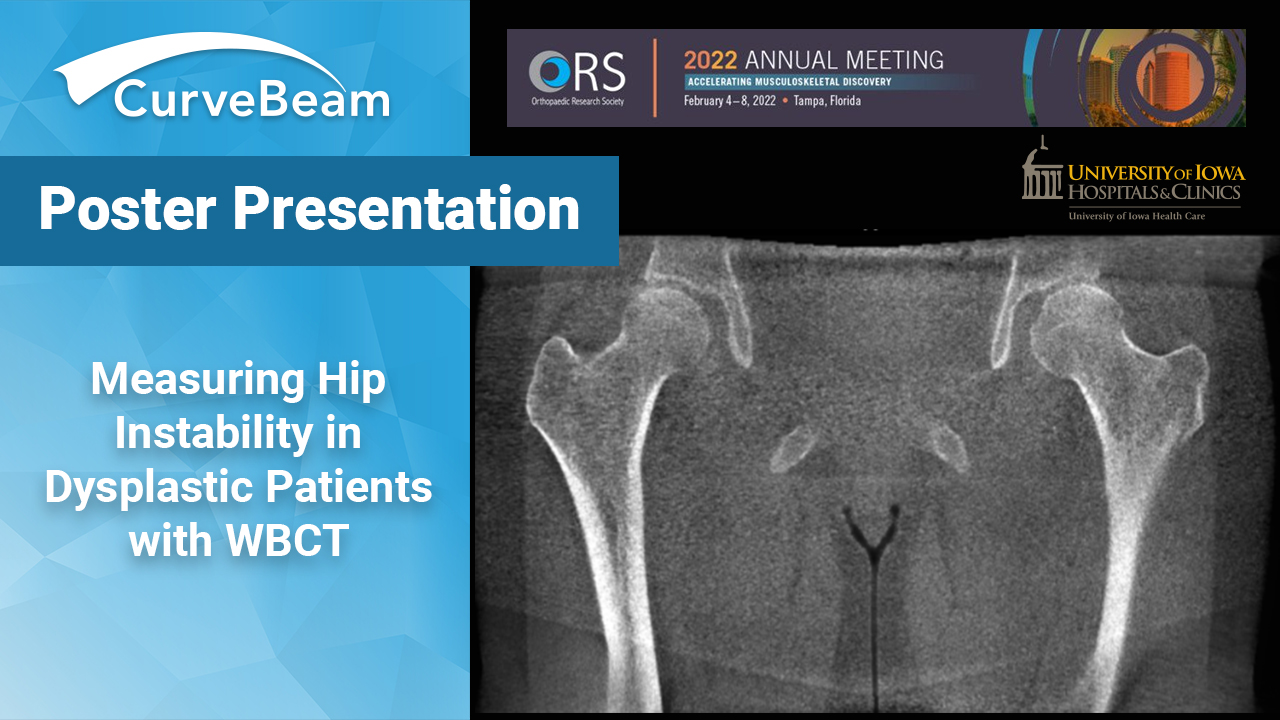
Measuring Hip Instability in Dysplastic Patients with WBCT
Hip dysplasia is a complex deformity of the acetabulum and femoral head that results in…
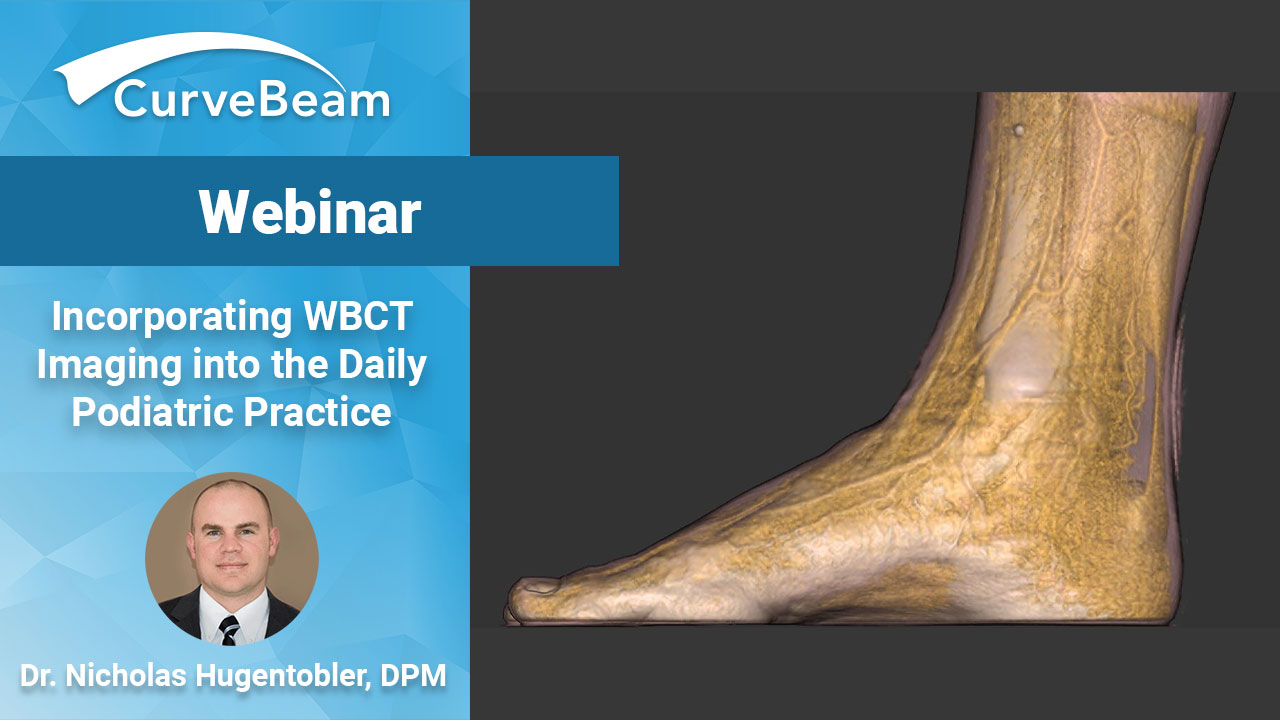
Hugentobler: pedCAT Improves Practice Workflow
It is feasible for a single-practitioner podiatry practice to add weight bearing CT (WBCT) imaging and realize economical…
First Knee Replacement Surgery Planned with Weight Bearing CT Imaging Successfully Completed
HATFIELD, Penn. – March 19, 2024 – CurveBeam AI, Ltd., the leader in weight bearing…
Redefining Patient Care with Cone Beam CT
"Cone beam CT reduces the barriers for orthopedic practices to offer CT imaging in-house," says…
CurveBeam AI’s AI-Powered Foot & Ankle Measurements Have Good Reliability Compared to Manual Methods
Dr. Prof. Martinus Richter et al published a study in the March 2024 issue of…
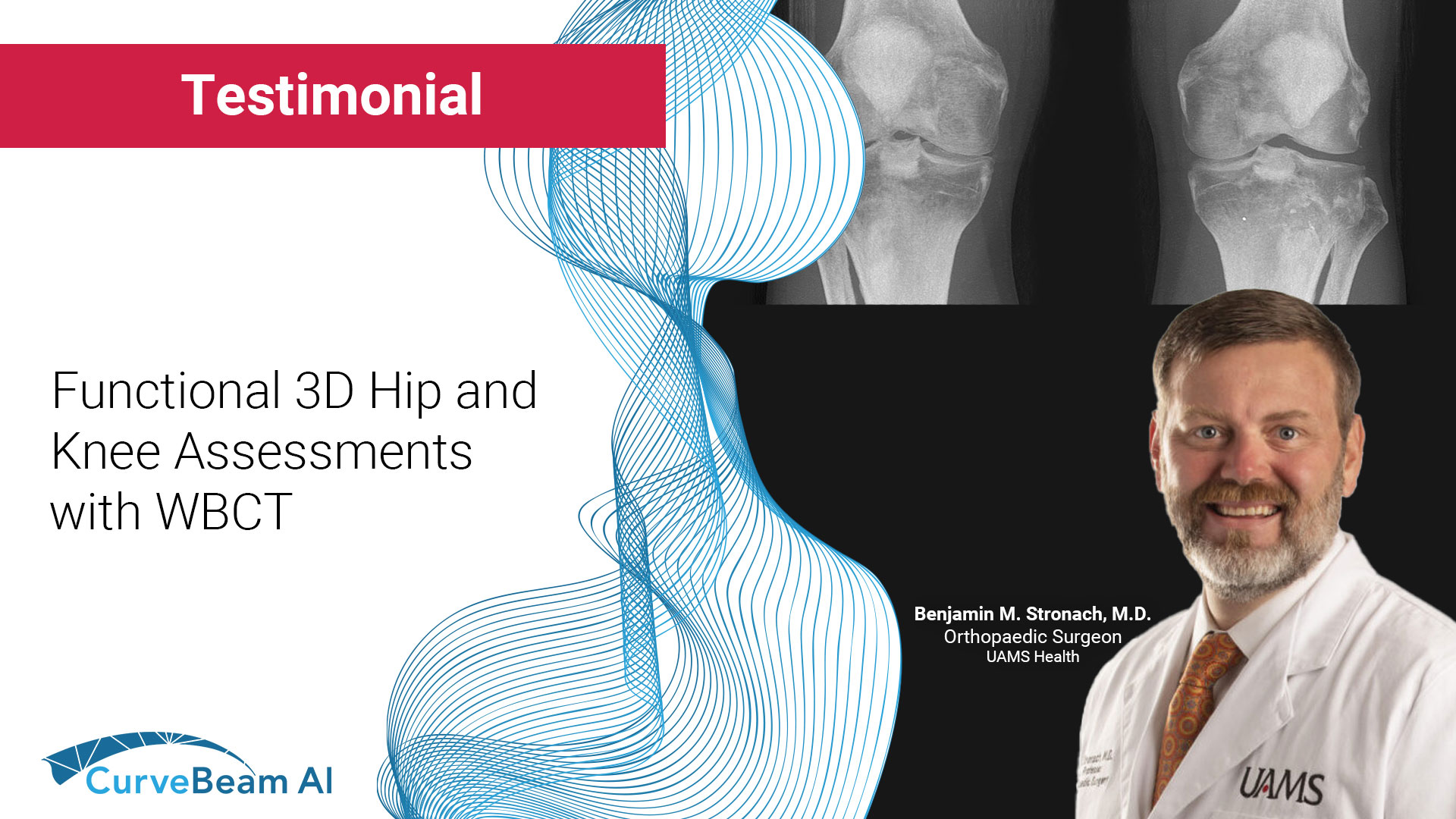
Functional Hip & Knee Assessment with WBCT
With Weight Bearing CT you can gain an increased understanding of the functional aspects of…
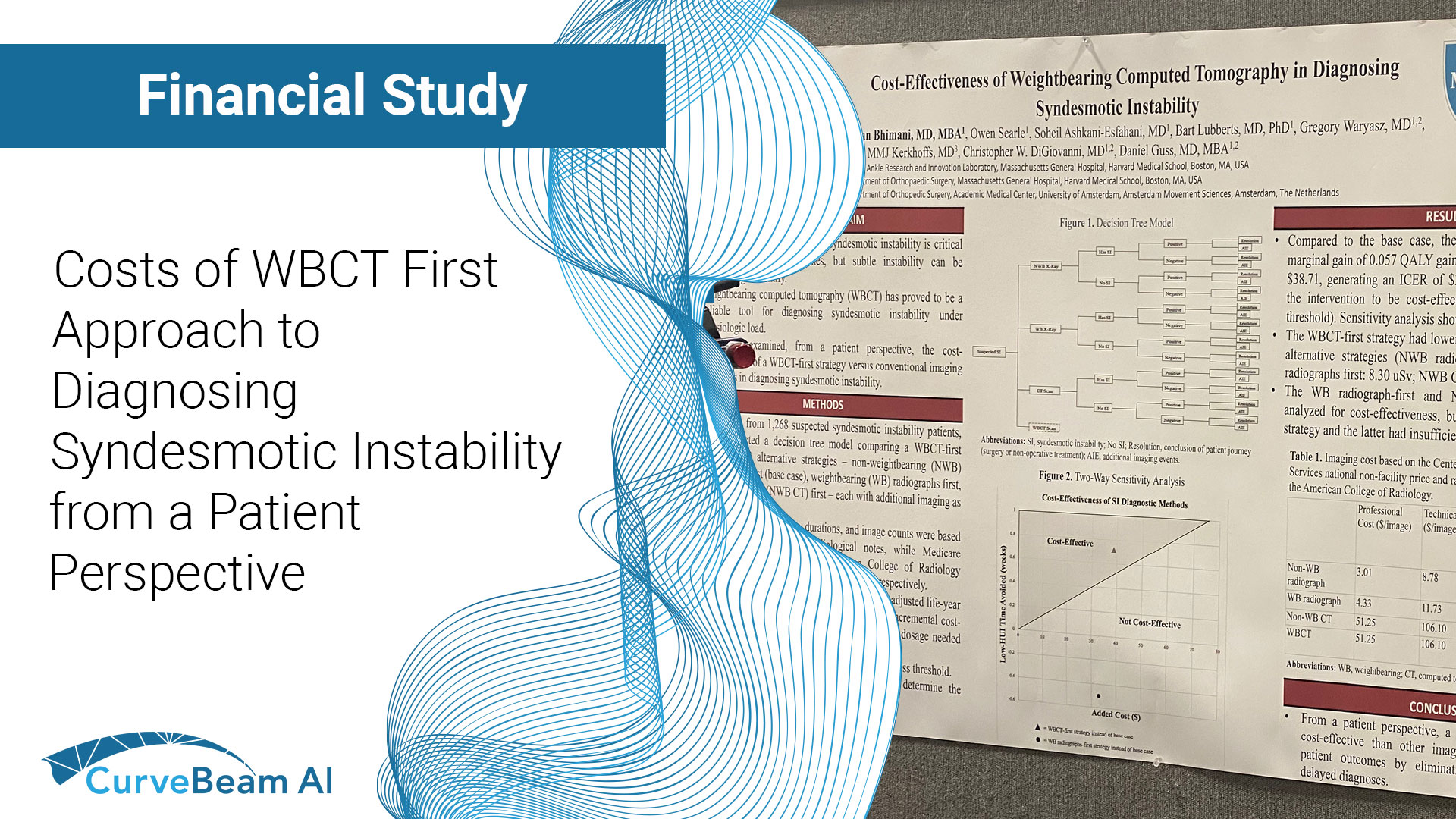
Costs of WBCT-First Approach to Diagnosing Syndesmotic Instability from a Patient Perspective
A weight bearing CT (WBCT)-first approach in diagnosing syndesmotic instability emerged as the most cost-effective…
Enhancing Patient Care: The Benefits of In Office Weight-Bearing CT
Weight bearing CT (WBCT) is a game changer in foot and ankle care. In-office access…
Decoding Ulnar Wrist Pain: Dr. Glenn Gaston’s Rapid Diagnosis
In a case study, Dr. Glenn Gaston, MD, studied a 19-year-old female patient presenting ulnar…
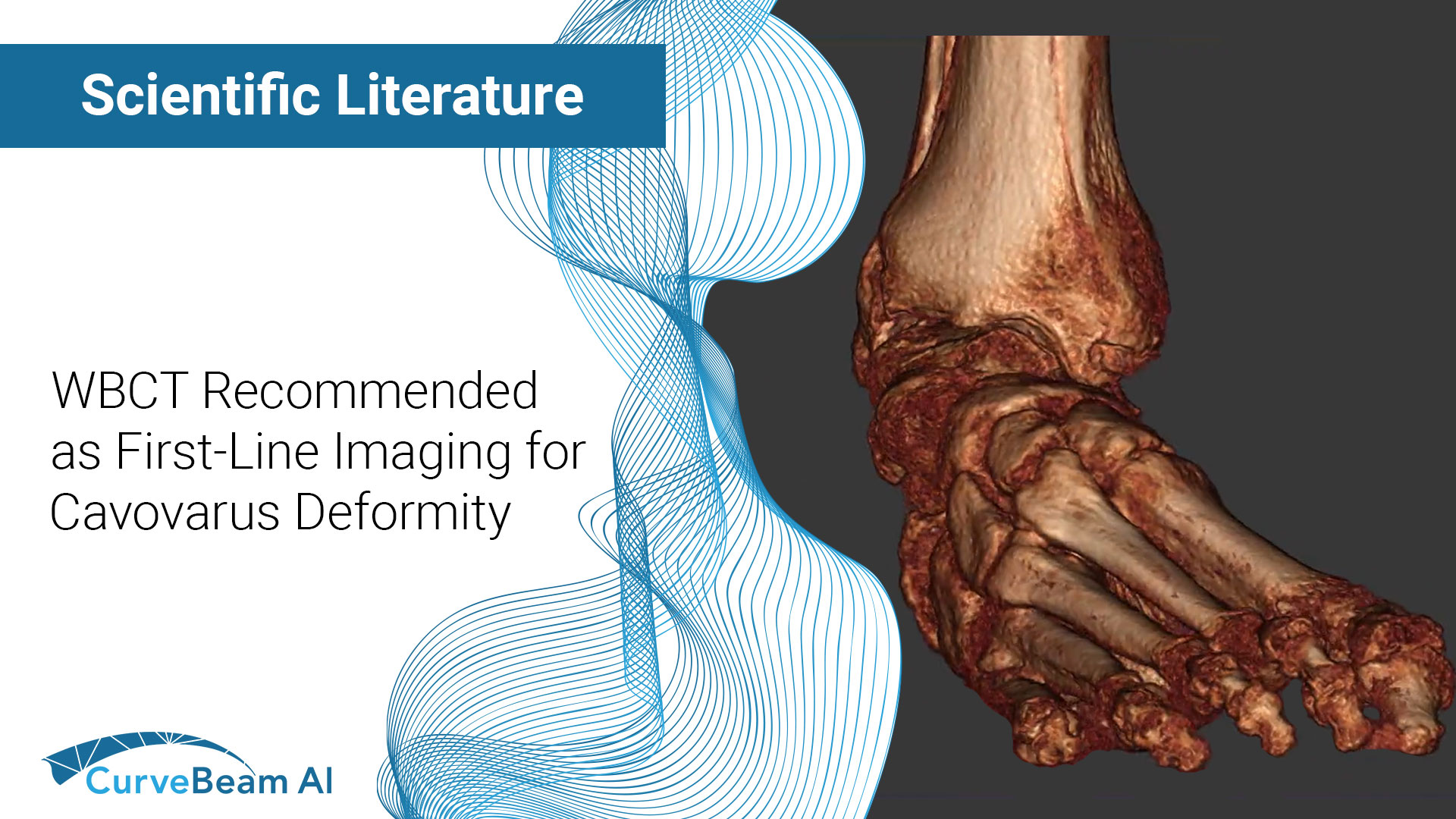
Bernasconi & Lintz: WBCT Should be First-Line Imaging for Cavovarus Deformity
Cavovarus deformity in the foot involves the forefoot, midfoot, hindfoot, and often the talus. Because…
CurveBeam AI Cast: The Evolution of Orthopedic Technology and PeekMed’ s Role in Streamlining Surgical Processes
Rapid advancements in surgical planning techniques are propelling the field of orthopedic technology into a…
Countdown the Top 5 of 2023 with CurveBeam AI
As we approach the end of this remarkable year, we want to take a moment to…
Planning Ankle Replacements using WBCT Seminar at EFAS
Highlight from IWBCTS session at EFAS in Madrid: Andy Goldberg discusses the importance of assessing…
CurveBeam AI Cast: An Entrepreneurial Physician in France Opens a Weight Bearing CT-Centered Private Clinic
In the latest CurveBeam AI Cast episode, recorded at the AOFAS Annual Meeting 2023, host…
Join the CurveBeam AI Connect Monthly Newsletter
Don't miss out on what is going on with weight bearing CT and all the…
WBCT is Helping Orthopedic Surgeons Understand Foot Deformities in Cerebral Palsy Patients
Weight bearing CT scans are helping us understand pediatric foot deformities better than ever before…
WBCT Shines Light on Foot Deformities in Pediatric Patients with Pes Planus
There’s a lot about pediatric pes planus, or flexible flat foot deformity, that orthopedic surgeons…
Stay Ahead Of The Curve With CurveBeam AI
At CurveBeam AI, we strive to stay ahead of the curve. We are thrilled to…
Beyond Traditional Imaging: Dr. Blake Moore on Weight-Bearing CT Scans
The introduction of weight-bearing CT scans has revolutionized how surgeons assess and treat various orthopedic…
Using WBCT to Explore Patient Specific Knee Kinematics
Most recently, CurveBeam AI and Vent Creativity explored the use of weight bearing CT (WBCT)…
Doug Chalker, BS R.T.(R)(CT): A Journey Through Radiologic Technology
In the realm of radiologic technology, there are professionals who stand out not just for…
Honoring Medical Imaging Professionals: CurveBeam AI Celebrates National Radiologic Technology Week® 2023
Every year, radiologic technologists come together to celebrate the tireless efforts and dedication of medical…
Reducing Patient Leakage with On Site Imaging
Leakage in healthcare can be a significant challenge, and it's important to address it effectively…
CurveBeam AI Cast: A look into The Benefits of Weight Bearing CT with Dr. Francois Lintz Part 2
The benefits of Weight Bearing CT (WBCT) usher in a new era of orthopedic diagnostics,…
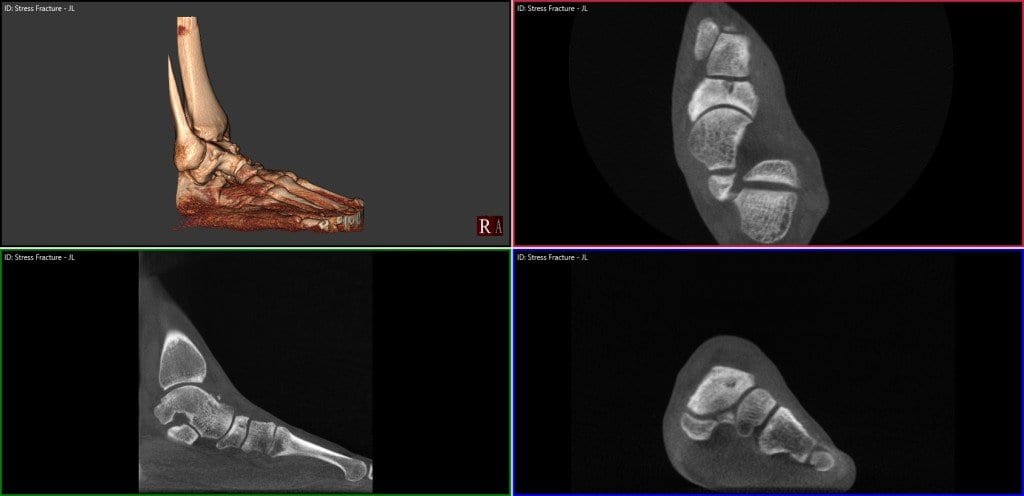
Mayo Clinic Case Report: WBCT Reveals Tibial Plafond Stress Fracture in Competitive Athlete
Key Points: Stress fractures should always be considered in athletes with ankle pain. This is…
CurveBeam AI Cast: A look into The Benefits of Weight Bearing CT with Dr. Francois Lintz
In recent years, the orthopedics field has been gravitating towards using 3D imaging technologies to…
Testimonial: Weight Bearing CT Can Help Pinpoint the Diagnosis
Weight bearing CT (WBCT) imaging has advantages over traditional forms of imaging such as X-Ray…
IWBCTS Session on End Stage Ankle Arthritis with a Focus on TAR
The International Weight Bearing CT Society (IWBCTS) had a session on the use of weight…
Ista Dispatch 2023
Direct from ISTA, hear how knee specialists are using weight bearing CT in daily practice…
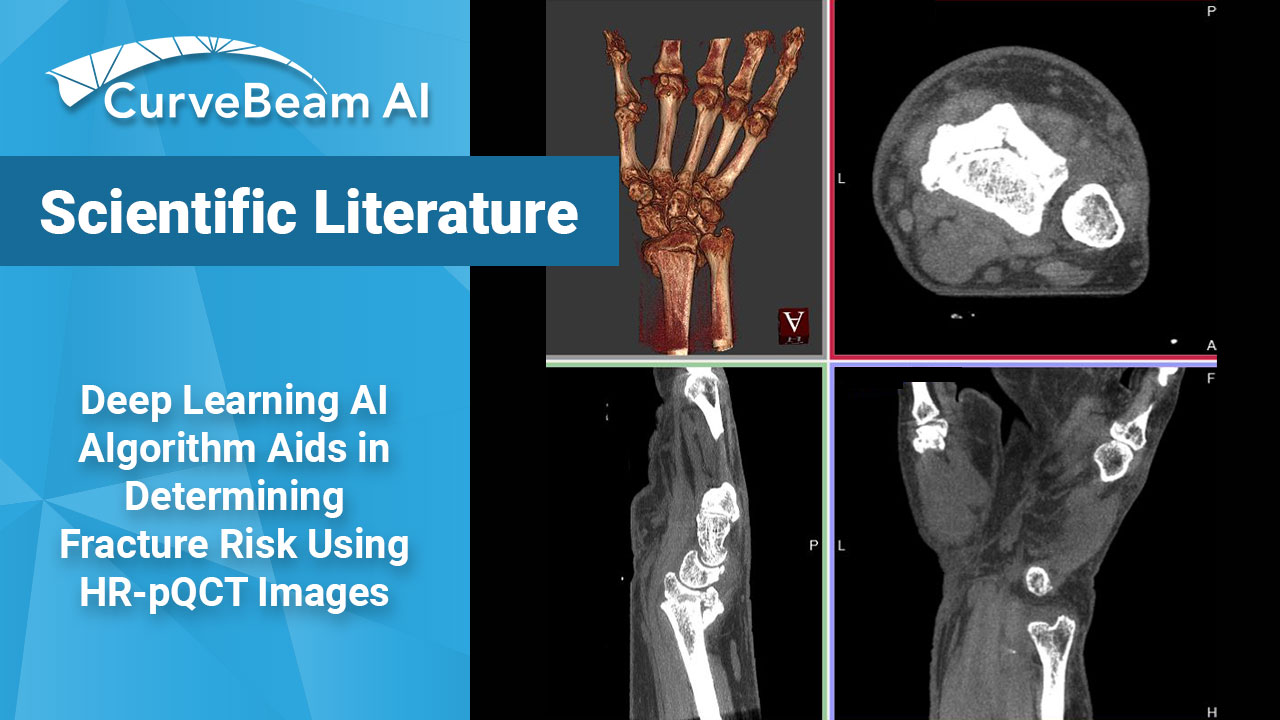
Deep Learning AI (DLAI) Algorithm Aids in Determining Fracture Risk Using High Resolution Peripheral Quantitative CT (HR-pQCT) Images
A recent publication in the online journal eLife has described the world’s first application of…
Why is WBCT an essential modality in an MSK Imaging Suite?
The use of upright CT, also known as weight bearing CT (WBCT), is one of…
WBCT Indications Series: PCFD
Progressive Collapsing Foot Deformity Progressive Collapsing Foot Deformity (PCFD), previously known as adult-acquired flatfoot deformity,…
WBCT Indications Series: Midfoot Arthritis
Midfoot Arthritis Midfoot arthritis occurs when there is loss of cartilage at the tarsometatarsal and/or…
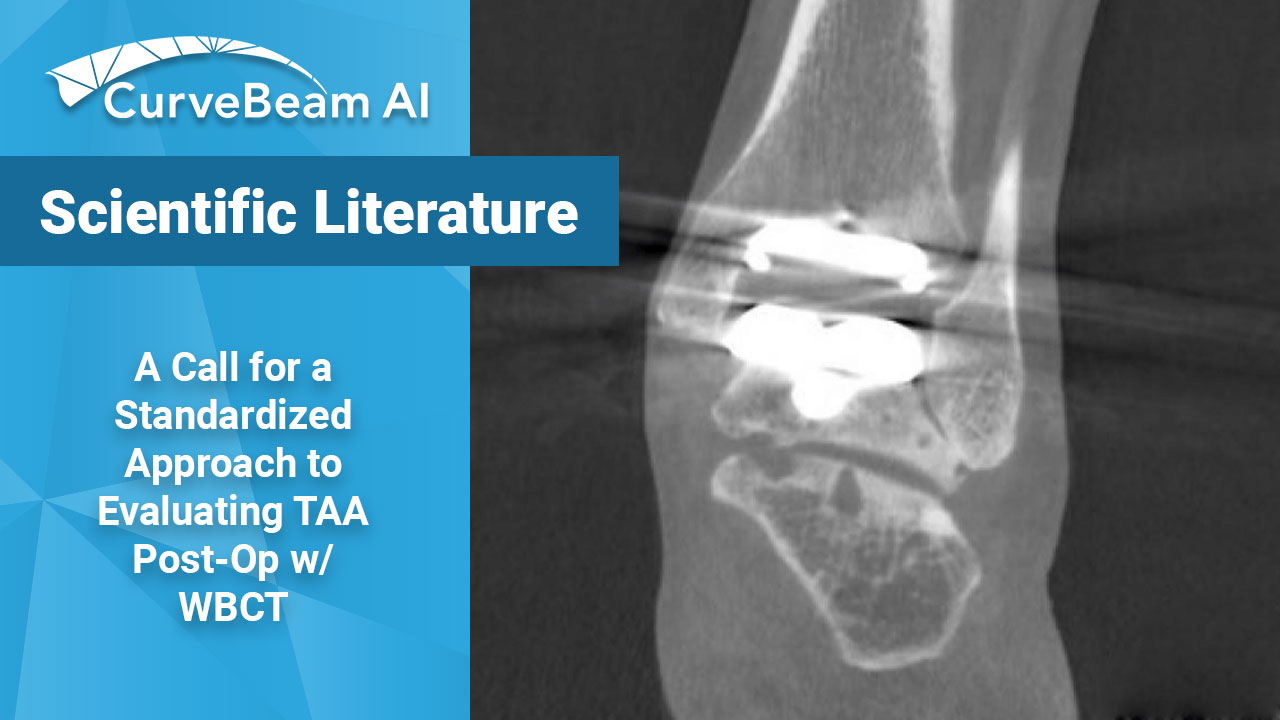
A Call for a Standardized Approach to Evaluating TAA Post-Op w/ WBCT
A paper published in Foot and Ankle International is calling for a weight bearing CT…
WBCT Indications Series: Lisfranc
Lisfranc Injuries A Lisfranc injury is characterized by an injury to the ligament that connects…
WBCT Indication Series: End Stage Ankle Arthritis
End Stage Ankle Arthritis End-stage ankle arthritis occurs when there is loss of cartilage at…
WBCT Indications Series: Syndesmosis
Syndesmosis A syndesmotic ankle sprain, also known as high ankle sprain, is an injury to…
WBCT Indication Series: Charcot Arthropathy
Charcot Arthropathy Charcot arthropathy is characterized by a destructive process that affects the bones, joints,…
Cone Beam CT: A Technical Explanation of White Paper Image Quality
Extremity cone beam CT (CBCT) systems are used as a point-of-care modality to improve workflow…
Introducing SkyRise
Introducing SkyRise, CurveBeam AI's fourth generation weight bearing CT platform for the orthopedic specialties. The…
CurveBeam AI Cast – How AI Imaging Technology is Revolutionizing Orthopedics
Imaging and diagnostics areimportant facets in healthcareand nearly every area of medicine relies on this…
CURVEBEAM AI CAST EPISODE PREVIEW: Stuti Singh discusses CurveBeam AI Autometrics
"It's easy for to me to look at a 3D model, but to then measure…
WBCT Indication Series: Hallux Valgus
Hallux Valgus Hallux valgus is a three-dimensional deformity of the first ray in which the…
CurveBeam AI Cast – Reimaging Patient-Specific Deformity Correction Planning
The foot and ankle are complex structures comprised of 26 small bones and 29 muscles…
CURVEBEAM AI CAST EPISODE PREVIEW: DR Perler discusses patient specific deformity correction
CurveBeam AI Cast guest host Dr. Lew Schon, MD, interviews Dr. Adam Perler, DPM, FACFAS,…
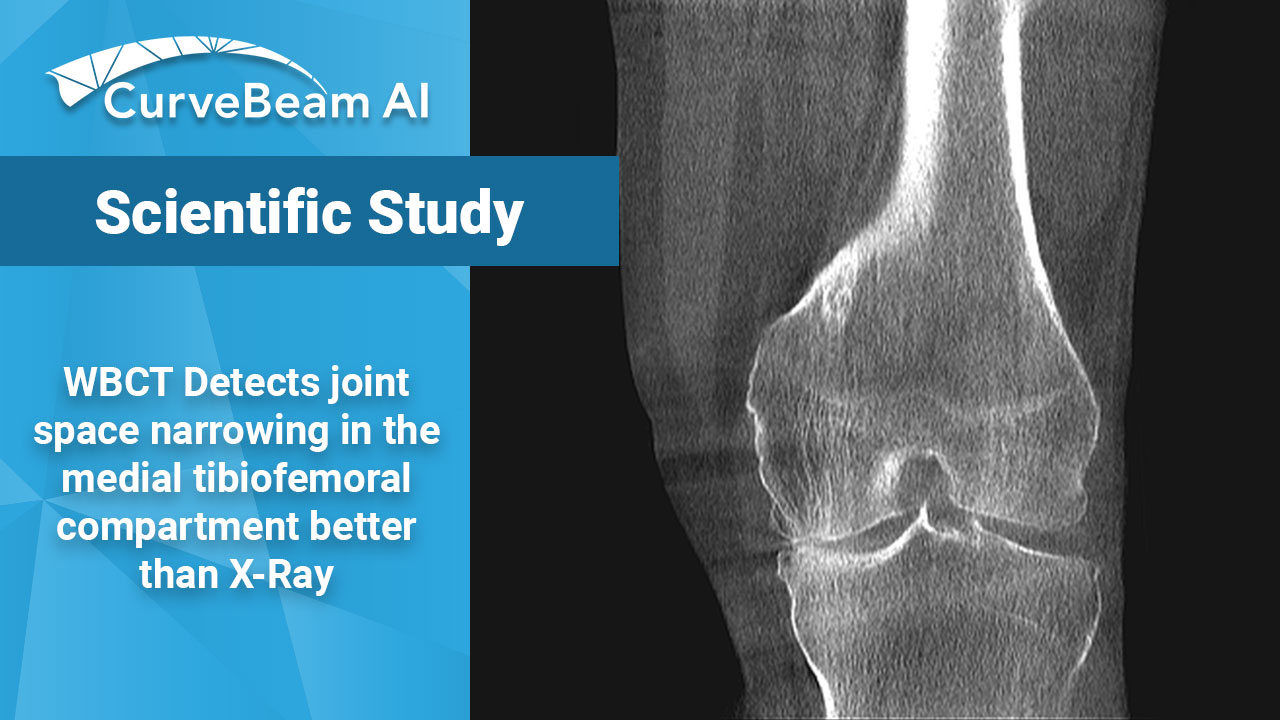
WBCT Better Detects Changes in Knee Joint Space Width than X-Ray in OA Patients
Key Points: Weight Bearing (WB) radiographs are limited by 1) their 2D nature and 2)…
CurveBeam AI Cast – The Origin Story: Dr. Lew Schon Interviews Dr. Cesar de Cesar Netto about his Journey in WBCT Research
Dr. Lew Schon, MD, makes his debut as guest host on CurveBeam AI Cast in…
CurveBeam AI Cast Episode Preview: Dr Netto shares research project
The first CurveBeam AI Cast episode guest hosted by Dr. Lew Schon.MD, drops on curvebeamai.com…
Dr. Ortiz Shares Insights on WBCT Imaging for Research Initiatives
Dr. Cristian Ortiz, MD, a foot and ankle orthopedic surgeon at Clinica Universidad de los…
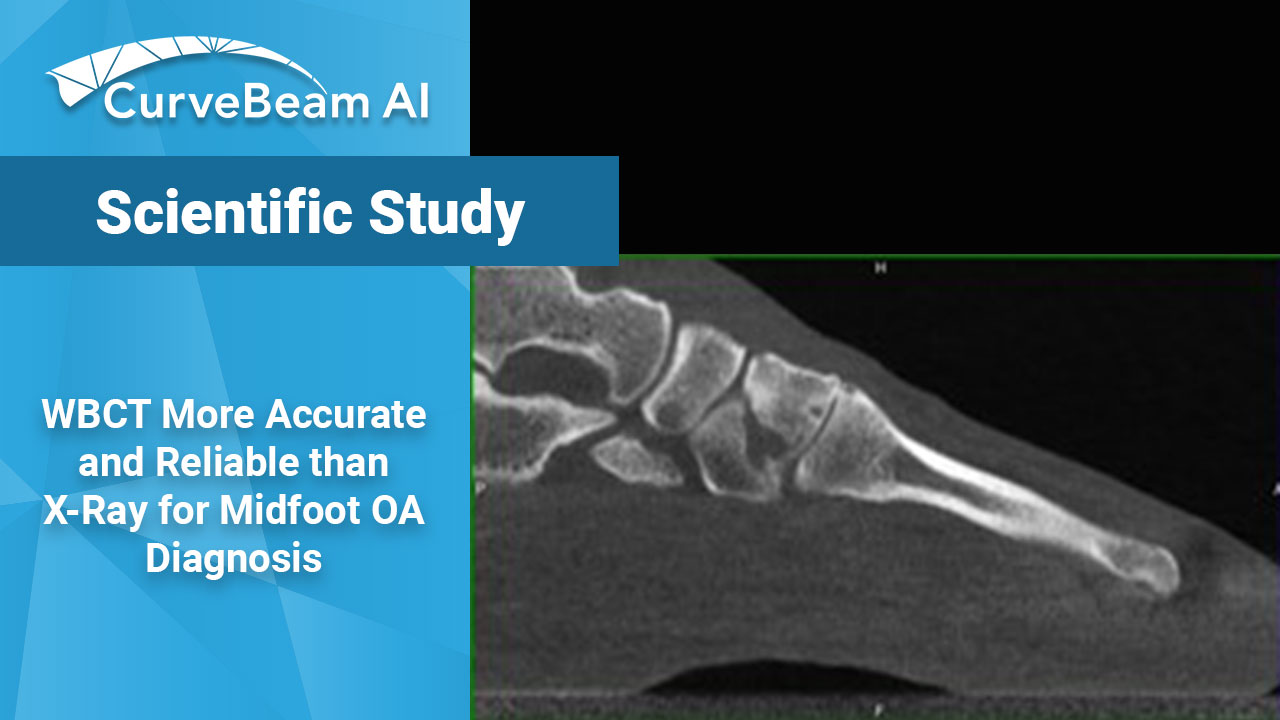
Comparative Assessment of Midfoot Osteoarthritis Diagnostic Sensitivity Using Weight Bearing Computed Tomography vs Weight Bearing Plain Radiography
Key Points: When evaluating midfoot arthritis osteoarthritis (OA), Weight Bearing X-Ray shows many false negatives…
Researchers Discuss Clinical Relevance of Knee OA Clinical Trials Using WBCT
Knee osteoarthritis (OA) clinical trials results show that weight bearing CT (WBCT) imaging may offer…
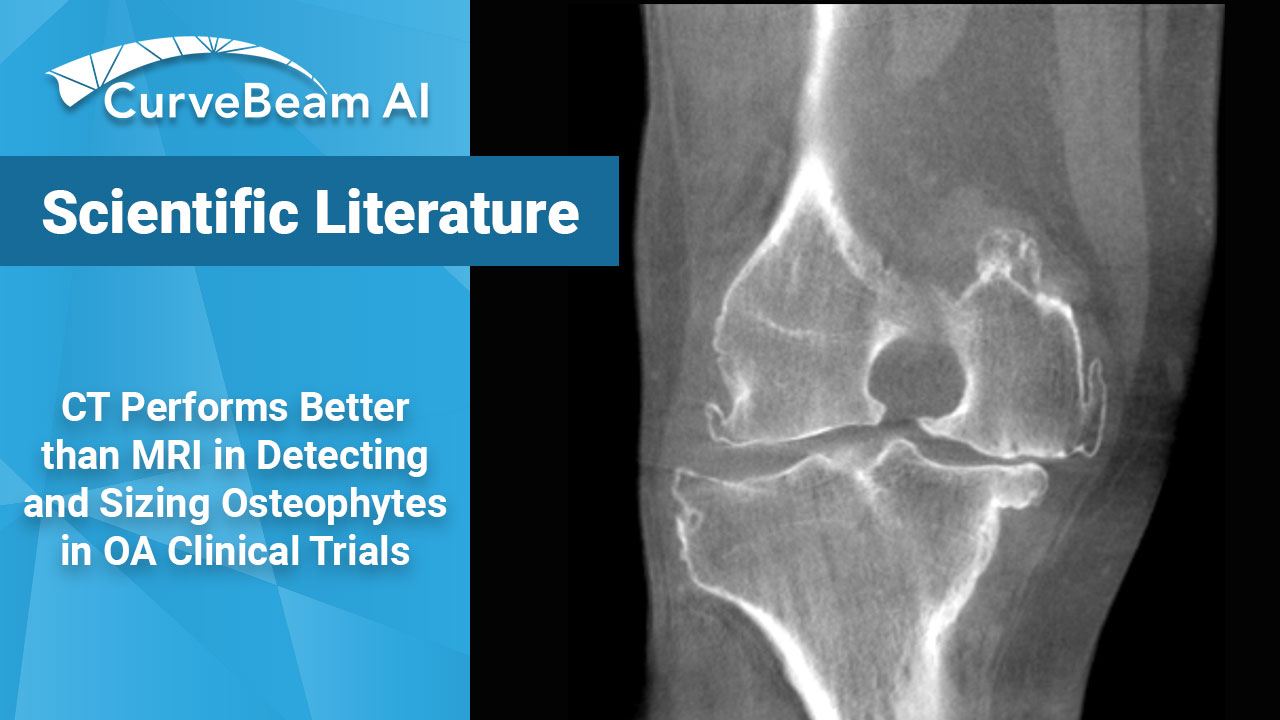
CT Outperforms MRI in Osteophyte Detection in Knees
Key Points: While magnetic resonance imaging (MRI) adequately detects the size and presence of osteophytes…
Dr. Cesar de Cesar Netto, MD, PhD, Awarded Top Paper in Foot and Ankle Classification
Dr. Cesar de Cesar Netto, MD, PhD, received the award for Top Paper in Foot…
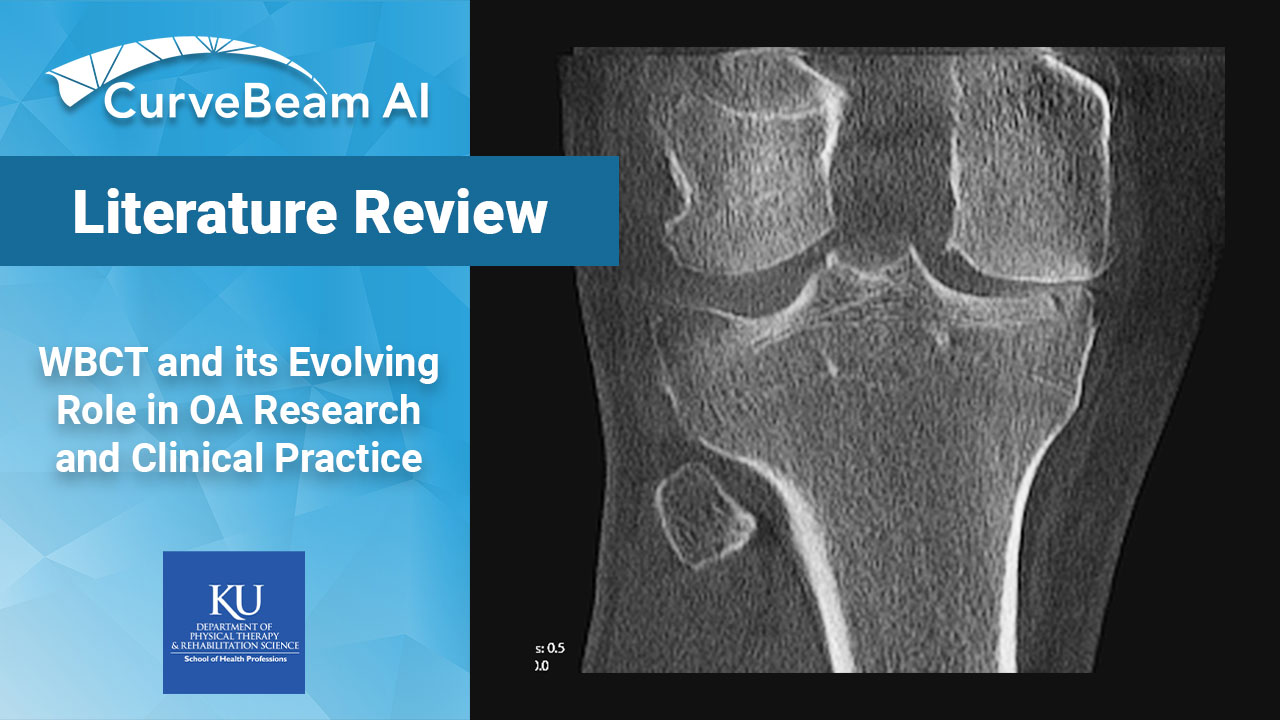
WBCT and its Evolving Role in OA Research and Clinical Practice
Key Points: Weight bearing CT (WBCT) can detect signs of osteoarthritis (OA), such as osteophytes,…
CURVEBEAM AI CLOSES A$25M PRE-IPO PRIVATE PLACEMENT SUPPORTED BY PROMINENT INVESTORS
The Private Placement was oversubscribed and increased from an initial target of A$15m Investors in…
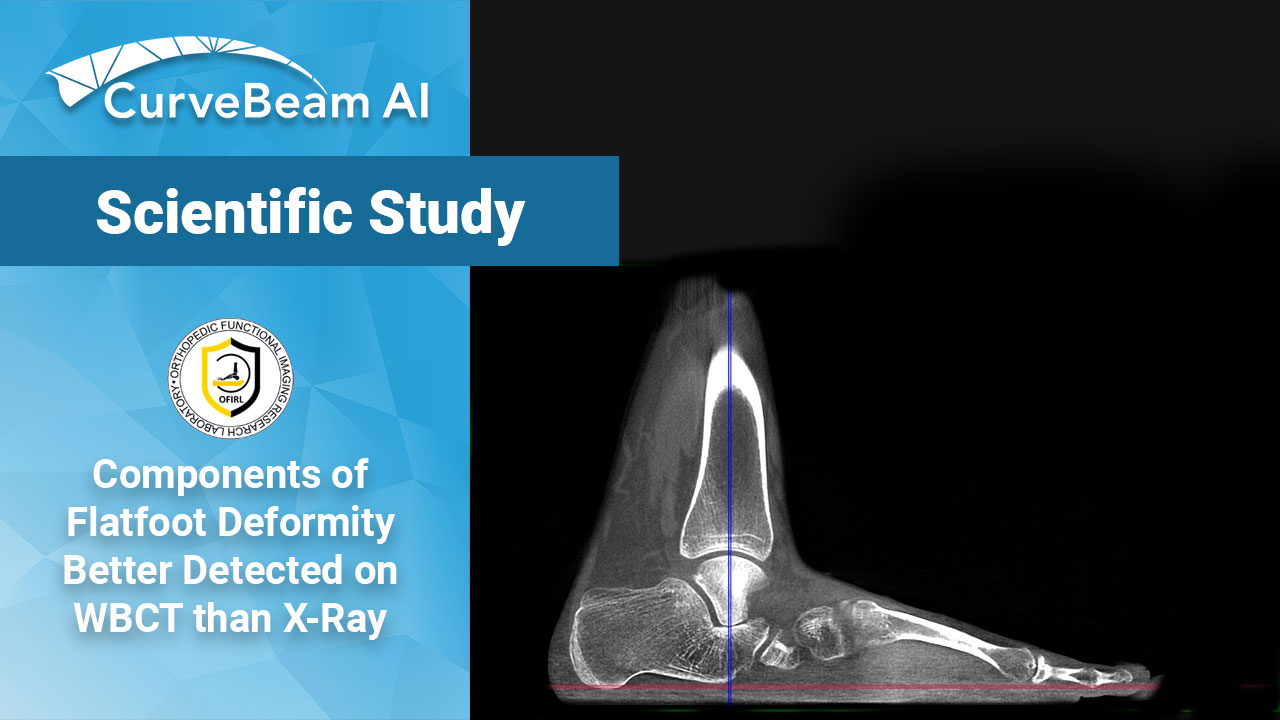
WBCT Better Detects Complexities of Flatfoot
Key Points: Weight Bearing CT (WBCT) imaging has facilitated the introduction of a new classification…
RedPoint Medical 3D Announces Weight Bearing CT, When Available, is Preferred Input for Its Surgical Planning Solution
<Hatfield, Penn – January, 31, 2023> RedPoint Medical 3D (RPM-3D) announced that, when available, weight…
Happy Holidays from CurveBeam AI
As the year comes to a close we want to wish all of our team,…
WBCT + Coverage Mapping Finds Significant Subluxation of the TTJ in PCFD Patients
Key Points Weight bearing CT (WBCT) combined with coverage mapping (CM) and 3D distance mapping…
Made in America: Countdown to the CurveBeam AI Premiere
MarketScale's Made in America series explores industries that are thriving in the USA. In it's…
CurveBeam AI Cast: Orthopedic CT Imaging Accreditation in a Changing MedTech Landscape
The medtech landscape is constantly evolving. Keeping up with those changes to maintain accreditation standards…
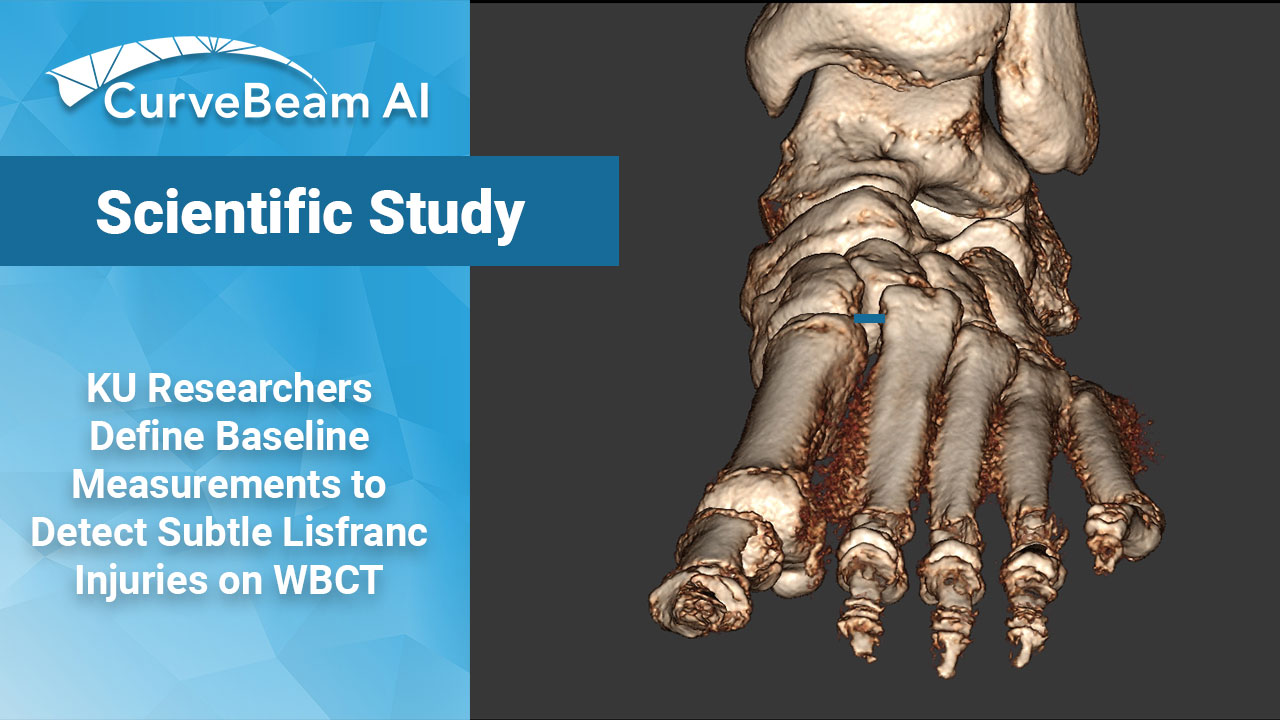
KU Researchers Define Baseline Measurements to Detect Subtle Lisfranc Injuries on WBCT
Key Points: Up to 20% of subtle Lisfranc injuries are missed on initial plain films…
CurveBeam and StraxCorp Close Merger
<Hatfield, PA – October 20, 2022> Medical imaging companies CurveBeam, LLC and StraxCorp, Ltd., announced today…
CurveBeam AI Enters Strategic Partnership with Healthy Bones Australia
<Australia - October 13, 2022> To mark its 21st anniversary of pioneering bone health in…
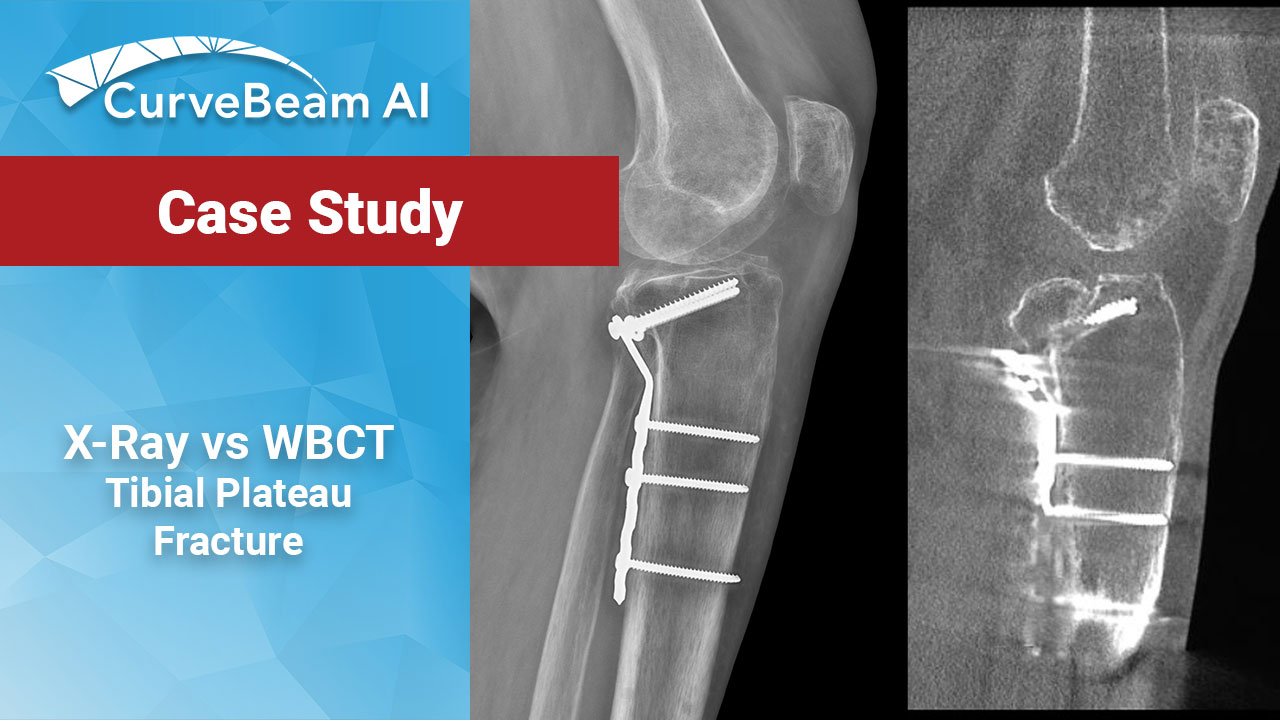
Tibial Plateau Fracture Case Study
The benefits of using weight bearing CT (WBCT) imaging for pre-operative planning have been explored…
Researchers Propose New OA Classification Method Using WBCT Imaging
Key Points: The Kellgren Lawrence (KL) grade is the most common system used to classify…
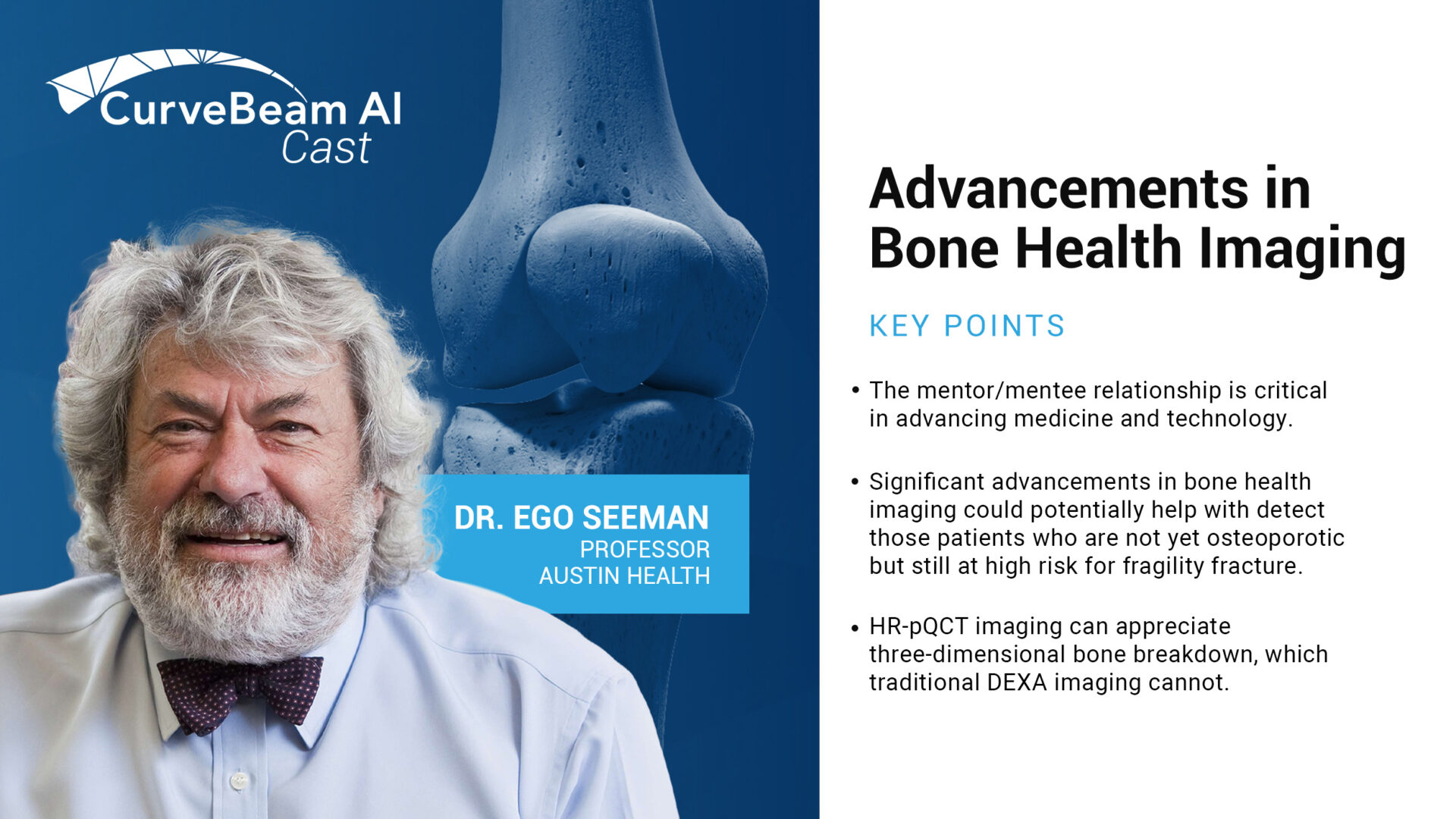
CurveBeam AI Connect: Advancements in Bone Health Imaging
CurveBeam AI Medical Director - Endocrinology Prof. Ego Seeman recently sat down with Vinti Singh,…
CurveBeam AI Medical Director– Endocrinology Receives Prestigious Award at ASBMR 2022 Annual Meeting
CurveBeam AI Medical Director – Endocrinology Ego Seeman was awarded the Gideon A. Rodan Excellence…
FDA Grants Breakthrough Device Designation for CurveBeam AI’s OssView™ Bone Fragility Software
<HATFIELD, Penn. - September 12, 2022> CurveBeam AI, Ltd announced its medical diagnostic software, OssView™,…
CurveBeam and StraxCorp Announce Definitive Merger Agreement to Become CurveBeam AI
<Hatfield, Penn. – September 6, 2022> – CurveBeam, LLC of Hatfield PA, USA and StraxCorp…
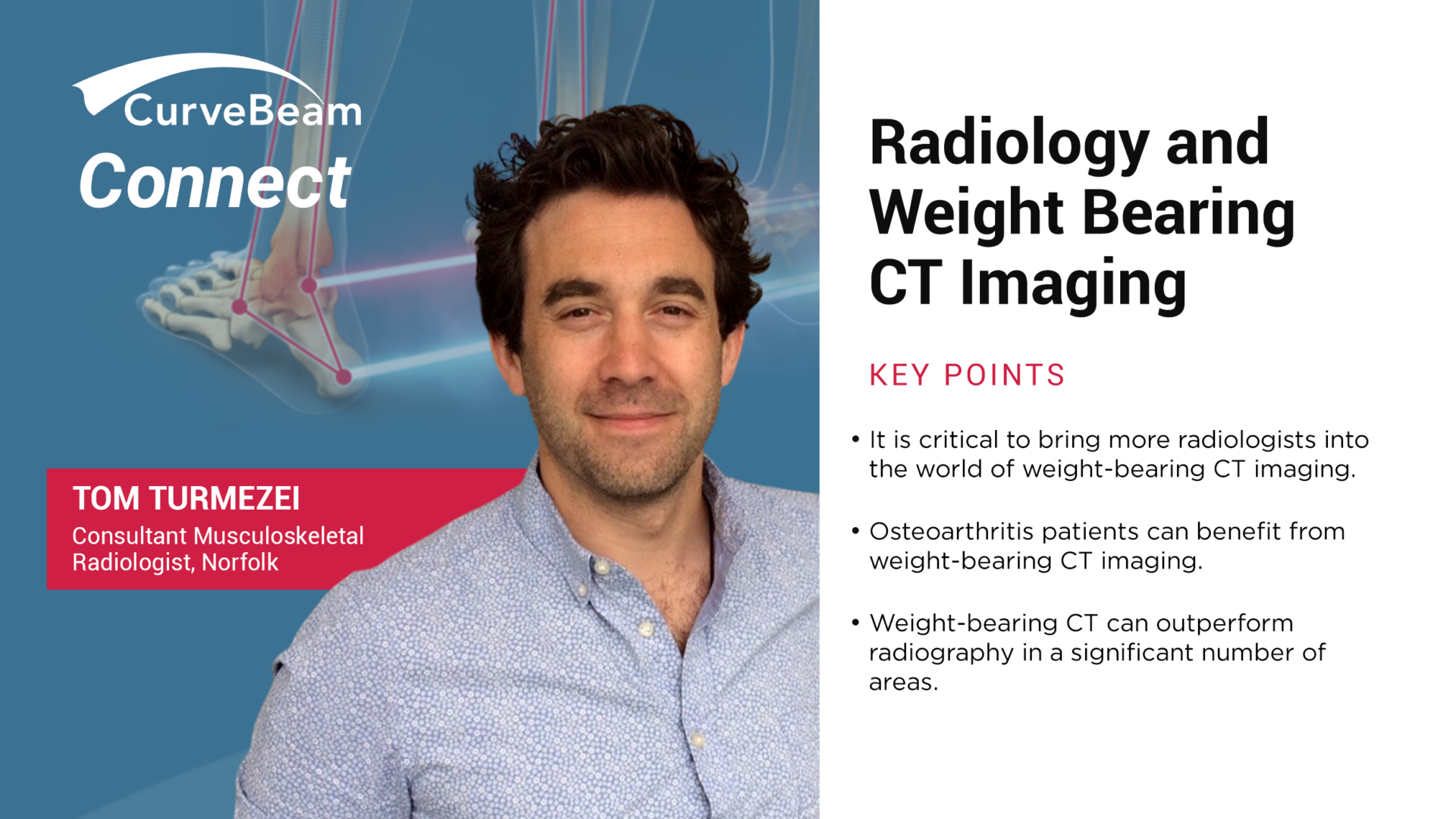
CurveBeam Connect: Radiology and Weight Bearing CT Imaging
In the lead-up to CurveBeam’s Annual User Meeting, initially planned for mid-October, 2022, CurveBeam Connect…
CurveBeam User Meeting Speaker Series: Meet Dr. Sudheer Reddy
Fall is coming, and so is CurveBeam’s Annual User Meeting, held this year from Oct…
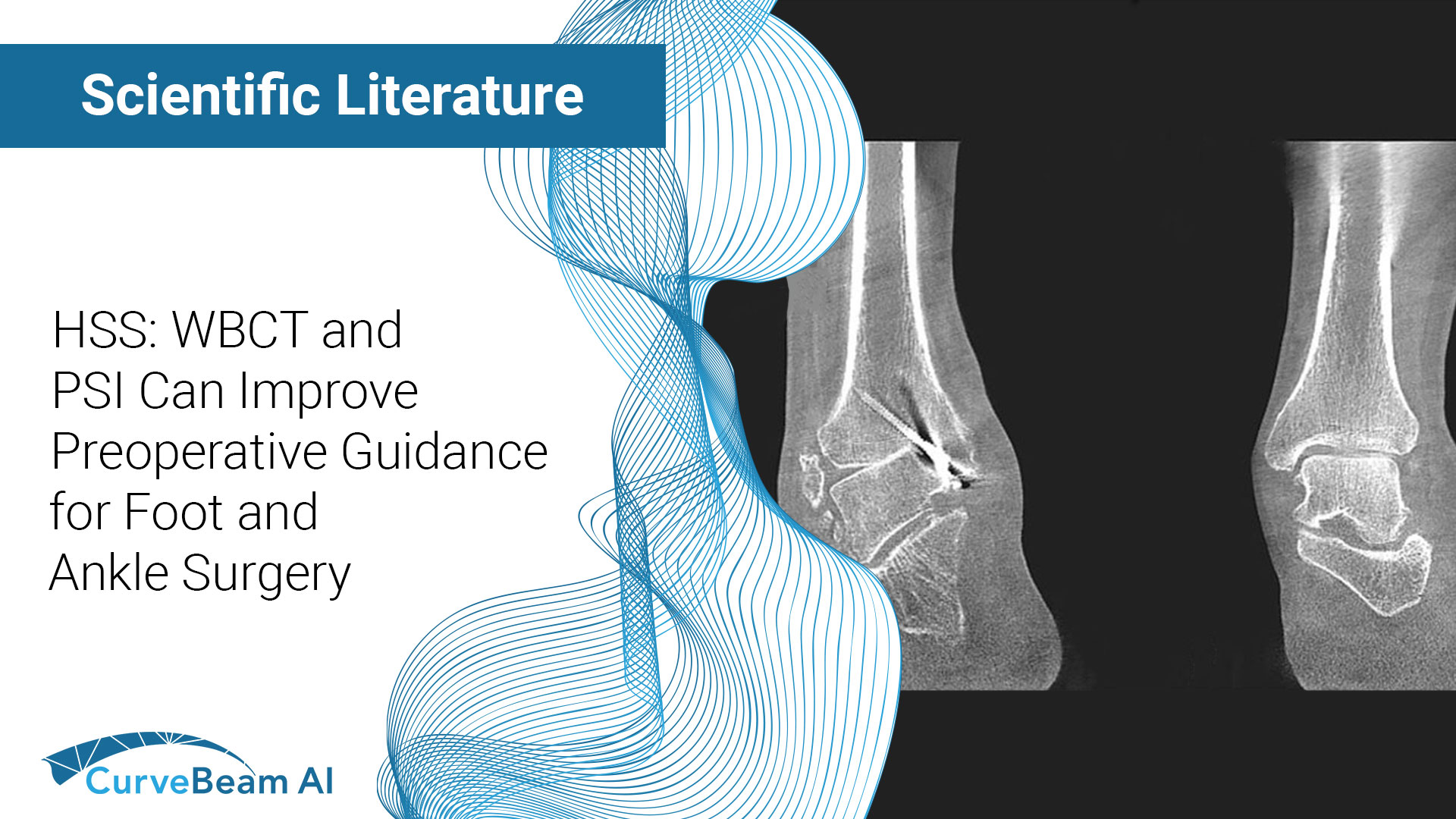
HSS: WBCT and PSI Can Improve Preoperative Guidance for Foot and Ankle Surgery
Key Points: Weight Bearing CT (WBCT) has helped surgeons better understand the orientation and specifications…
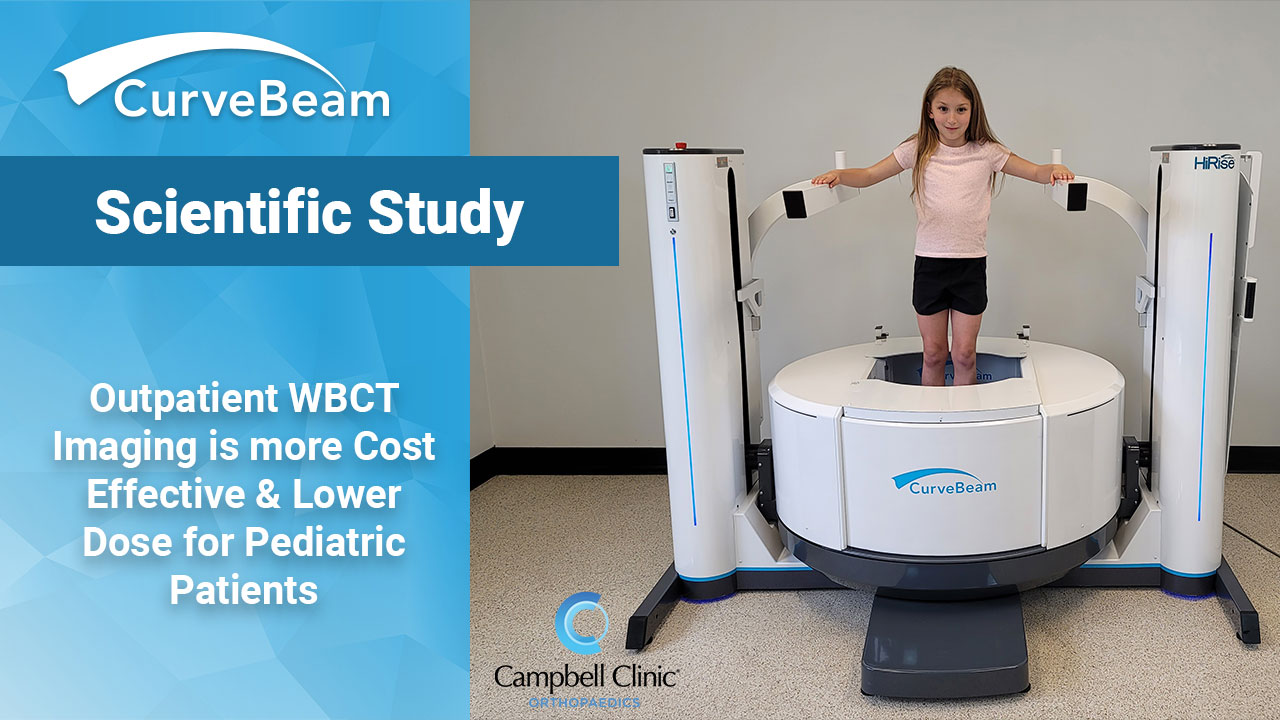
Campbell Clinic Analyzes Pediatric WBCT Scans
Key Points: This study compared clinical indications, demographic data, radiation dose and cost of weight…
Sana Hospital Rummelsberg Adds HiRise WBCT System
Sana Hospital Rummelsberg in Bavaria, Germany initially made headlines in 2013, when it became one…
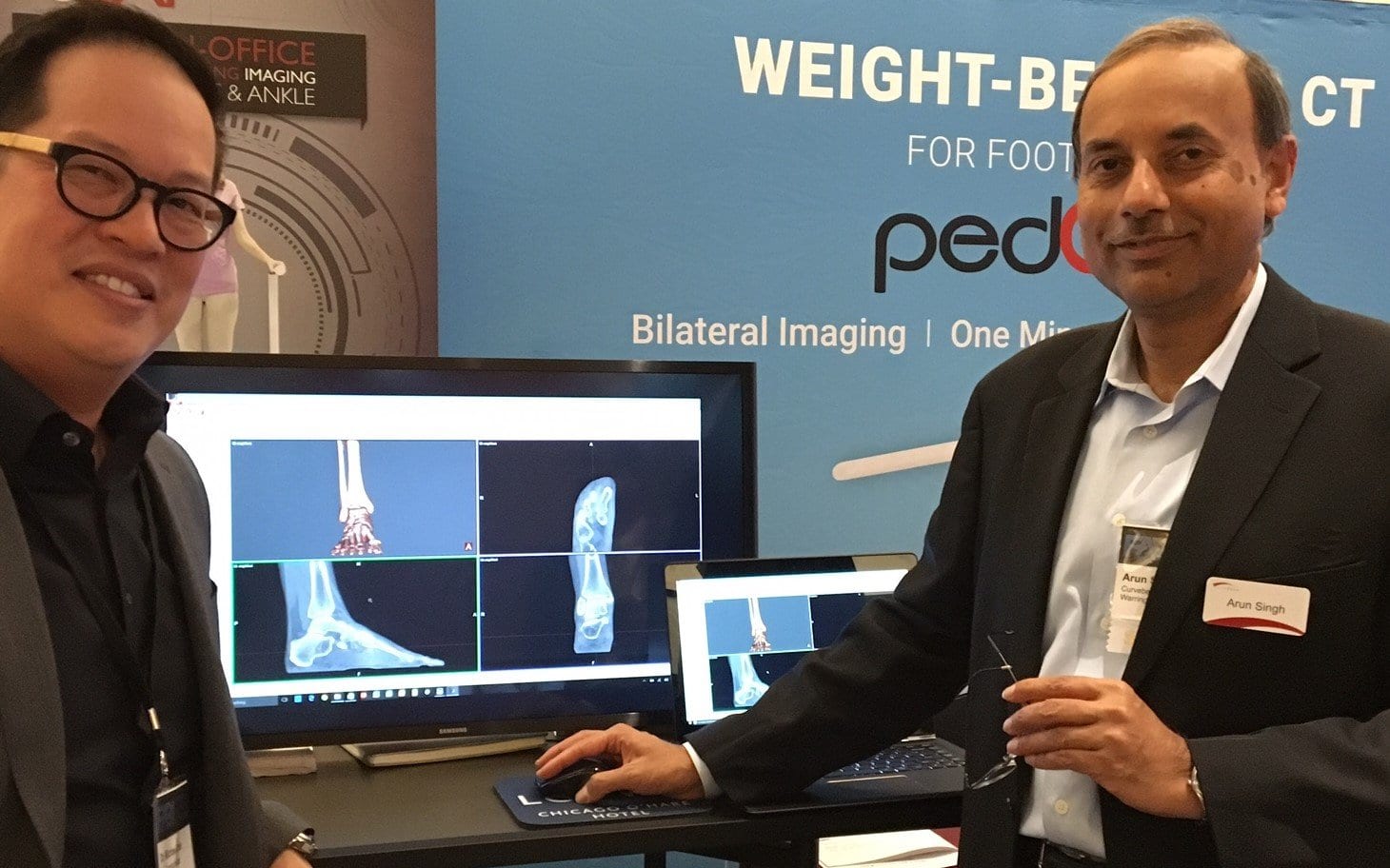
Meet CurveBeam President & CEO Arun Singh
CurveBeam President & CEO Arun Singh founded CurveBeam in 2009, two years after his first…
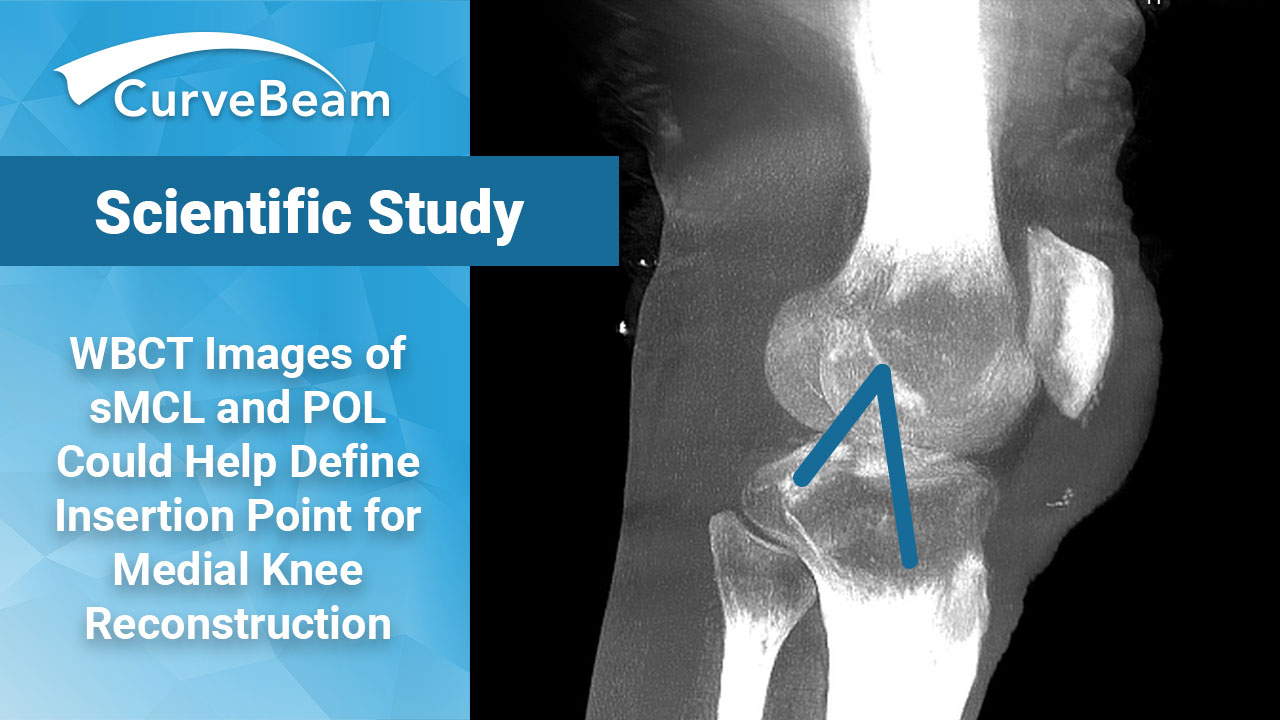
WBCT Images of Knee Ligaments Provide Insights Into Medial Knee Reconstruction Techniques
Key Points: Weight Bearing CT imaging was used to observe elongation patterns of the superficial…
Tennis Player Rafael Nadal Diagnosed with Müller Weiss Syndrome
Sports fans and sports medicine specialists alike are all watching sports star Rafael Nadal as…
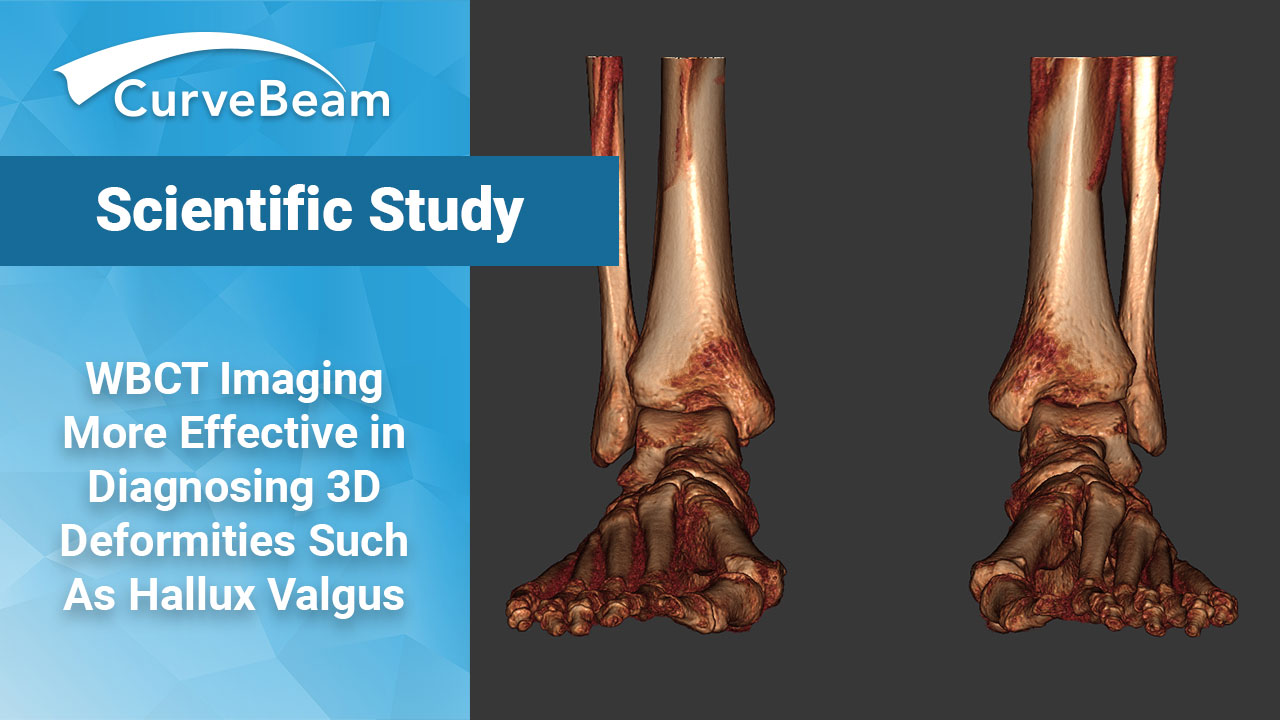
Study Finds 3D WBCT Measurements are Reliable for Hallux Valgus Planning
Key Points 2D imaging has limitations for pathology such as Hallux Valgus (HV) as pronation…
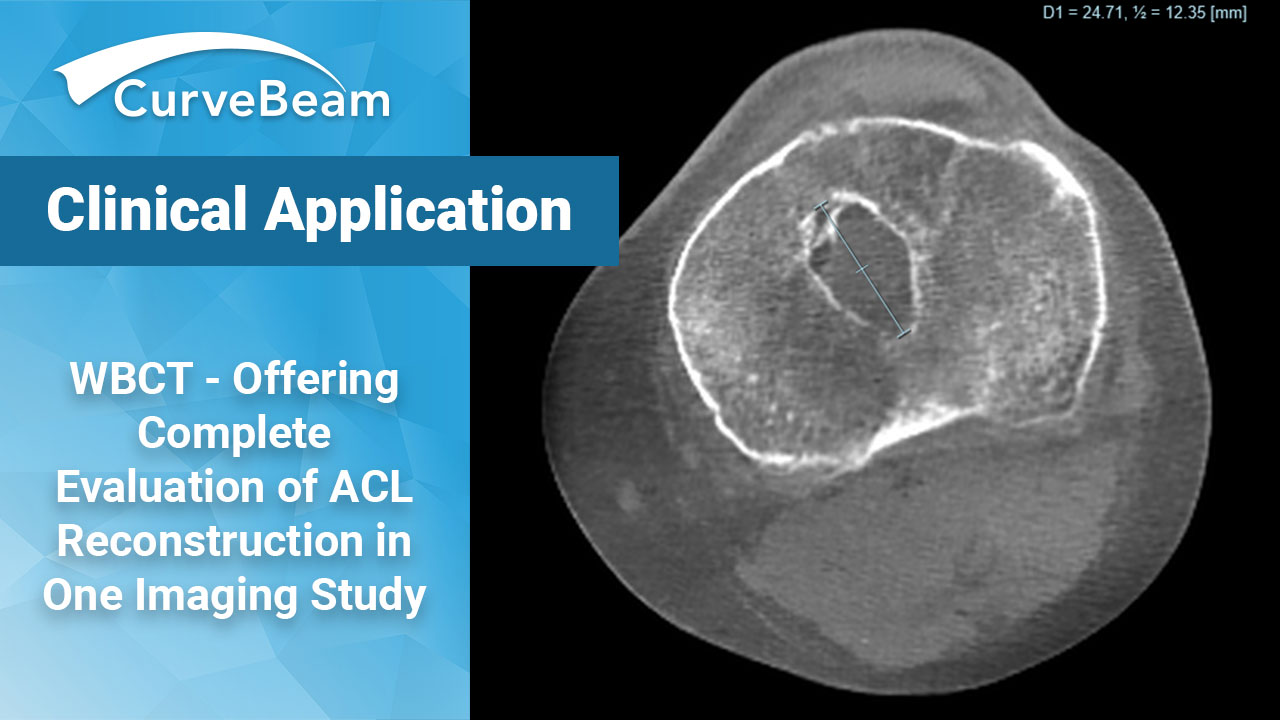
How To Get a Complete Evaluation of ACL Reconstruction in One Imaging Study
With approximately 100,000 – 175,000 Anterior Cruciate Ligament (ACL) reconstructions taking place annually in the…
A Novel Anteroposterior Axis of the Tibia for TKA: A WBCT Analysis
Key Points: On weight bearing CT (WBCT) scans of healthy subjects, the AP axis was…
Effective Dose of HiRise Hip Scan “Substantially Lower” than MDCT
Weight Bearing CT (WBCT) imaging of the hip and extremities permits diagnosis and surgical management…
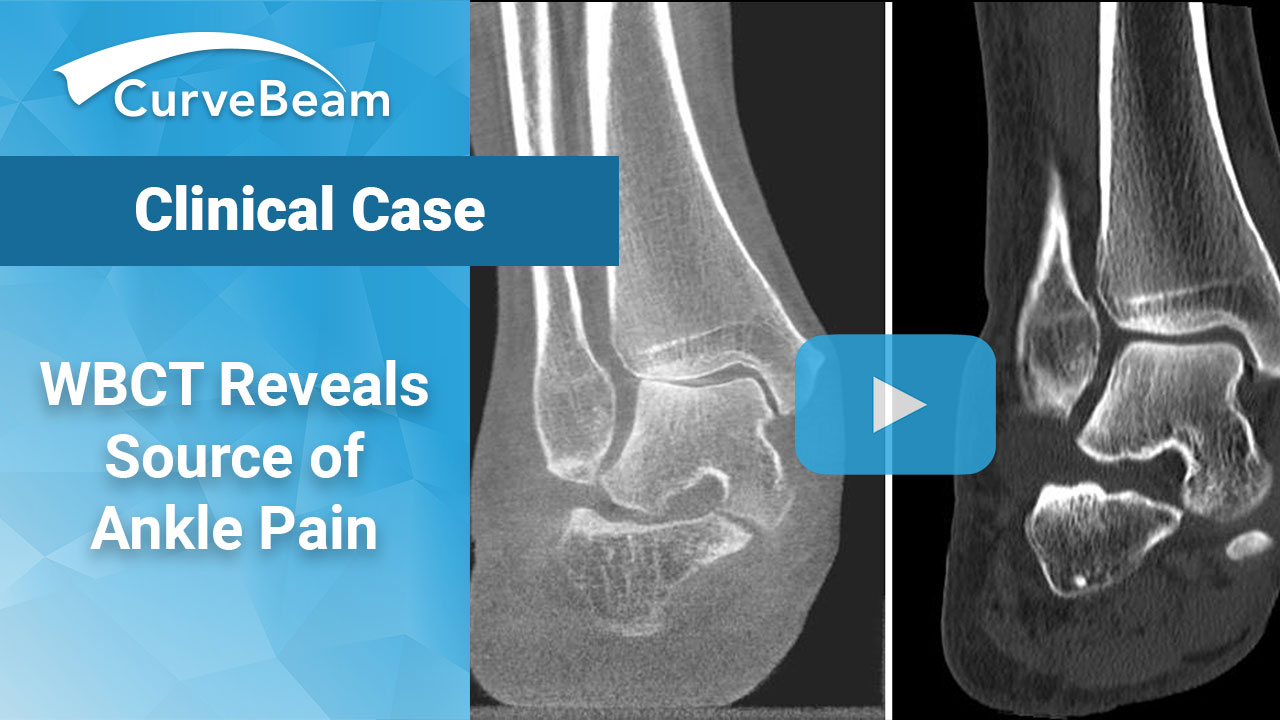
Case: WBCT Helps Diagnose Source of Ankle Pain
Prof. Dr. Markus Walther is the Director of Foot and Ankle Department of Schön Klinik…
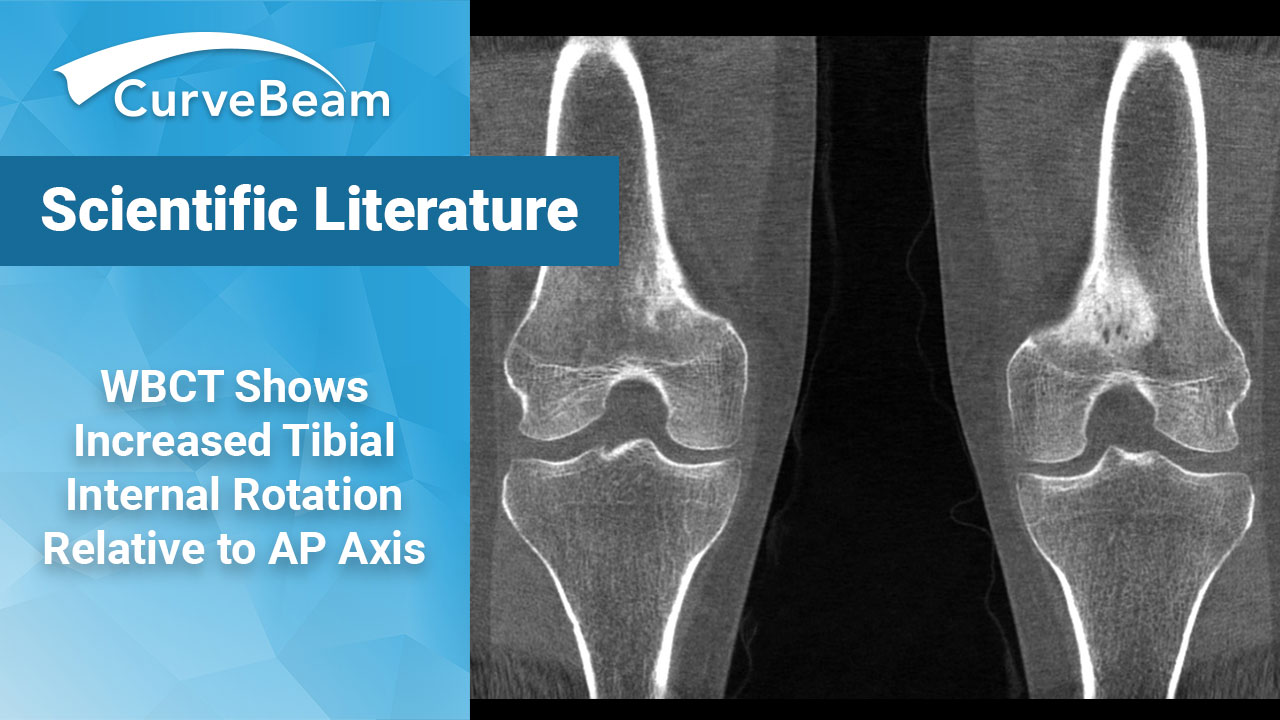
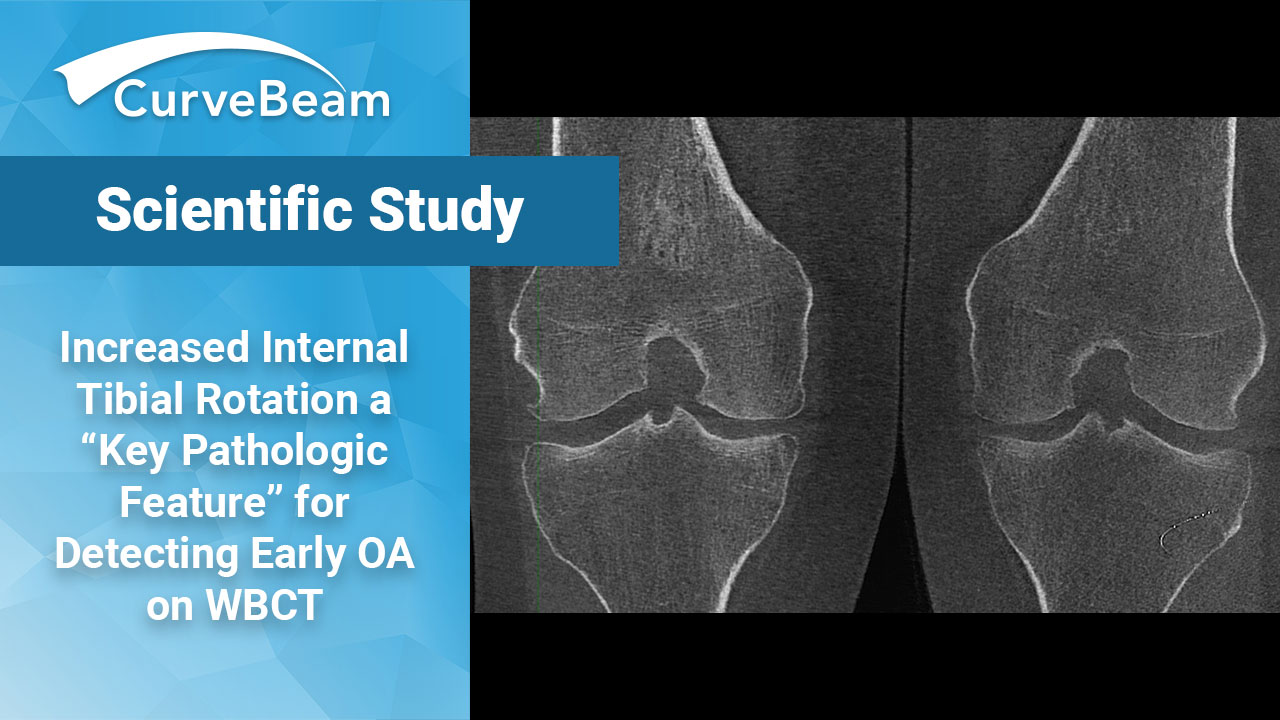
An Increase in Tibial Internal Rotation Due to Weight Bearing is a Key Feature to Diagnose Early-Stage Knee Osteoarthritis
Key Points: Greater flexion, adduction, and tibial internal rotation can be observed in the standing…
Hospices Civils de Lyon Acquires First HiRise in France
The CurveBeam HiRise was recently featured on the French media channel BFM Lyon in a…
WBCT Helps Define Safe Zone of FAO in Hindfoot Alignment Assessment
In 2019, an award-winning study theorized the development of periprosthetic cysts post a total ankle…
Orthopaedic Institute for Children Acquires HiRise for Pediatric Imaging
Pictured above: Luis Carrasco, Jr., 18, was the first patient to be scanned in Orthopaedic…
Dr. Walther: WBCT Imaging Has Improved Pediatric Imaging
Weight bearing CT (WBCT) imaging holds tremendous promise for improving pediatric diagnostics. The CurveBeam HiRise…
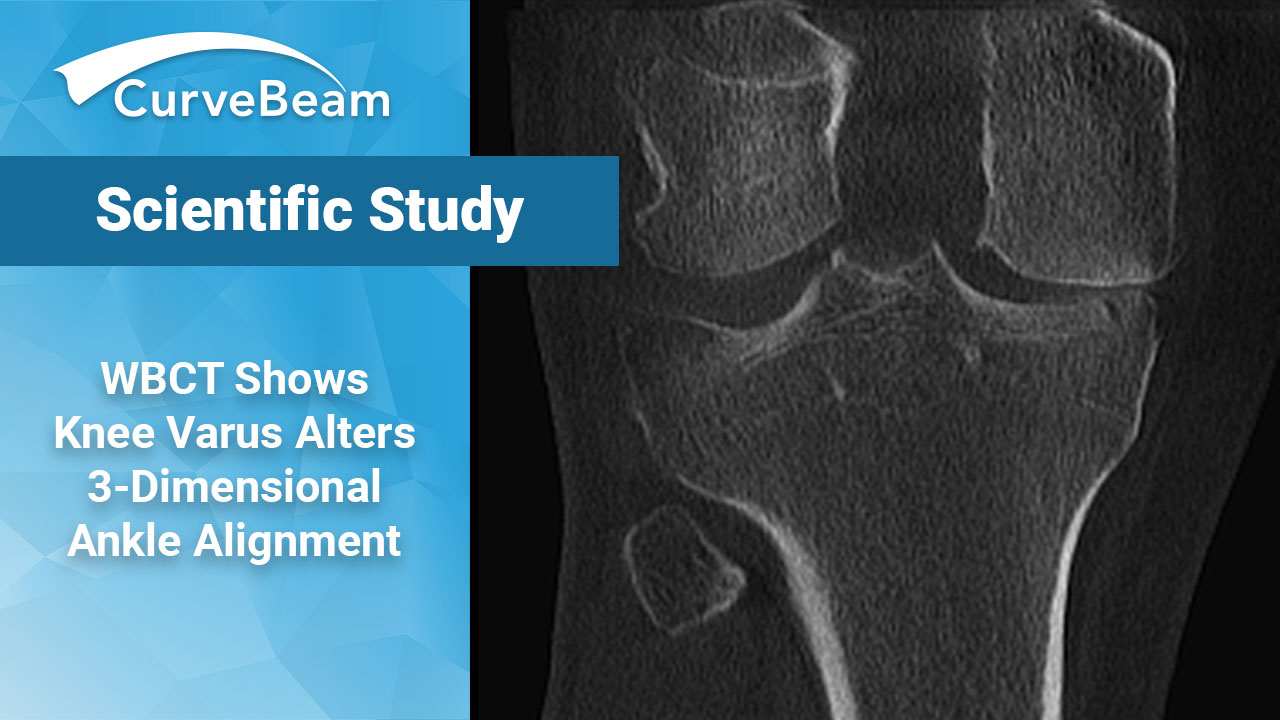
WBCT Study Shows Knee Varus Alters Three-Dimensional Ankle Alignment in Standing
Key Points: WBCT studies showed each Kellgren-Lawrence grade of knee osteoarthritis has its own characteristic…
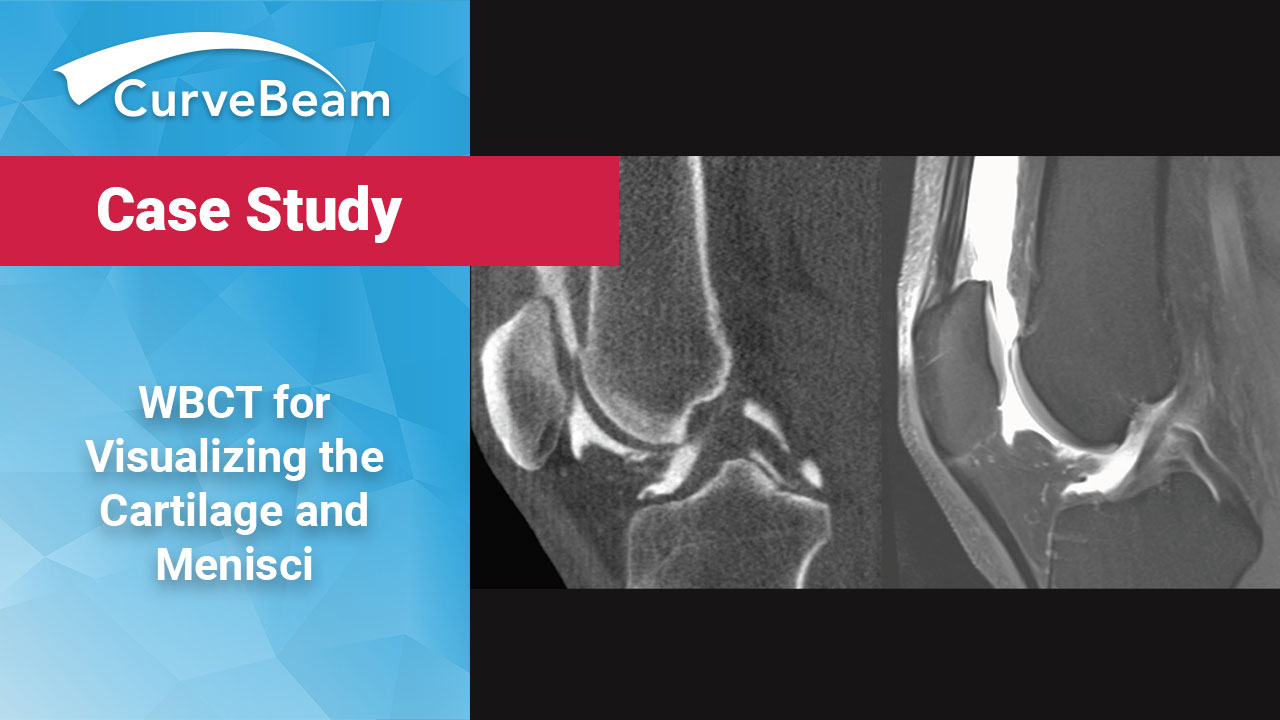
Case Study: WBCT for Visualizing the Cartilage and Menisci
Current Standard of Care MRI is the current standard of care for non-invasive visualization of…
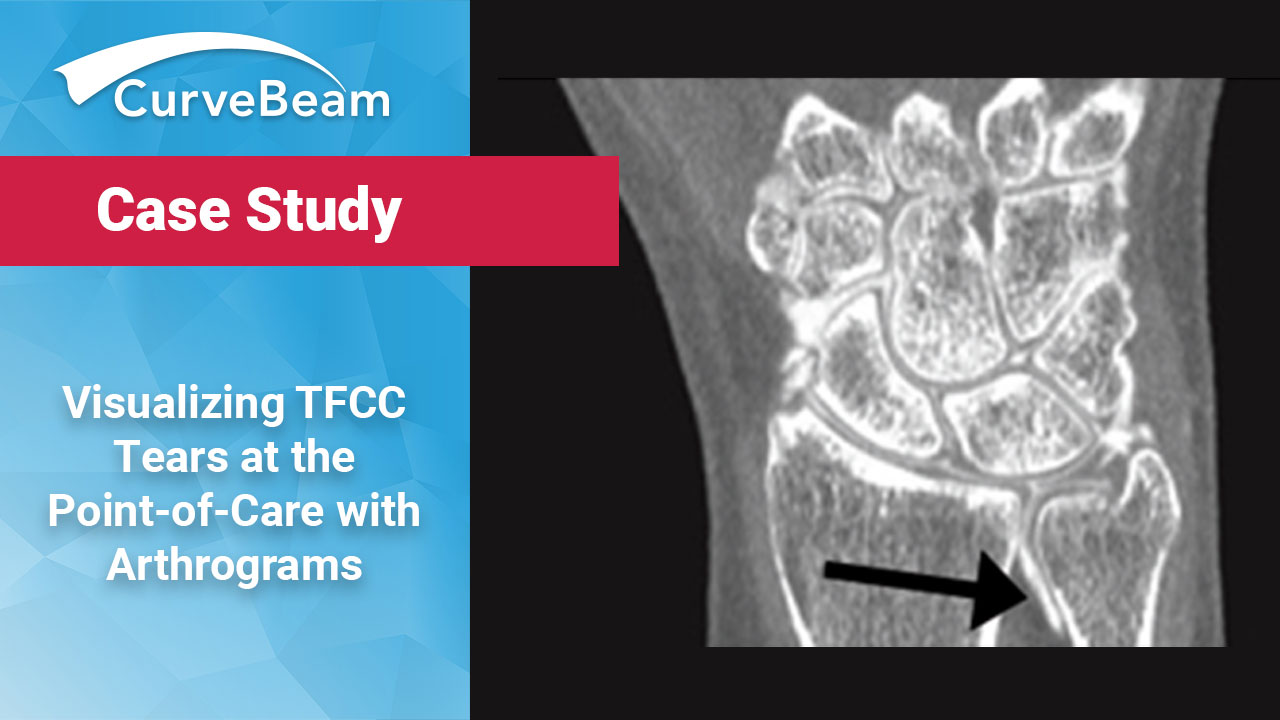
Visualizing TFCC Tears at the Point-of-Care
Ritesh Durgapersad, RT(R) ARMIT The triangular fibrocartilage complex (TFCC) is a major stabilizer of the…
Researchers Measure TSA on WBCT to Inform Syndesmosis Reduction
Weight Bearing CT (WBCT) has been shown to be a reliable method to diagnose syndesmotic…
CurveBeam Connect: Innovative Technologies are Changing the Economic Landscape of the Healthcare System – Part 3 of 3
KEY POINTS: Layers of security are built into Efferent Health’s technology solutions to protect critical…
CurveBeam Connect: Innovative Technologies are Changing the Economic Landscape of the Healthcare System – Part 2 of 3
KEY POINTS: Current healthcare processes are cumbersome and costly. Common healthcare IT applications automate processes…
CurveBeam Connect: Innovative Technologies are Changing the Economic Landscape of the Healthcare System – Part 1 of 3
KEY POINTS: Understanding the economic landscape of healthcare in the United States. Cuellar completed a…
Live from ACFAS: The Future of Custom Insoles
CurveBeam, T-Soles, and PodoPrinter team up to bring you the future of custom insoles. Watch…
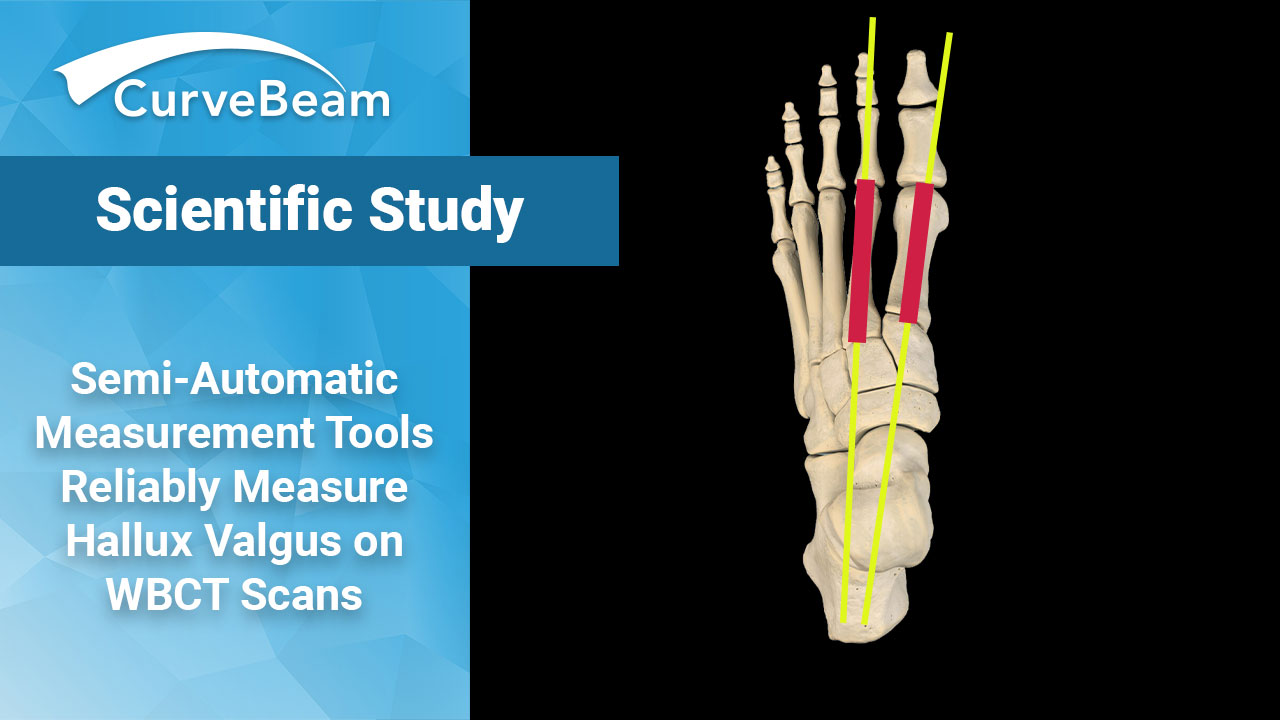
Comparison Between Weight Bearing CT Semi-Automatic and Manual Measurements in Hallux Valgus
Key Points Semi-automatic measurements made on WBCT scans are reproducible and comparable to measurements performed…
CurveBeam Connect: Preparing for CurveBeam’s Annual User Meeting
Those in the medical field who have heard of or used weight bearing computed…
Utility of WBCT to Diagnose Syndesmotic Instability in Patients with Weber B Lateral Malleolar Fractures
Key Points WBCT is able to discern a stable from an unstable syndesmosis. WB X-Rays…
Measuring Hip Instability in Dysplastic Patients with WBCT
Weight bearing CT (WBCT) may be able to visualize changes in measures of hip instability…
The Achille Medical Center offers WBCT with pedCAT Premium
The Achille Medical Center in Montpellier, France, in January, celebrated with an opening ceremony in…
Researchers Find Two WBCT Measurements that Diagnose PCFD Almost Perfectly
Key Points A study from Clinique de I’Union found that a combination of 2D and…
CurveBeam Connect: Harnessing Medical Imaging Data to Supercharge Orthopedics
The orthopedic specialty has progressed since the first robotic applications were first introduced 30…
Andy Goldberg: Hip Version May Correlate with Foot & Ankle Pathologies
Internal rotation of the hip could be the root cause of ankle pathology, and weight…
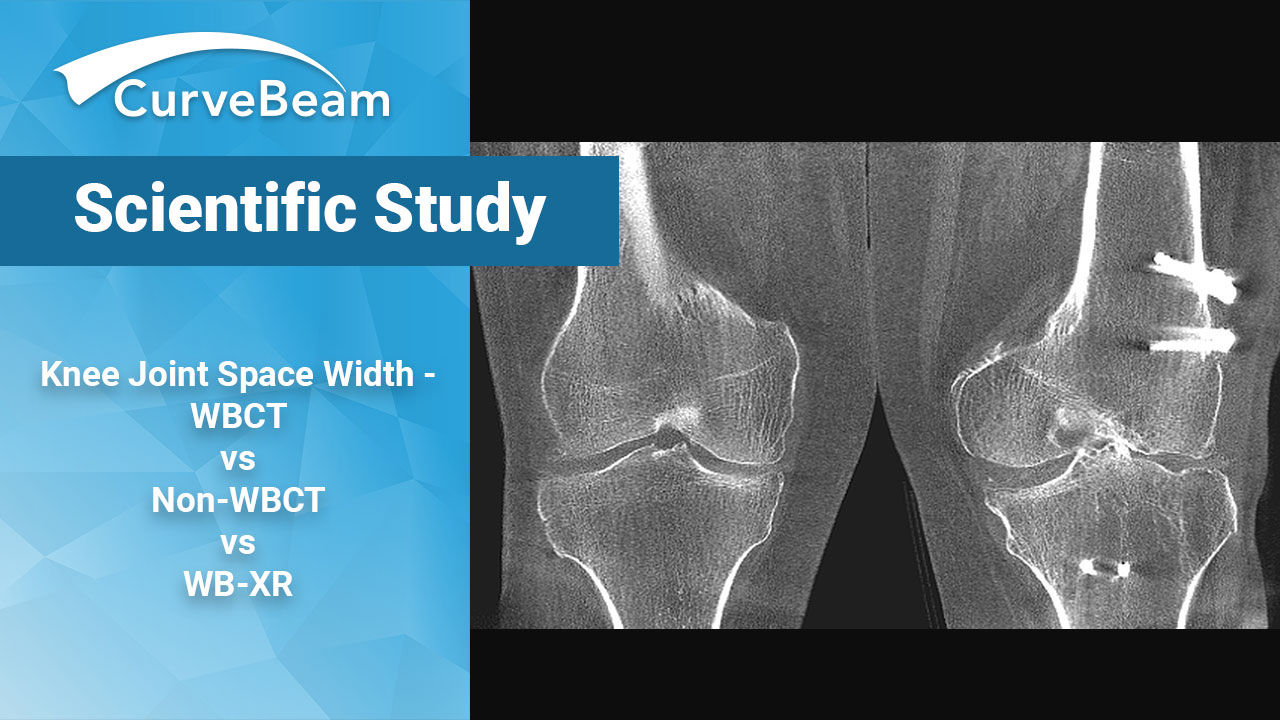
Knee Joint Space Width – WBCT vs Non-WBCT vs WB-XR
3D Quantification of Knee Joint Space Width with WBCT, Non-WBCT & WBXR: A Comparison Key…
CBCT Arthrogram Effectively Diagnoses Cartilage and OCD Lesions in Ankle
Cone Beam CT (CBCT) arthrogram can diagnose osteochondral (OCD) lesions in the ankle in high…
Testimonial: Dr. Hodges Davis, MD
Dr. Hodges Davis, MD said in the future, weight bearing CT will be the…
Testimonial: Dr. Lew Schon, MD
Dr. Lew Schon, MD, says the CurveBeam’s HiRise WBCT system provides an extremely detailed…
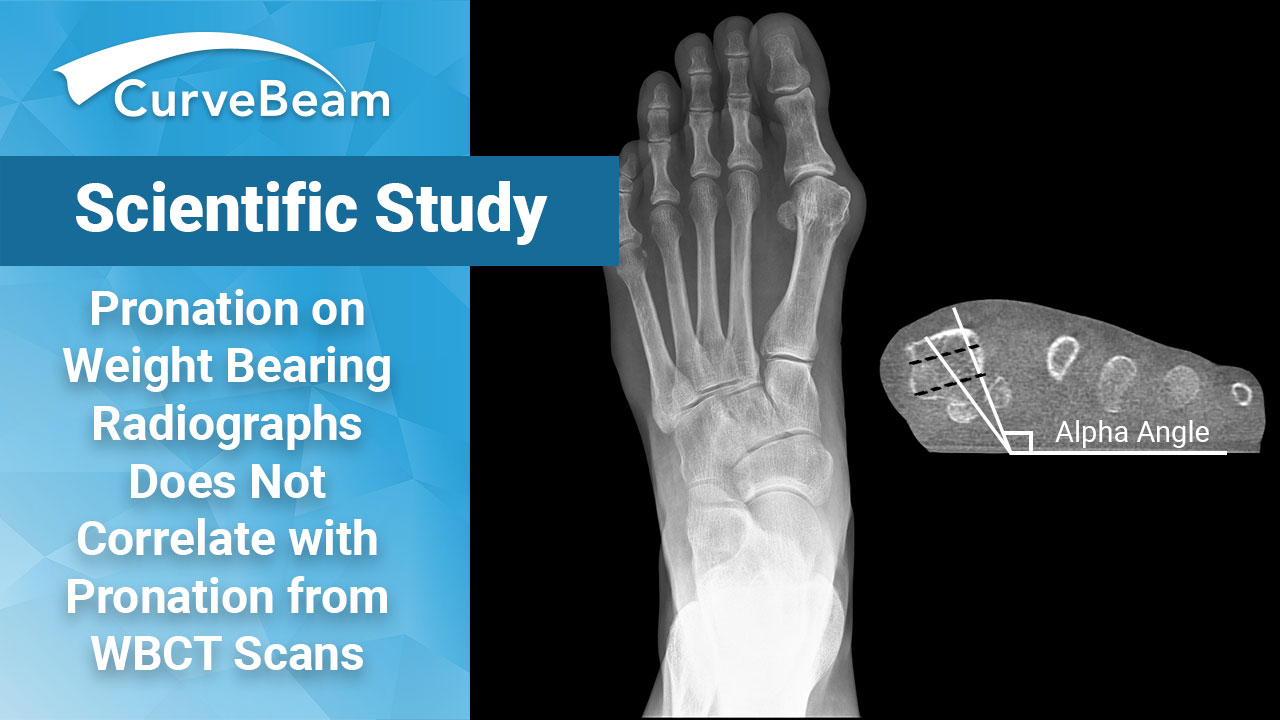
WBCT – “New Gold Standard” for HV Pronation Measurements
Key Points A study from Hospital for Special Surgery found there is no correlation between…
Ellington: WBCT Improves Planning for restor3D Solutions
Dr. Kent Ellington, MD, a foot and ankle surgeon at OrthoCarolina, said he orders…
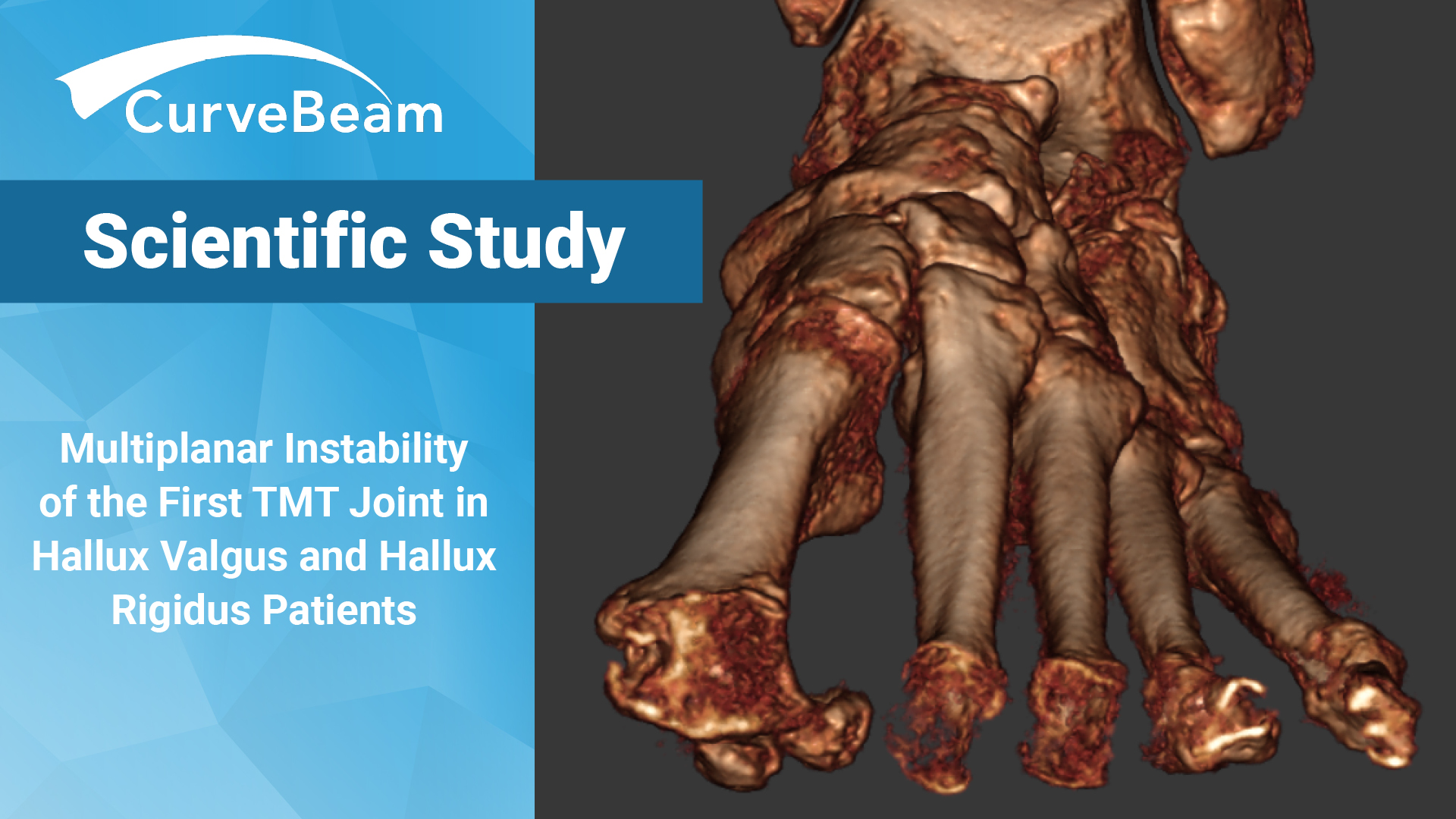
WBCT Confirms 1st TMT Instability in HV & HR Patients
Key Points Compared to controls, WBCT scans of hallux valgus (HV) & hallux rigidus (HR)…
WBCT is “Financially Viable” for Ortho Practices
Researchers from Campbell Clinic presented a poster on the cost effectiveness of WBCT for orthopedic…
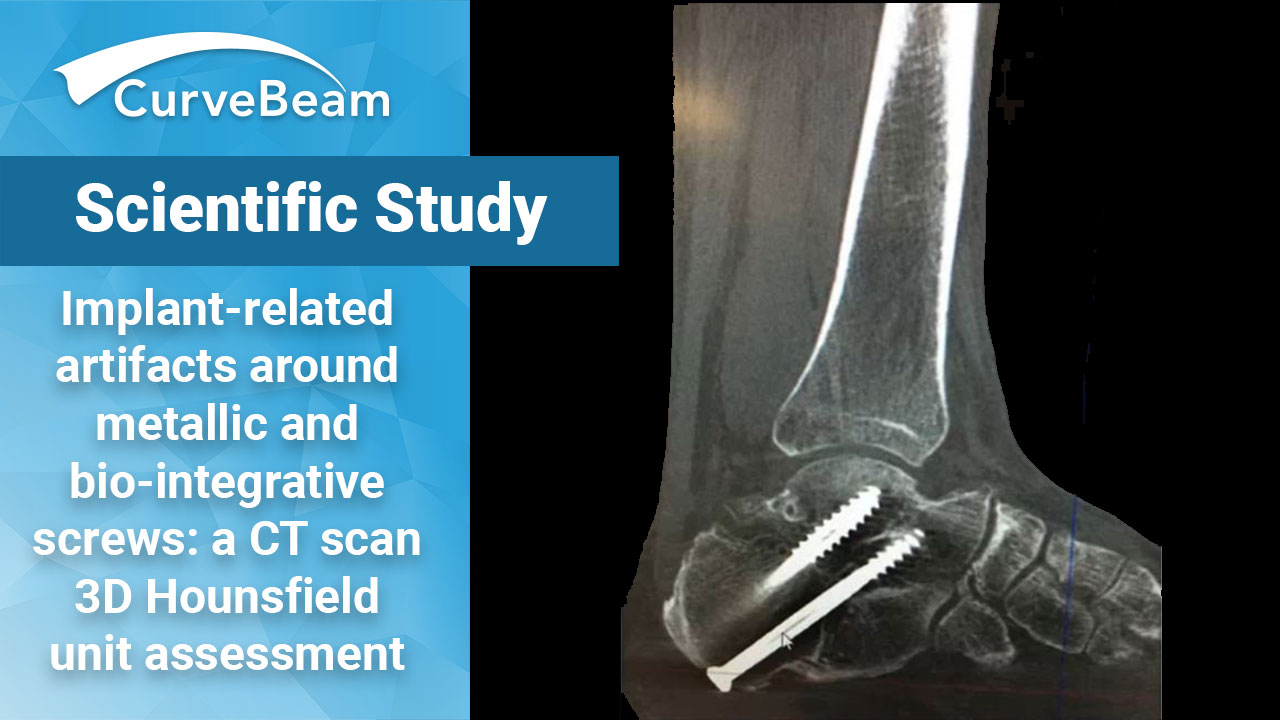
Bio-Screws Could Eliminate Need for MAR Protocols in Post-Op Imaging
Bio-integrative (BI) cannulated screws may be a preferred alternative to metallic implants to decrease implant-related…
Promoted Podcast: Device Nation w/ Joseph McGinley
Click here to listen on Apple Podcasts. Click here to listen on Spotify.
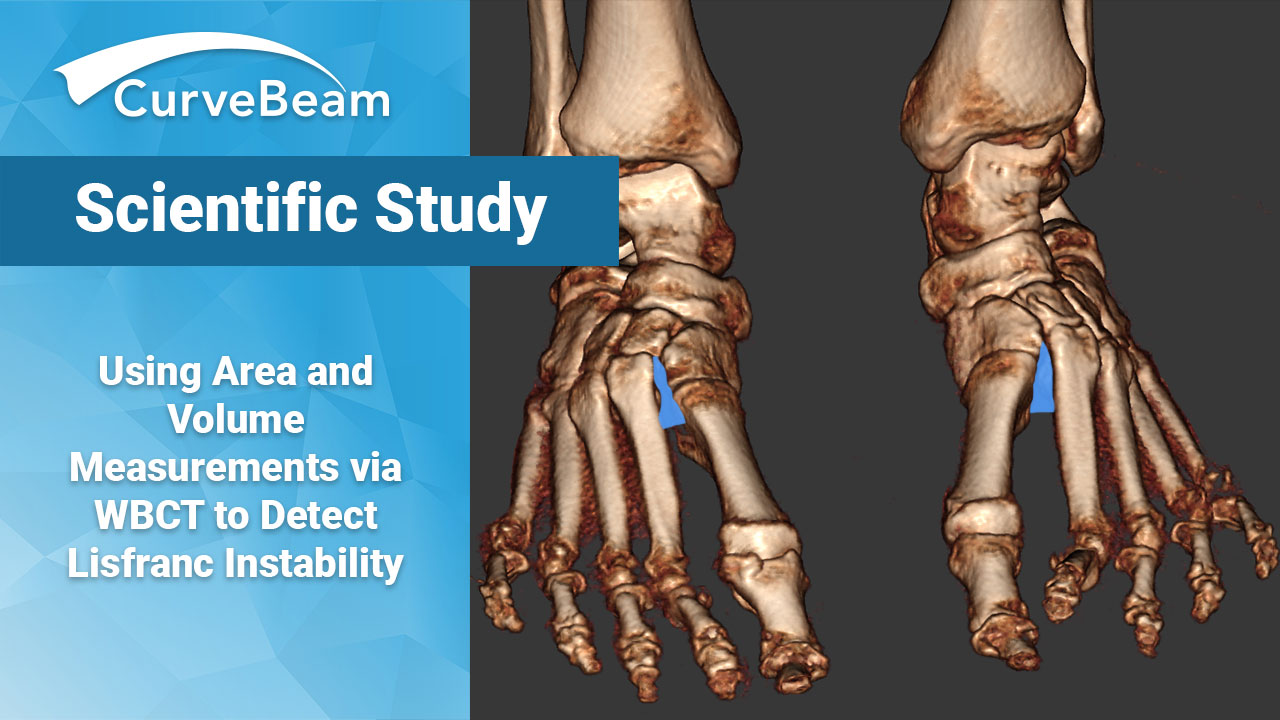
Using Area and Volume Measurements via WBCT to Detect Lisfranc Instability
Weight bearing CT (WBCT) can effectively differentiate between stable and unstable Lisfranc injuries, according to…
CurveBeam Announces Effort to Improve Insta-X Performance
AAOS attendees will get a first look at CurveBeam's improved digitally reconstructed radiographs, or…
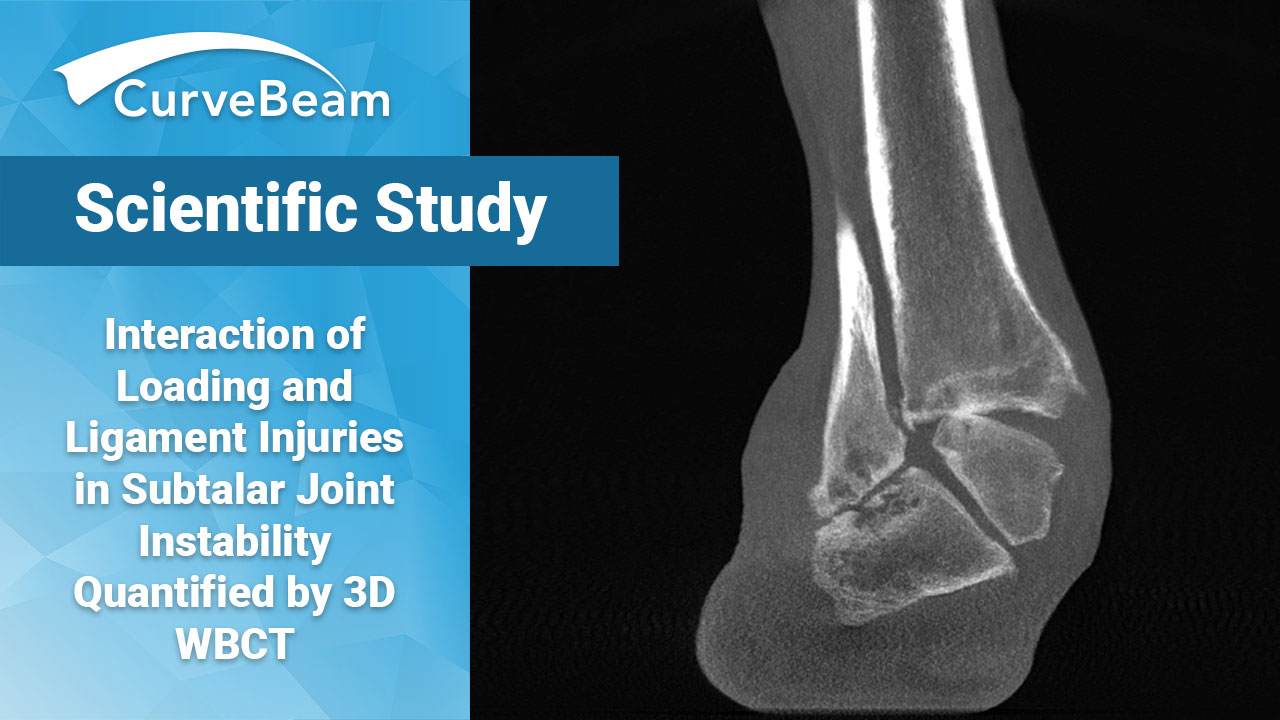
WBCT Reveals Subtalar Joint Instability Mechanisms
Are two-dimensional imaging techniques to blame for the diagnostic enigma of subtalar joint instability? An…
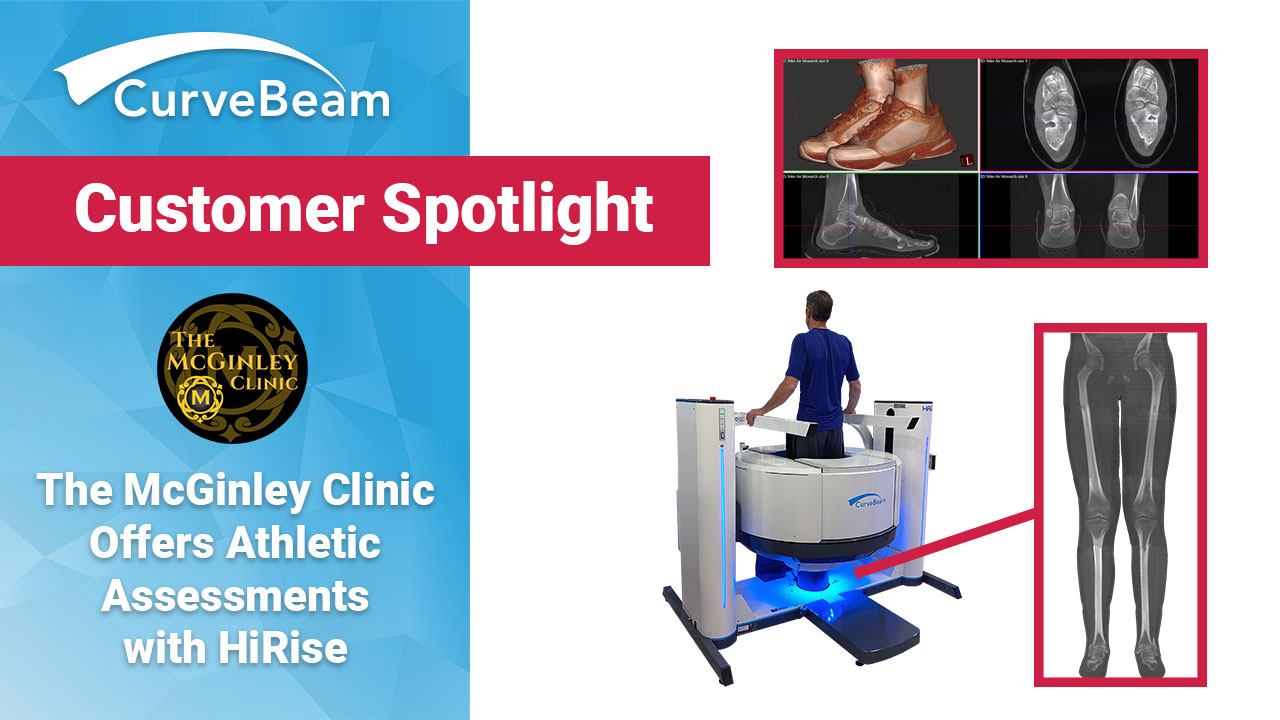
The McGinley Clinic Offers Athletic Assessments with HiRise
With the 2021 Olympic Games underway, The McGinley Clinic in Casper, Wyoming is letting athletes…
Dr. John Sigle: Tailor Bunionectomies to the Patient w/ WBCT Imaging
Weight bearing CT imaging is a useful clinical tool in the podiatric setting, especially when…
Videocast: Revealing the 3D Perspectives of Hindfoot Alignment with WBCT
Weight bearing CT imaging could change how surgeons approach hindfoot alignment (HA) & deformity. Multiple…
Researchers Define Normal Values for MT Rotation via WBCT Imaging
Key Points Researchers from RNOH Stanmore analyzed 182 feet via WBCT to define normal values…
Assessment of Hindfoot Alignment Comparing WB X-Ray to WBCT
Hindfoot Alignment View (HAV) radiographs, originally described by Saltzman and El-Khoury, have been the standard…
CBCT Increases Patient Turnover in ER by 23%, Study Finds
Introducing a cone beam CT (CBCT) device into a French emergency room’s radiology department resulted…
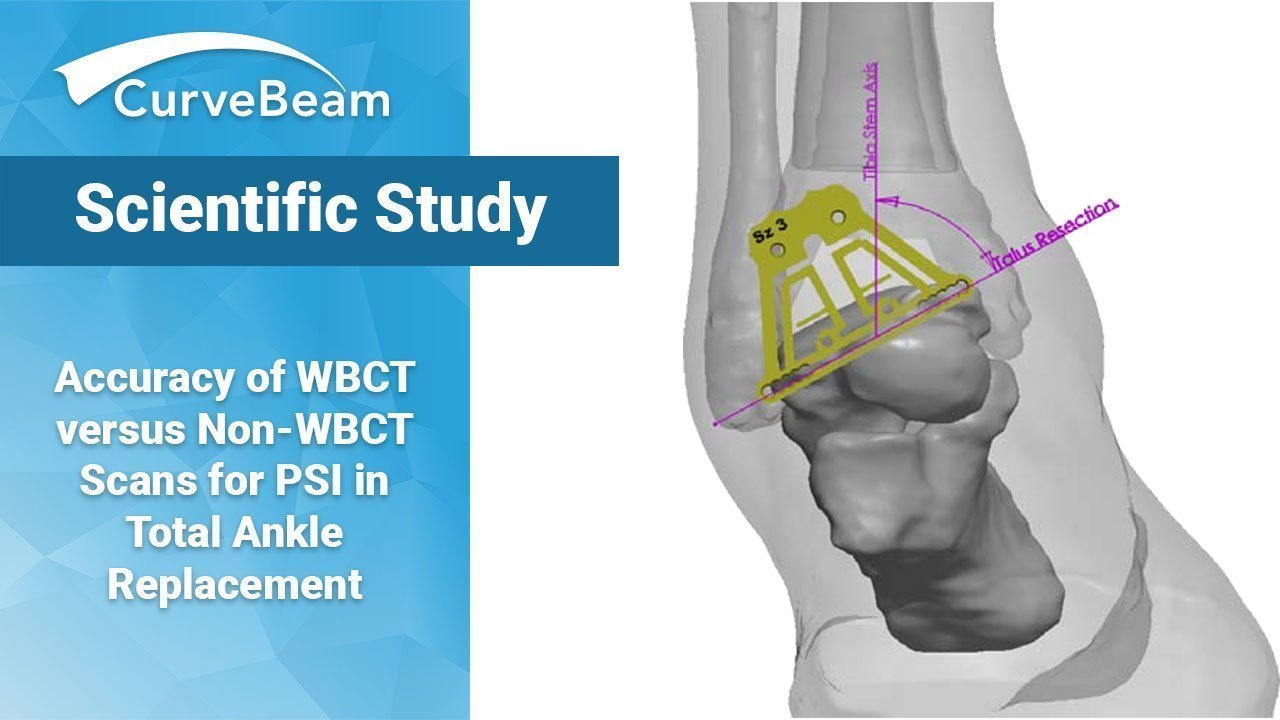
Accuracy of Weight Bearing CT versus Non-Weight Bearing CT Scans for Patient-Specific Instrumentation in Total Ankle Replacement
As Total Ankle Replacement (TAR) becomes the gold standard for end stage ankle arthritis, methods…
VIDEOCAST & LIVE Q&A: WBCT SOCIETY PRESIDENT-ELECT DR. FRANCOIS LINTZ REBRANDING WBCT
Join CurveBeam and Dr. Francois Lintz, President-Elect of the WBCT Society, to discuss the future…
Videocast: Is it Time to Rebrand WBCT as 3D X-Ray?
In CurveBeam's latest videocast, we sit down with Dr. Francois Lintz, MD, PhD, FEBOT, to…
Dr. Don Anderson: HiRise Helps Track Arthritic Joint Degeneration in the Knee
Donald D. Anderson, PhD is Vice Chair of Research, Orthopaedics and Rehabilitation at the University…
An Inside Look at CubeVue Autometrics
CubeVue Autometrics uses artificial intelligence to identify each individual bone and automatically calculates key biometrics…
Iowa Researchers Use WBCT to Investigate Achilles Tendinopathy and Affects of Heel Lifts
Assistant Professor of Physical Therapy and Rehabilitation Science, Ruth Chimenti, DPT, PhD, is researching the…
MedShape And CurveBeam Announce Initiation of Joint Prospective Clinical Study
Research will evaluate the efficacy of a sustained dynamic compression device using longitudinal weight-bearing CT…
CurveBeam Connect Videocast: Announcing CurveBeam Cloud
On this episode of the CurveBeam Connect videocast, host and CurveBeam Marketing Director Vinti Singh…
Scientific Literature: The New Standard? A Comparison of 2D and 3D Measurements of the Foot & Ankle Via Weight Bearing CT Imaging
A group of radiologists from the Netherlands and Italy are asking if 3D angle measurements…
CurveBeam Announces Development of CurveBeam Cloud
CurveBeam announced it has begun development on CurveBeam Cloud, a web-based platform that will simplify…
CurveBeam Weight Bearing CT Systems Offer Protocol for Paragon 28’s MAVEN Patient-Specific Instrumentation System
HATFIELD, PENN. – Apr. 6, 2021 – CurveBeam, a leader in weight bearing CT (WBCT)…
A Day in the Life at the University of Iowa’s Functional Imaging Lab
The University of Iowa’s Orthopedic Biomechanics Laboratories overarching mission is advancing “the application of innovative…
CurveBeam Weight Bearing CT Systems Offer Protocol for Exactech VANTAGE Ankle PSI
HATFIELD, PENN. – March 16, 2021 -- CurveBeam, a leader in weight-bearing CT (WBCT) imaging,…
CurveBeam HiRise Receives Regulatory Clearance in Canada
CurveBeam announced it has received Health Canada clearance for the HiRise weight bearing CT (WBCT)…
Webinar Highlights: Custom Insoles 3D Printed from WBCT Scans
The foot insoles industry is prime for disruption. Current methods are labor intensive, result in…
CurveBeam Connect: Innovative and Minimally Invasive – Meet Dr. Alireza Khosroabadi, Foot & Ankle Specialist
Dr. Alireza Khosroabadi, DPM, opened the Khosroabadi Institute in Los Angeles, CA to shift his…
Make Your Practice Stand Out with Extremity CT
Patients use technology in their everyday lives, and recognize when orthopedic practices make investments to…
Integrate Pre-Op CTs into Your Clinical Workflow
Provide a Personalized Assessment of Knee Arthritis Custom solutions for knee osteoarthritis (OA) deserve custom…
Philadelphia Inquirer Profiles CurveBeam President & CEO
The Philadelphia Inquirer featured CurveBeam's President & CEO Arun Singh on the front page of…
Improve Case Migration to ASCs with In-Office Extremity CT
Orthopedic leaders are embracing the migration of surgical cases to ambulatory surgery centers (ASCs). To…
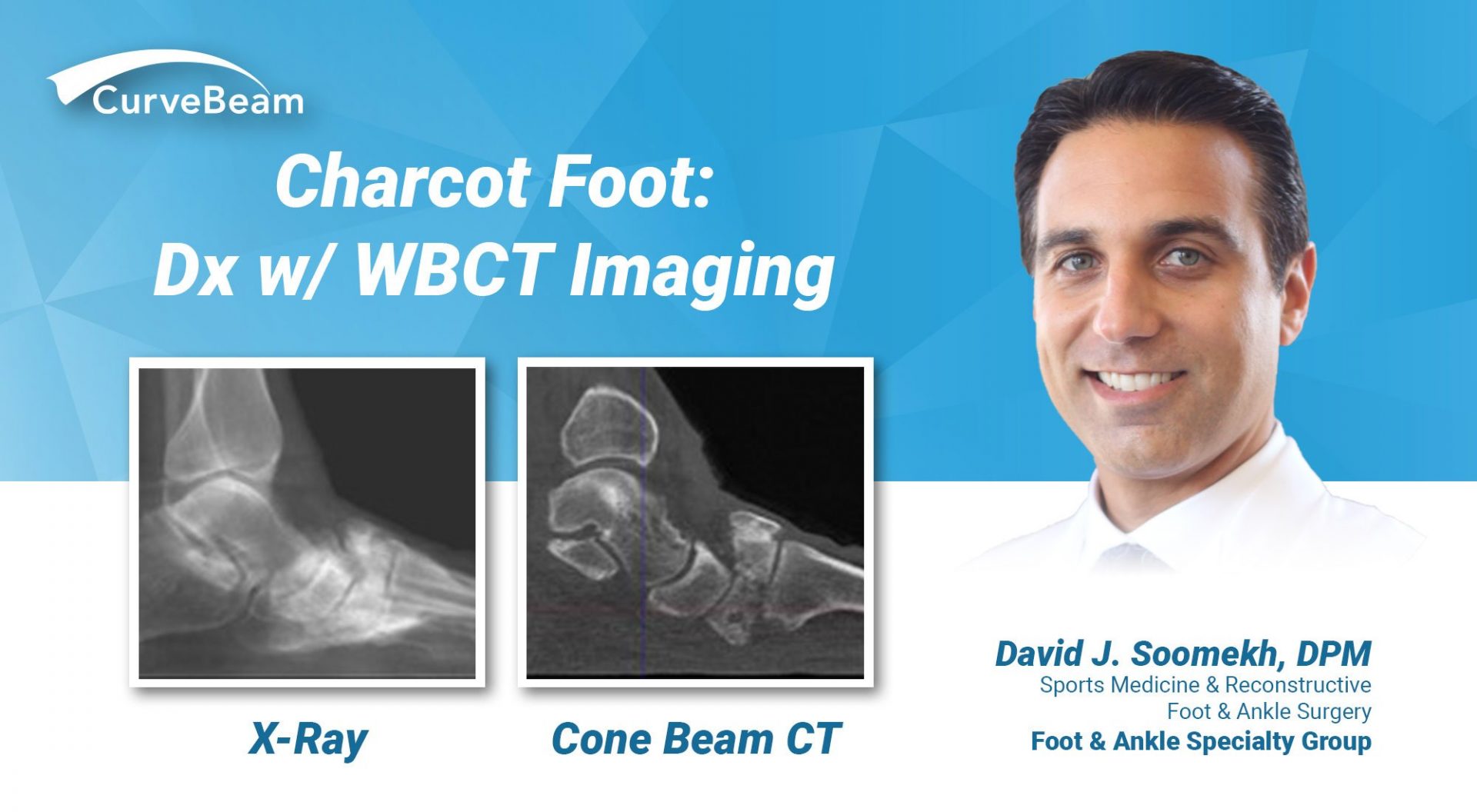
Charcot Foot: Dx w/ WBCT Imaging
By Dr. David J. Soomekh, DPM, FACFAS Guest Contributor Charcot foot is a complex foot…
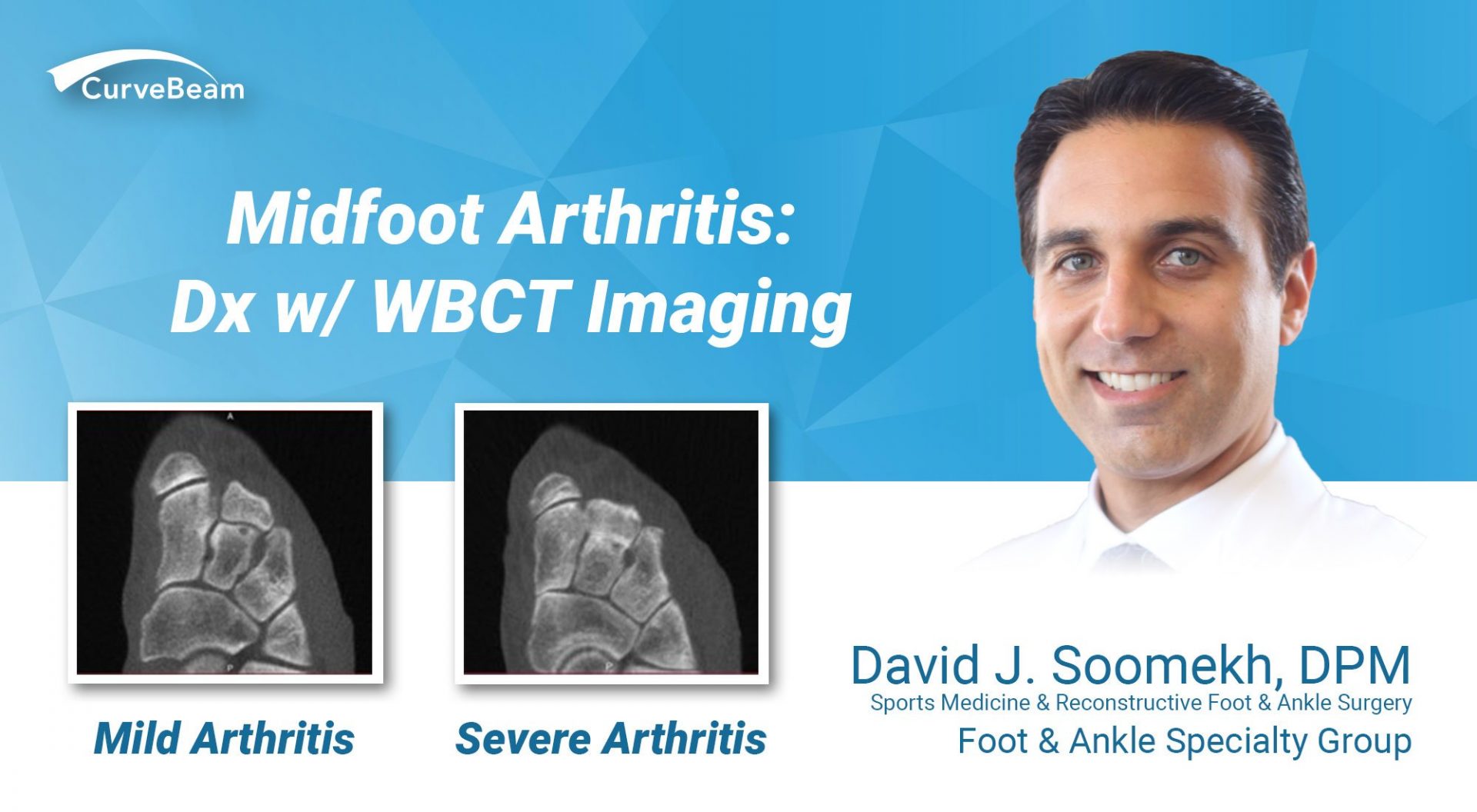
Midfoot Arthritis: Dx w/ WBCT Imaging
By Dr. David J. Soomekh, DPM, FACFAS Guest Contributor Osteoarthritis is the loss of cartilage…
Future-Proof Your Orthopaedic Clinic with Advanced Orthopaedic Imaging
Safe, consistent access to advanced diagnostic imaging can give orthopedic practices a competitive edge post-pandemic…
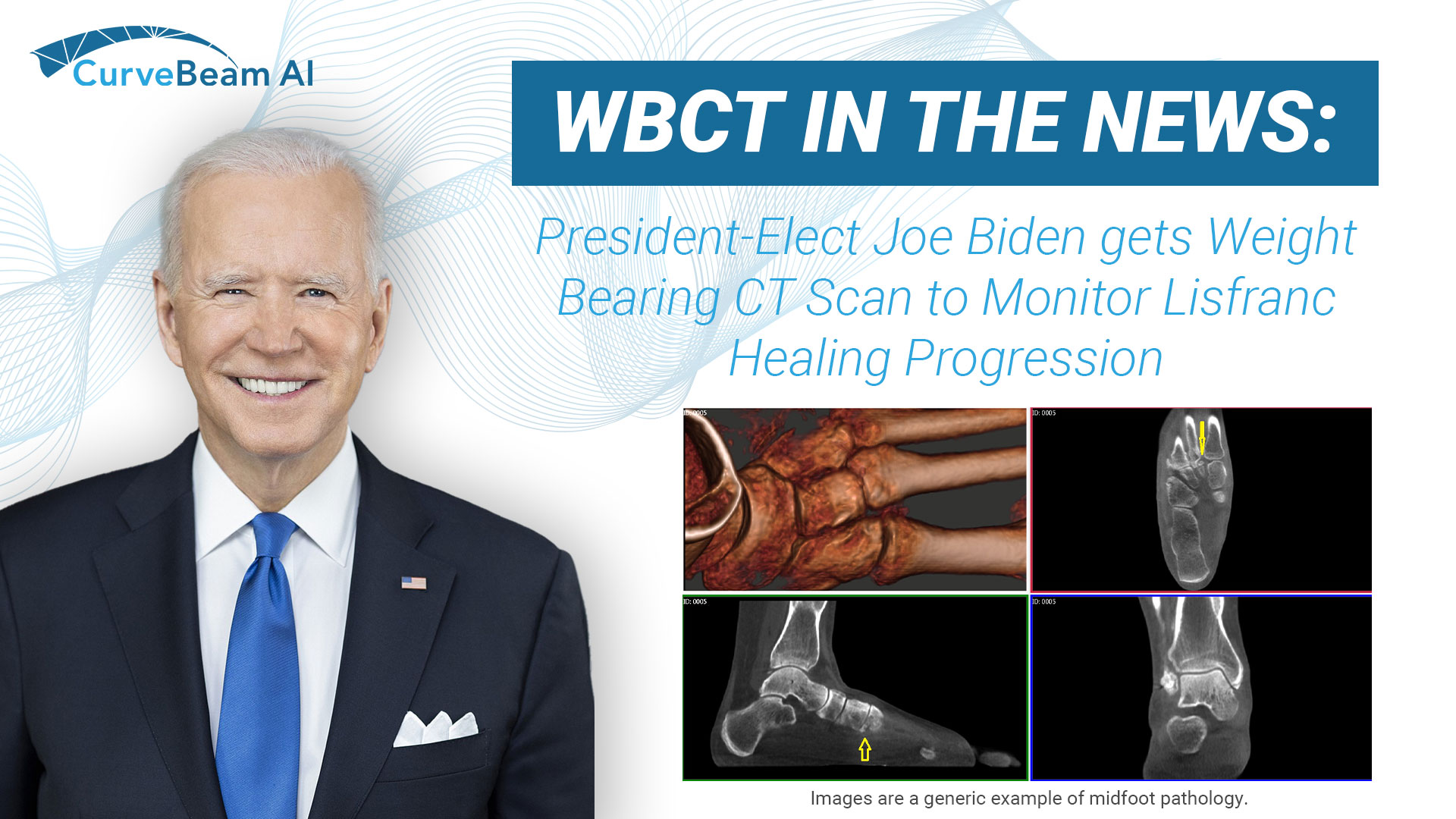
President Joe Biden gets WBCT Scan to Assess his Lisfranc Injury
President-elect Joe Biden's physician recommended he get a weight bearing CT scan to monitor the…
Right Test, Right Time – Ensure Your Ortho Practice is the Right Place
Modern medicine has made some major advances. Procedures that used to require a three-day inpatient…
Hairline Fractures: Dx w/ In-Office CT Imaging
By Dr. David J. Soomekh, DPM, FACFAS Guest Contributor “I think I have a hairline…
CurveBeam Connect: The Fantasy Doctor Breaks Down the Latest Trends in Foot & Ankle Injury Care
Dr. Selene Parekh, MD, MBA, wears several different hats in the orthopedic world…
First Weight Bearing CT System in Netherlands Is Accepting Patients
Healthcare professionals and patients in the Netherlands now have access to weight bearing CT imaging…
Improve TKA & Osteotomy Planning w/ Full Leg WBCT
Preoperative plans for arthroplasty and corrective osteotomies rely on a weight-bearing long leg AP X-Ray…
CurveBeam Announces FDA 510(k) Clearance for HiRise Weight Bearing CT System for the Entire Lower Extremity
CurveBeam announced the HiRise, its weight bearing CT (WBCT) imaging system for the entire lower…
Promoted Podcast: Device Nation Features Dr. Kent Ellington
Dr. Kent Ellington, an OrthoCarolina Foot and Ankle Surgeon and team physician for NASCAR’s Stewart-Hass…
Webinar Highlights: Designing a Mobile Imaging Suite to Serve Multi-Location Practices
In a recent CurveBeam Academy webinar, Turner Dean, COO of CurveBeam Mobile, explored the doors…
CurveBeam Announces Certification for HiRise WBCT System for Entire Lower Extremities
CurveBeam announced the HiRise, its weight bearing CT imaging system for the entire lower extremity,…
Benefits of InReach over Traditional X-Ray by Dr. Gaston
Dr. Glenn Gaston. MD presented the benefits of InReach in-office CT imaging in a CurveBeam…
Webinar Recap: Radiologist Sajid Butt Presents Basics of CBCT
Dr. Sajid Butt, Consultant Musculoskeletal Radiologist for the Royal National Orthopedic Hospital in the United…
Dr. Soomekh: How CT Became My Primary In-Office Modality
Dr. David Soomekh is a board-certified foot and ankle specialist and surgeon and founder of…
Standing CT: Enabling Treatment & Triage for Orthopedic Patients During COVID-19
To access the full-length webinar, please click here. COVID-19 is still causing ripple effects across…
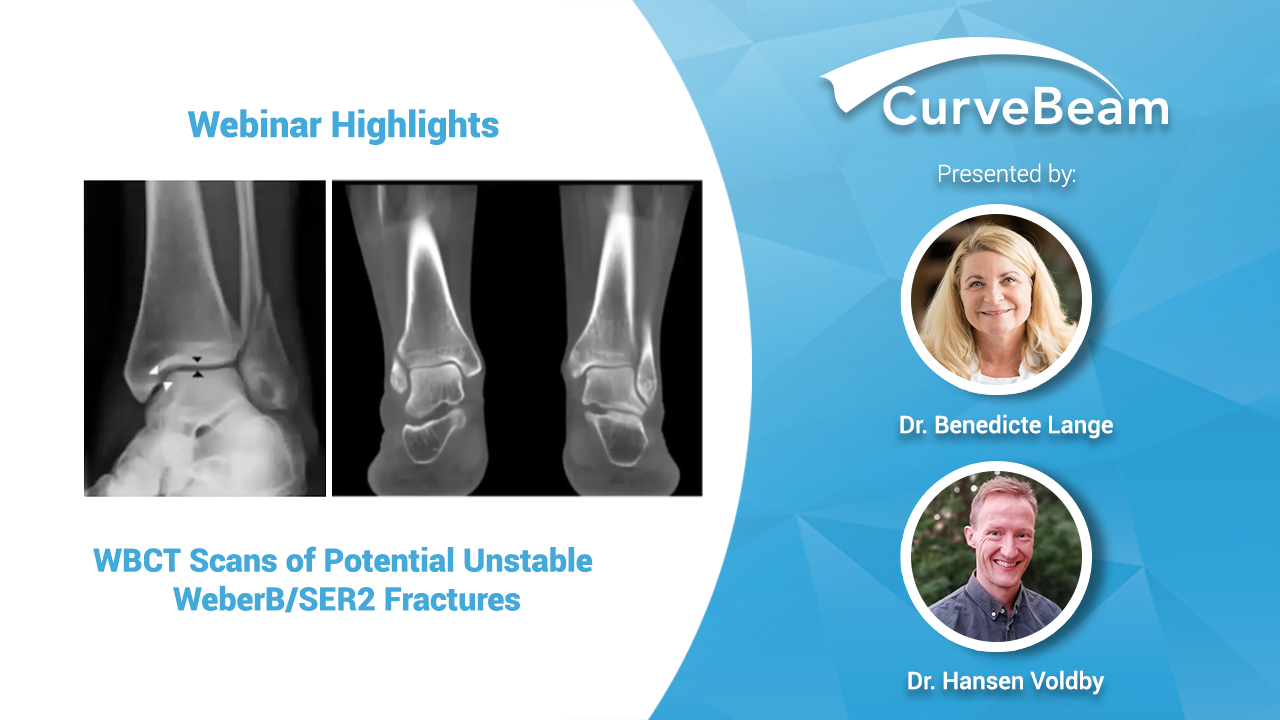
Webinar Recap: WBCT Scans of Potentially Unstable WeberB/SER2 Fractures
In a recent CurveBeam webinar held during the ECR Virtual Congress, Radiologist Dr. Benedicte Lange…
Curvebeam Connect: Robot Surgeons Aren’t Here, But Innovation Is
Dr. Chris O’Grady wants to see joints. The Florida-based Orthopedic Surgeon who specializes in shoulder…
CurveBeam Announces Patent for TALAS 3D Hindfoot Measurement Tool
CurveBeam has formally been issued a patent (both in the U.S. and internationally) for TALASTM, or…
CurveBeam Announces Trial Site for the HiRise Weight Bearing CT System for Entire Lower Extremities
CurveBeam announced a top research university has received Institutional Review Board approval to conduct clinical…
CurveBeam Connect: Educating Lawmakers About Cone Beam CT
Belgian surgeons started using weight bearing cone beam CT several years ago. Then, radiologists like…
Video: During COVID-19, Mobile 3D Imaging Provides Uninterrupted Care for Ortho Patients
When the coronavirus halted elective care at his UK hospital, orthopedic surgeon Mr. Matthew Solan…
CurveBeam Connect: WBCT Imaging of the Diabetic Foot with Dr. Jarrett D. Cain, DPM, FACFAS
Dr. Jarrett Cain, DPM, FACFAS knows he’s in a unique position. As a researcher at…
WBCT Imaging Assists with Preoperative Planning for Total Talar Replacement
Dr. Kent Ellington, MD, MS, FAAOS, a foot and ankle orthopedic surgeon at OrthoCarolina, discussed…
CurveBeam Connect: Cone Beam CT Mobile Unit Deployed to Manage Orthopedic Imaging During COVID-19
Doctors all over the world have been pressed into duty taking care of patients with…
CurveBeam Connect: T-Soles Set to Disrupt Insole Industry
The Netherlands may be famous for their wooden shoes, but with an estimated four million…
The Importance of Dynamic Imaging in Facilitating Biomechanical Research Outcomes
The Queen’s University Skeletal Observation Laboratory in Kington, Ontario, Canada, is exploring some critical topics…
COVID-19: Redefining Telemedicine’s Role in Orthopedics
Dr. Francois Lintz, an orthopedic foot and ankle senior consultant at UCP Toulouse…
CurveBeam Connect: The AI Advancements that Could Change Orthopedics Forever
Mention AI in a clinic, and many people’s minds go to the idea of robotic…
Dr. Robert Santrock: Using LineUP WBCT Scans with Wright PROPHECY
Watch Dr. Robert Santrock, Chief of Foot & Ankle Surgery at West Virginia University, discuss…
Webinar: WBCT – Financial and Operational Perspectives
Register today to participate in a virtual lecture delivered by Dr. Calvin Rushing, DPM, who…
CurveBeam Connect: Accreditation Options for Cone Beam CT Imaging
Garry Carneal, JD MA, is the President and CEO of RadSite, a CMS accrediting body for…
Queen’s University: Using LineUP to Better Understand Human Mobility
At the Queen’s University Human Mobility Research Lab, they’re studying some important and fundamental questions…
Low-Dose CT: No Effect on DNA
Low-dose CT scans have been proven to be effective in the diagnosis and treatment of…
CurveBeam Connect: New Software Developments Help Provide Superior Orthopedic Recovery and Care
Technology is changing the face of healthcare, especially in the sector of orthopedic rehabilitation. Amp Recover,…
Virtual Reveal Event: CubeVue Autometrics
CurveBeam Set to Offer Virtual Look at Latest Innovations With the recent cancellation of AAOS…
Virtual Reveal Event: HiRise – The Next Level of Weight Bearing CT Imaging
Register today to participate in a virtual lecture delivered by Vinti Singh, Marketing Manager for CurveBeam, who will demonstrate…
Virtual Session: CurveBeam Mobile for Centers with Multiple Locations
Register today to participate in a virtual lecture delivered by Turner Dean, President & COO of CurveBeam…
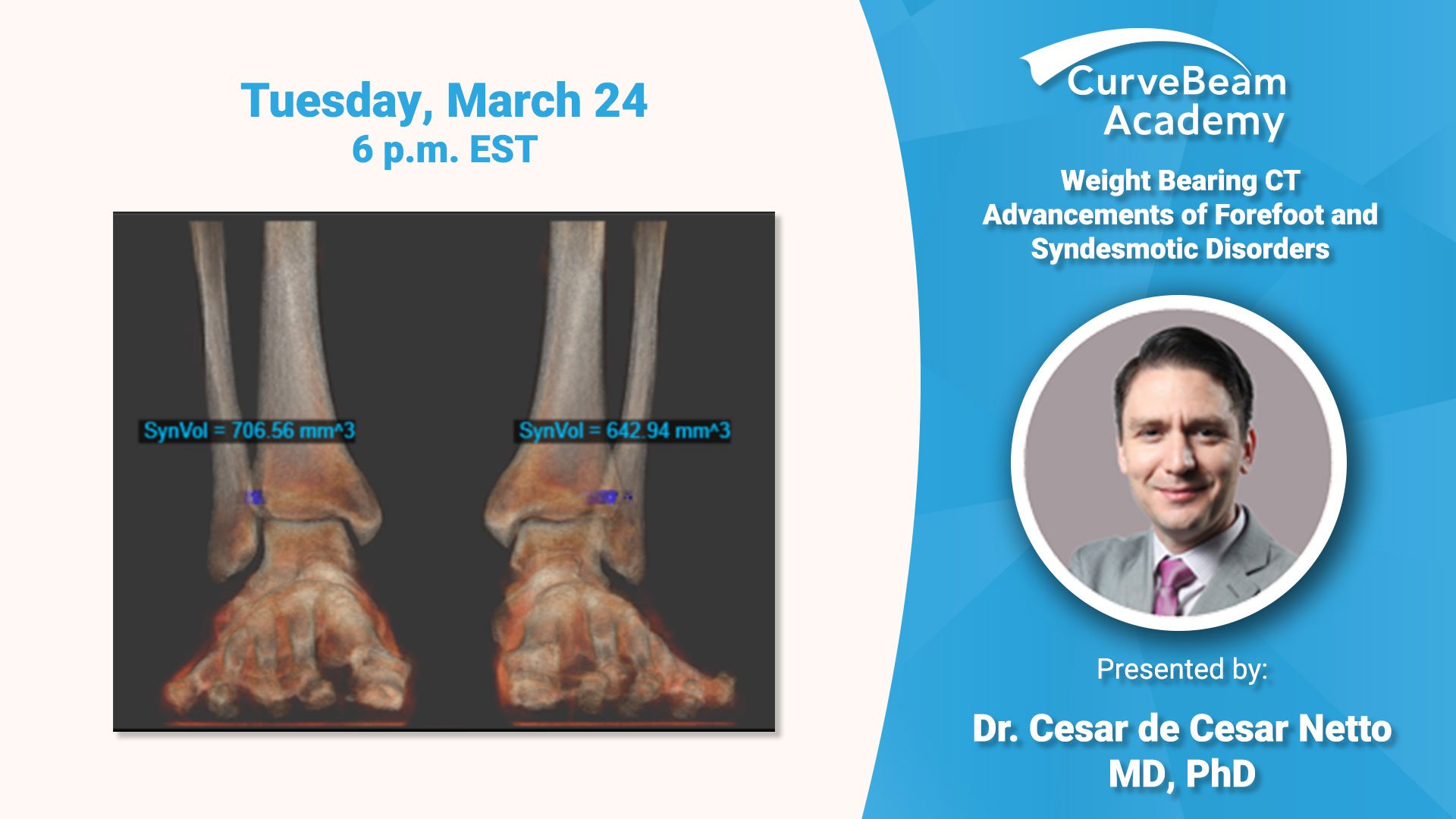
Virtual Lecture: Weight Bearing CT Advancements of Syndesmotic Disorders
Register today to participate in a virtual lecture delivered by Dr. Cesar de Cesar Netto, MD, PhD, of Univerity…
Virtual Lecture: Monitoring the Development of Cyst Formation After TAR
Register today to participate in a virtual lecture delivered by Dr. Francois Lintz, MD, FEBOT, of Clinique de…
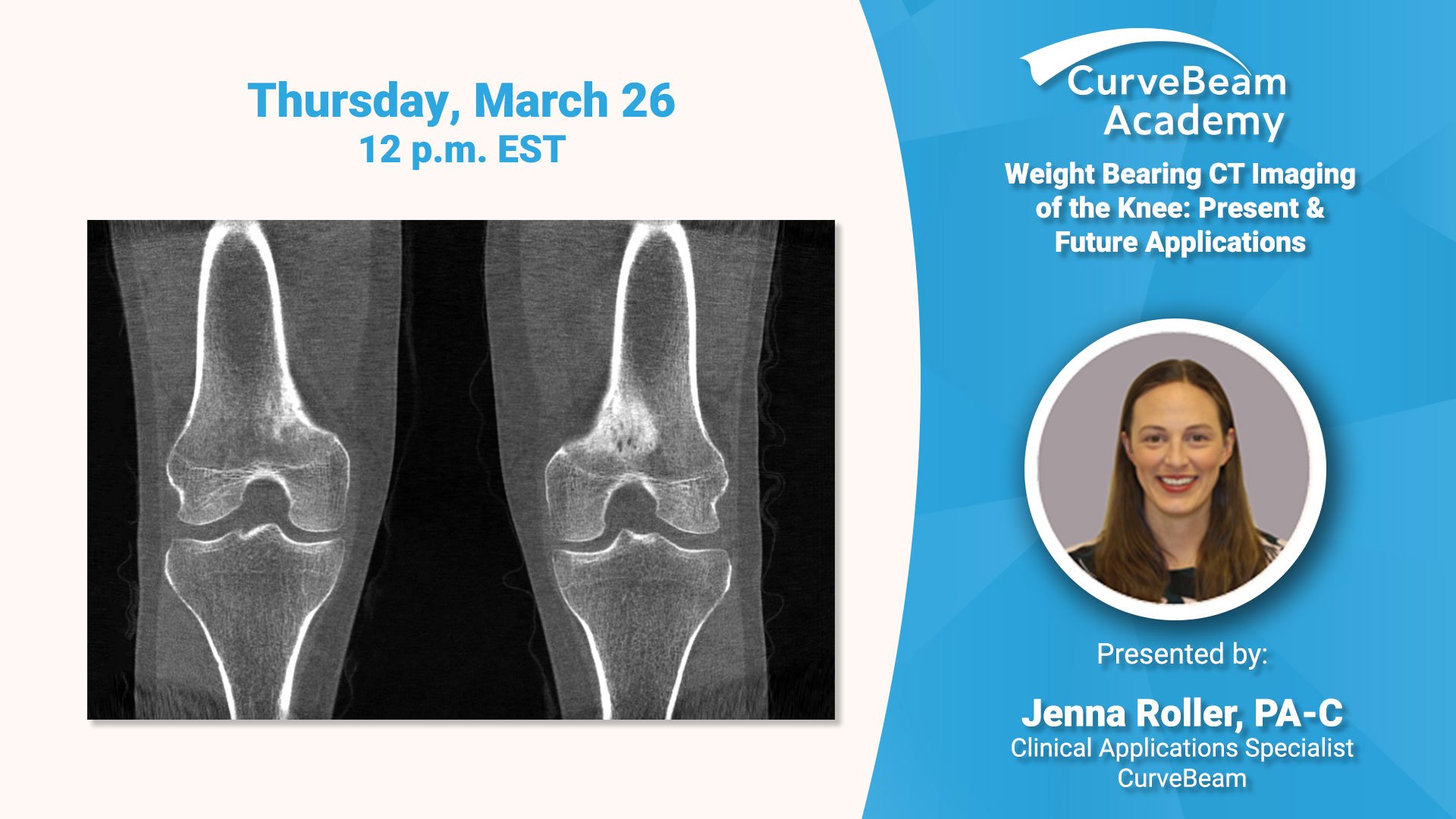
Virtual Lecture: WBCT of the Knee: Present & Future Applications
Register today to participate in a virtual lecture delivered by Jenna Roller, PA-C, Clinical Applications Specialist for…
Virtual Lecture: In-Office CT for Upper Extremity Conditions
Register today to participate in a virtual lecture delivered by Dr. Glenn Gaston, MD, an…
CurveBeam Announces Virtual Conference March 23 – 26
You are invited to attend CurveBeam Academy, an informative 3-day event featuring CurveBeam Reveal Events…
CurveBeam Connect: Presenting New Ideas and Challenging Old Ones
If CurveBeam was a band, it’d be the Foo Fighters. How’s that for a hook?…
CurveBeam HiRise – Live Demo of the Next Generation of WBCT Imaging
At CurveBeam, we pride ourselves on being at the forefront of weight bearing CT (WBCT)…
Cone Beam CT Helps Visualize Loose Bodies
A loose body is a bone or cartilage fragment that has chipped off inside a…
FOOTInnovate Webinar Recap: “Seeing 2020: My First 6 Months with Weight Bearing CT” with Dr. Blake E. Moore, MD, FAAOS
Dr. Blake E. Moore, MD, FAAOS, recently delivered a FOOTInnovate webinar titled “Seeing 2020: My…
3D Printing Custom Surgical Guides with Dr. Kristian Buedts, MD
Custom surgical guides help improve placement and surgical success. Dr. Kristian Buedts, MD, a foot…
FOOTInnovate Webinar Recap – Soomekh: How I Have Integrated Weight Bearing CT into My Practice
David J. Soomekh, DPM, is a Diplomate of the American Board of Foot and Ankle…
FOOTInnovate Webinar Recap: Weight-Bearing CT as a Diagnosis Tool in Podiatric Radiology, Dr. Albert Armstrong DPM
Albert Armstrong, DPM, MS, BSRS, Professor of Radiology and Medical Director of Advanced Imaging at…
CurveBeam’s Automatic IMA Angle Measurement Tool, Presented by Dr. Francois Lintz, MD, FEBOT, PhD
At the 2019 AOFAS Annual Meeting in Chicago, Dr. Francois Lintz, MD, FEBOT, PhD showcased…
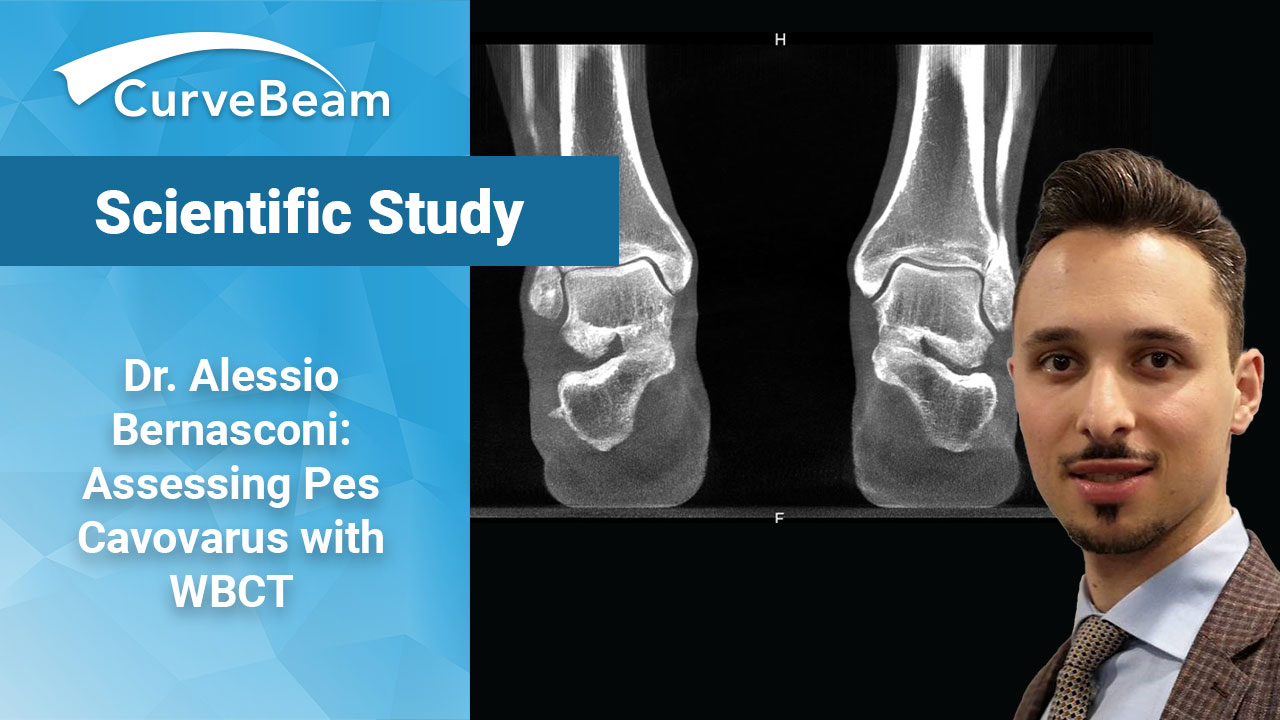
FOOTInnovate Webinar Recap: Dr. Alessio Bernasconi, “3D, Weight-Bearing CT’s Role in Assessing Pes Cavovarus”
Dr. Alessio Bernasconi, MD, FEBOT and his colleagues at the Royal National Orthopaedic Hospital in…
FOOTInnovate Lecture Preview: Dr. Blake Moore
Be sure to register for Dr. Blake Moore, MD, FAOOS' upcoming webinar on FOOTInnovate, titled…
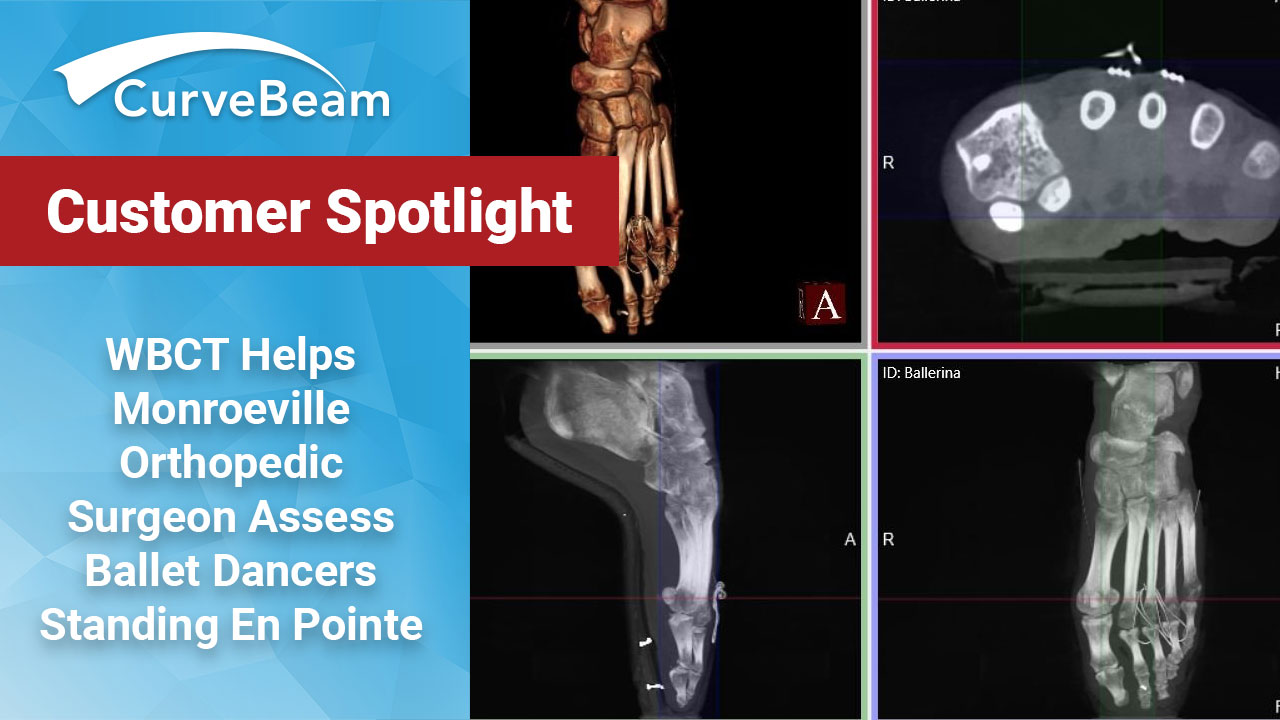
Weight Bearing CT Helps Monroeville Orthopedic Surgeon Assess Ballet Dancers Standing En Pointe
As a soloist for the Pittsburgh Ballet Theater, dancer Diana Yohe, 25, has sprained her ankle…
International WBCT Society Announces Book Release
The first textbook on weight bearing CT imaging, written by the board of the International…
CurveBeam Announces Development of Weight Bearing CT Imaging System that will Scan the Hip and Pelvis
CurveBeam is proud to introduce the next level of weight bearing CT imaging, which will…
CurveBeam AI Connect: Hand, Wrist & Elbow Injury Recovery Methods in Sports Medicine
How do professional athletes recover from sports injuries and what are the advancements in sports…
RSNA Preview: The Next Level of Weight Bearing CT Imaging
CurveBeam is going to the next level. Learn more at the RSNA Annual Meeting at…
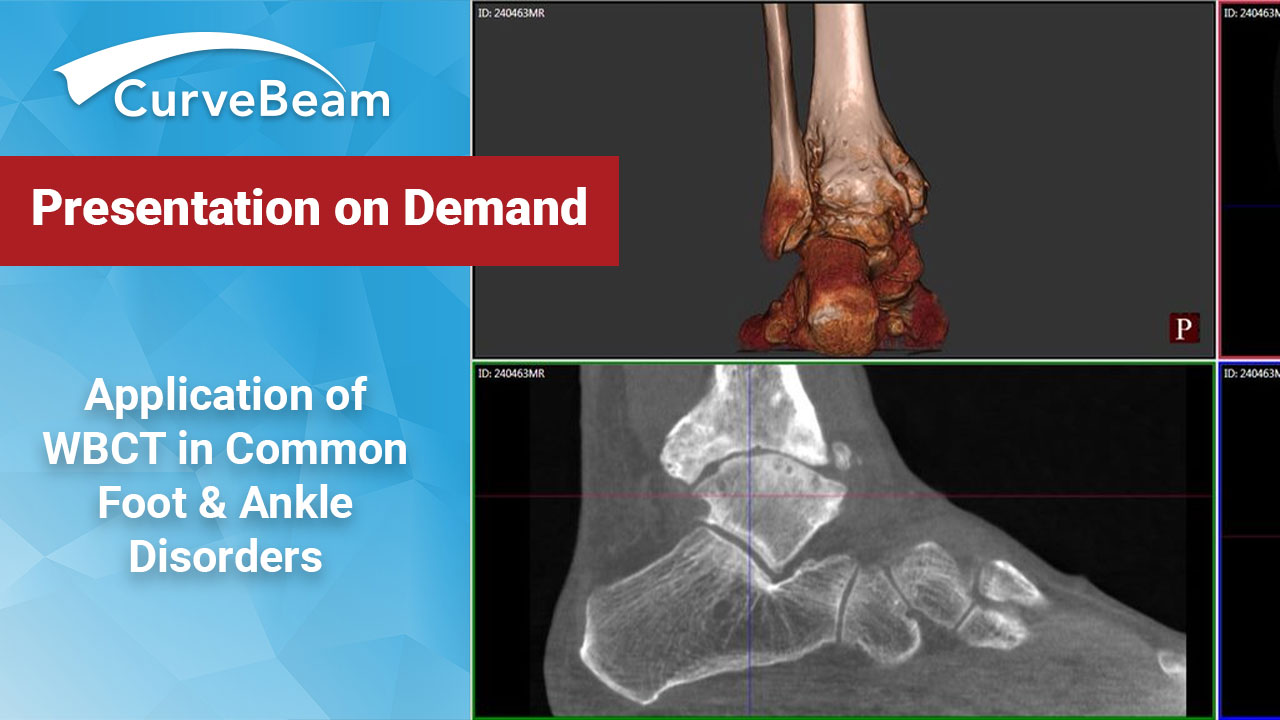
FOOTInnovate Webinar Recap: Application of Weightbearing CT (WBCT) in Common Foot & Ankle Disorders
Applications of Weight Bearing CT in Common Foot & Ankle Disorders (Click here to access…
CurveBeam Connect: Critical Thinking in Regulatory Affairs with Ryan Conlon
On this CurveBeam Connect Jobcast episode, Director of Regulatory Affairs Ryan Conlon said the most…
FOOTInnovate Webinar Preview: Dr. Albert Armstrong
Don't miss Dr. Albert Armstrong's forthcoming webinar titled "Podiatric Radiology – Weight Bearing CT Imaging…
IFFAS Award of Excellence Goes to Study Using Weight Bearing CT to Examine Residual Effects of Misalignment in TAR Patients
An international research team of orthopedic surgeons was awarded the prestigious International Federation of Foot &…
Press Release: CurveBeam Announces New Validated Protocols for LineUP™ Weight Bearing CT Imaging System
LineUP System Now Compatible with Wright Medical’s PROPHECY™ Preoperative Navigation System and Portfolio of Total…
CurveBeam Connect: The Humanitarian Effort with a Growing Global Footprint
When we sprain our ankle or break a toe, it’s assumed that proper care is…
CURVEBEAM CONNECT: South American Orthopedics is Making Strides with Dr. Cristian Ortiz
The foot & ankle orthopedic specialty in South America is burgeoning as the number of foot…
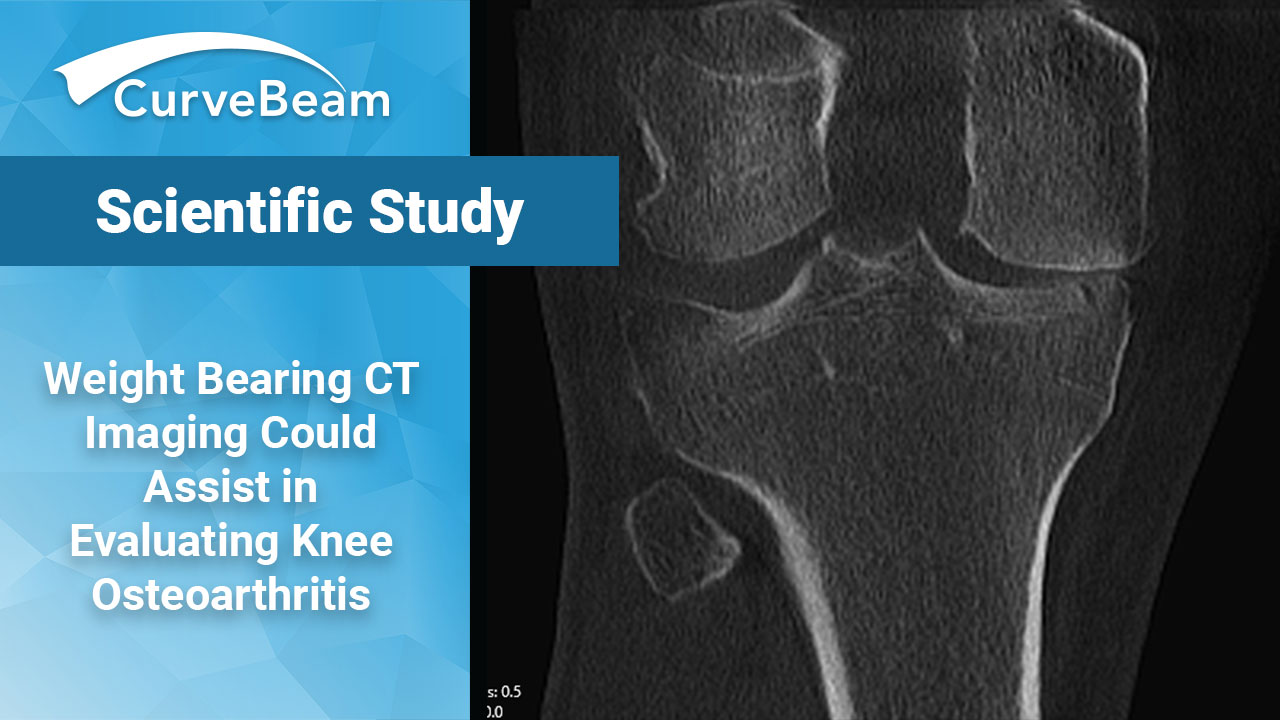
Weight Bearing CT Imaging Could Assist in Evaluating Knee Osteoarthritis
Many have long thought that the means of assessing osteoarthritis in the knee are less…
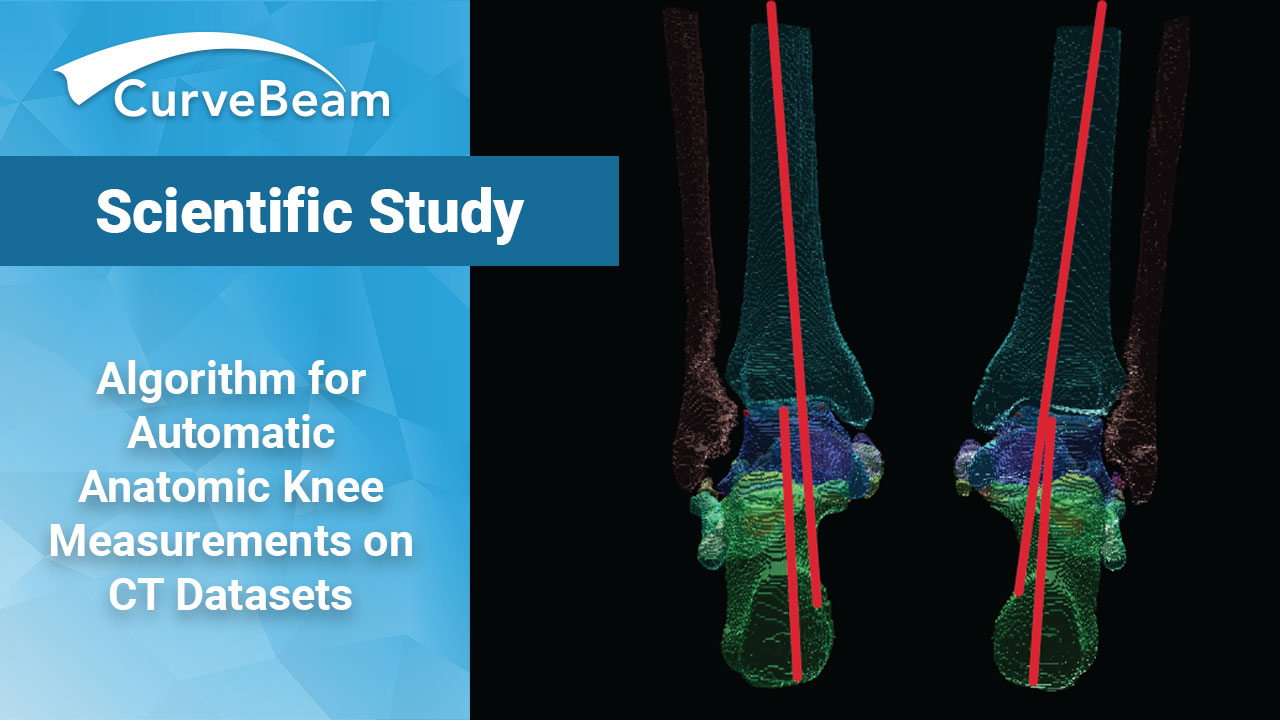
Researchers Develop Algorithm for Automatic Anatomic Knee Measurements on CT Datasets
Researchers at Johns Hopkins University have created an algorithm for automatic anatomical measurements in tomographic…
CurveBeam Connect: Breaking Down a New Study on Weight-Bearing CT’s Impact on Cost
In this episode of the CurveBeam podcast, host Vinti Singh sat down with Dr. Martinus Richter,…
AAOS: “Ortho Surgeons are Fully Competent to Interpret Imaging Studies”
Imaging technology has increased the convenience and quality of the diagnosis and treatment of musculoskeletal…
Medical Care May Have Swayed Kevin Durant’s Decision to Join the Nets
What’s worse than losing your patient’s confidence? Probably having it announced throughout national media coverage…
CURVEBEAM CONNECT: Getting to Know Dr. Cesar de Cesar Netto
For a premier orthopedic surgeon, you might be surprised to learn Dr. Cesar de Cesar…
El Equipo de Chile y la Clínica de la Universidad de los Andes Firman Acuerdo para el Cuidado de Atletas de Alto Rendimiento
En Abril, el Comité Olímpico de Chile firmo un nuevo acuerdo con la Clínica de…
Luminary South American Hospital Named Official Recovery Site for Chilean Olympic Athletes; Will Provide Weight Bearing CT Imaging
The Olympic Committee of Chile signed a new agreement with the Clinica Universidad de los…
CURVEBEAM CONNECT: A FOCUSED APPROACH TO TECHNOLOGY IN HEALTHCARE
Dr. Bob Baravarian Advocates a Focused Approach to Technology in Healthcare Dr. Babak (Bob) Baravarian…
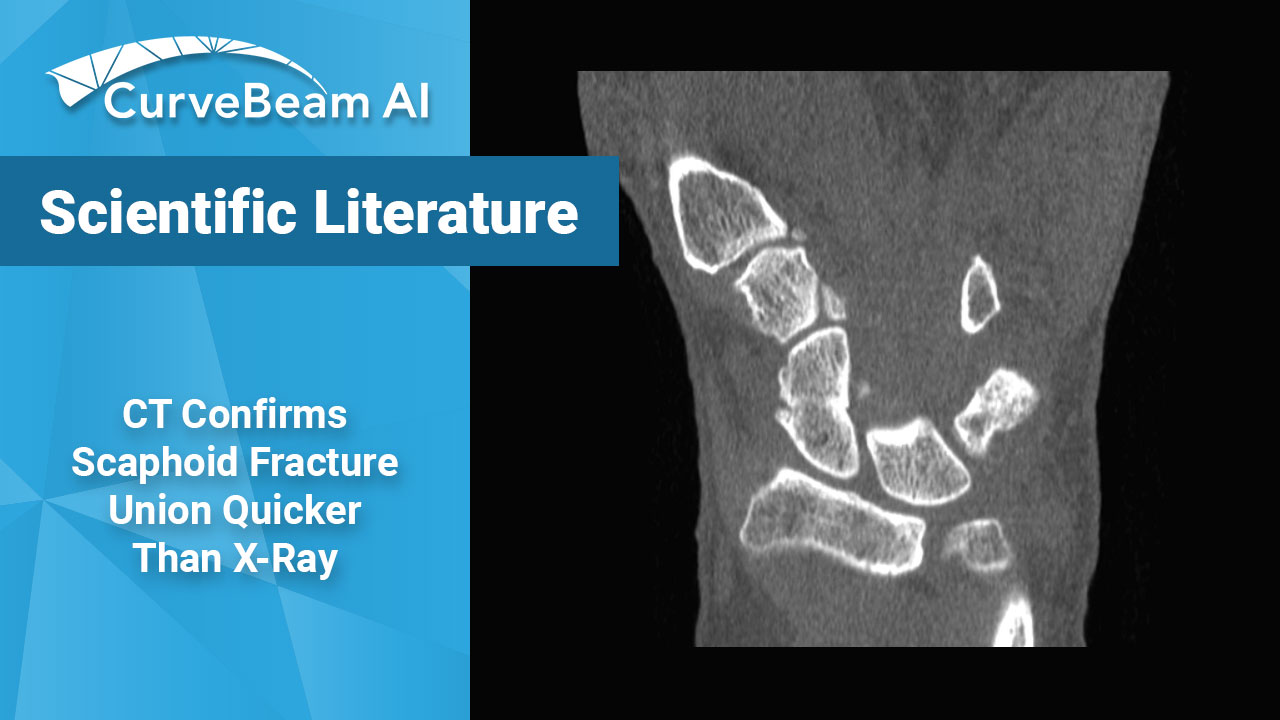
Cone Beam CT Confirms Scaphoid Fracture Union Quicker than X-Ray
Cone Beam CT (CBCT) is more accurate than X-Ray for predicting scaphiod healing at early…
Podcast: Interview with James Kraft, Founder of Standing CT Company
Weight-bearing CT scans have many benefits when compared to a weight-bearing X-ray. The…
WEBINAR: “Role of WBCT in the Assessment of Pes Cavovarus”
CurveBeam’s next FOOTInnovate webinar “Role of WBCT in the Assessment of Pes Cavovarus" will be delivered by Dr…
Study: Weight Bearing CT leads to better injury identification in NBA players
For professional athletes, injuries mean reduced playing time, impacted performance, and, in rare cases, an…
CURVEBEAM CONNECT: THE FUTURE OF SYNDESMOSIS EVALUATION
On this month’s edition of CurveBeam Connect, Vinti Singh, Director of Marketing at CurveBeam interviews Alexej Barg, M.D.,…
Webinar: “How I Integrated WBCT Into My Private Practice”
Sign up for CurveBeam’s next Corporate Webcast on FOOTInnovate.com. In his lecture titled, “How I…
pedCAT saves 800+ Hours of Imaging Time a Year
A study found that performing 2D radiographs & traditional medical CT imaging for foot &…
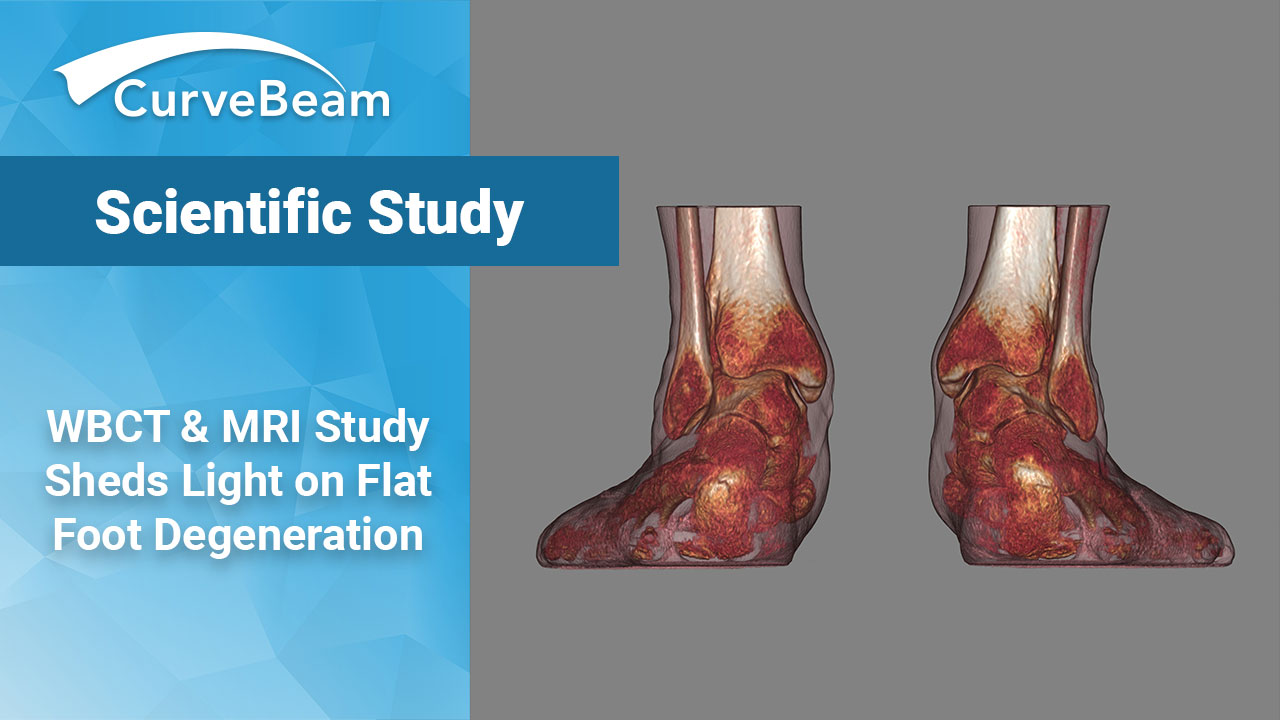
WBCT & MRI Study Sheds Light on Flat Foot Degeneration
Some degree of subtalar joint subluxation, as well as sinus tarsi impingement, was found in…
Demehri: WBCT Preferable Over MRI for Syndesmosis
Injury to the syndesmosis often requires advanced imaging. Up until recently, the most common advanced…
CurveBeam Connect Episode 1 – Dr. Francois Lintz, MD
CurveBeam is thrilled to announce the launch of its official podcast - CurveBeam Connect. Each…
Webinar: Incorporating Weight Bearing CT into Daily Practice
On Thursday, February 28, 2019, at 9 pm EST, CurveBeam will be sponsoring an…
Physicians Footcare and CurveBeam Mobile Hit the Road
CurveBeam Mobile makes weight-bearing CT imaging accessible and affordable to all providers, regardless of practice size…
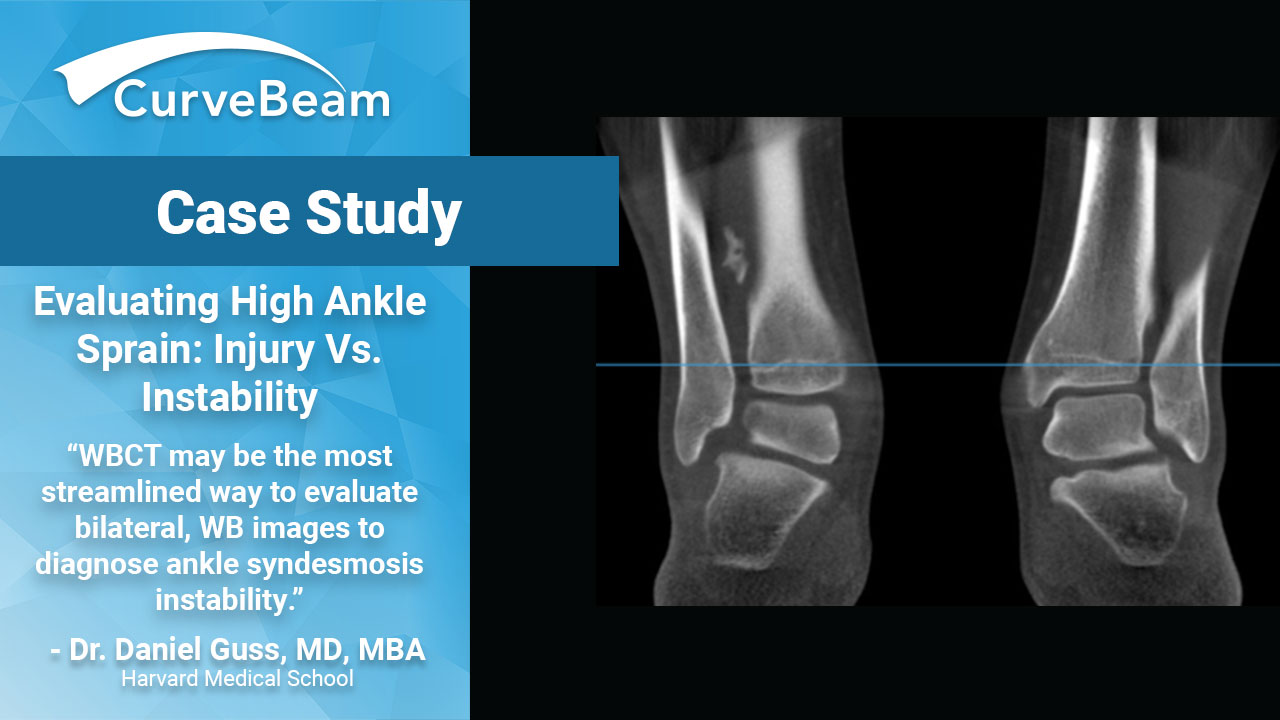
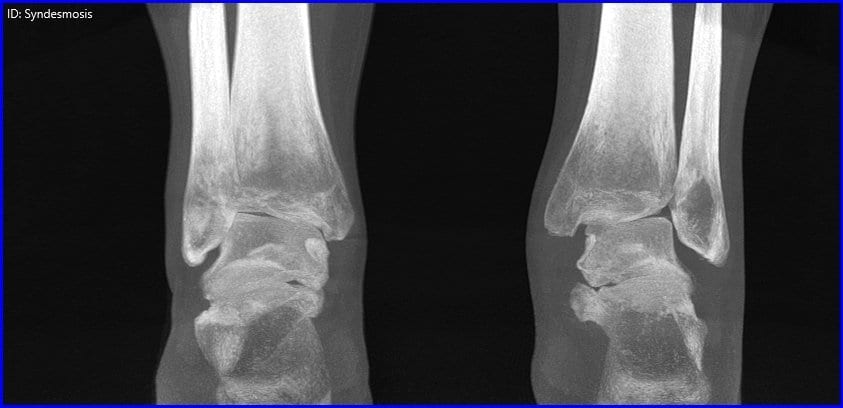
Evaluating High Ankle Sprain: Injury vs. Instability
Syndesmosis – A Functional Injury Ankle syndesmosis injury, commonly known as high ankle sprain, is…
Webinar: Applications of WBCT by Dr. Daniel Cuttica, D.O.
Webinar: Wednesday January 16, 2019 9:00 PM EST CLICK HERE TO REGISTER TODAY! Daniel…
Study Finds Weight Bearing CT Scans Could Provide Accurate Measurements of Adult Acquired Flatfoot Deformity
Gaining an accurate and complete measurement of the hindfoot alignment of a patient with adult…
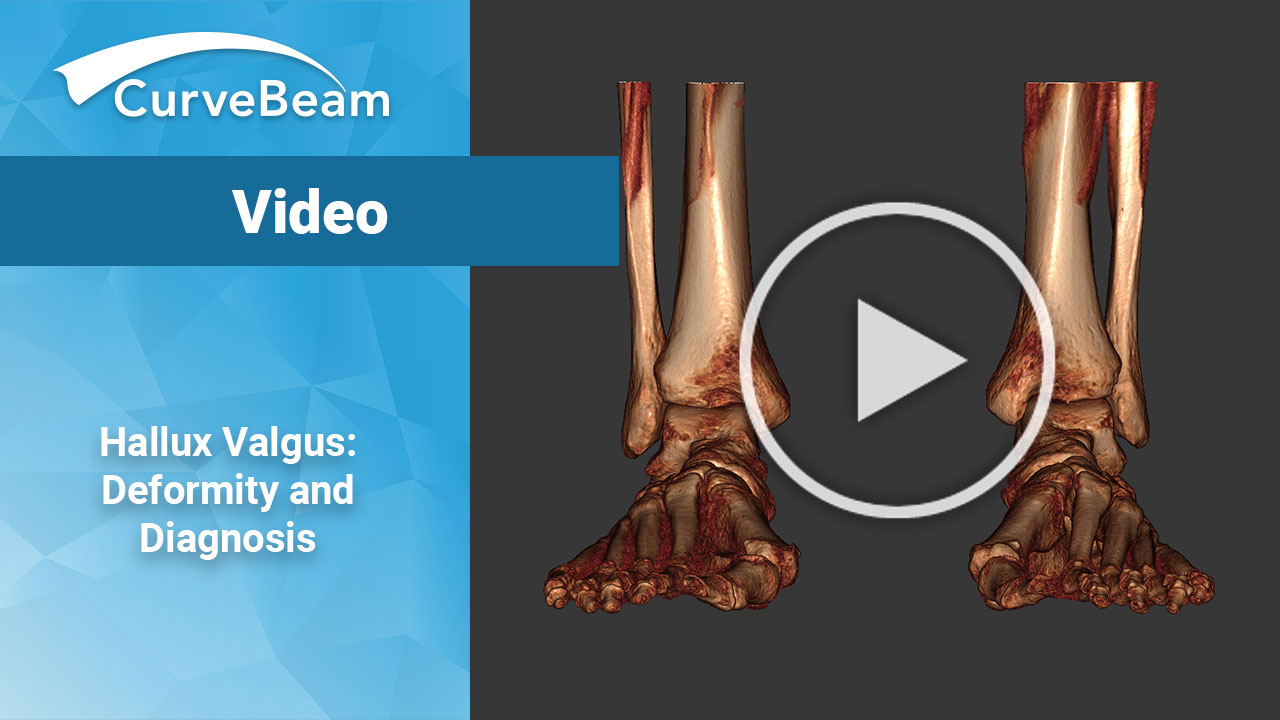
Hallux Valgus: Deformity and Diagnosis
Hallux Valgus, a deformity commonly called a bunion, occurs when there is a medial deviation…
Conventional Measurements of OLTS with CT Grossly Overestimate the Size of Lesions
When orthopedic surgeons attempt to evaluate and manage osteochondral lesions of the talus (OLTs) using…
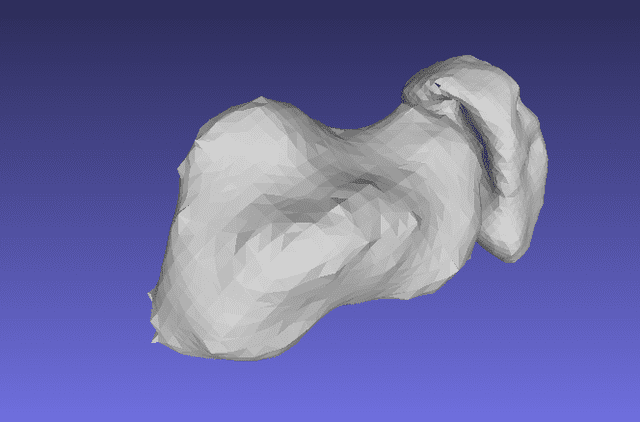
CurveBeam Plays an Important Role in the Best Research Awards at the EFAS Congress
The 12th annual European Foot and Ankle Society (EFAS) Congress was recently held in…
Dr. Alexej Barg WBCT ISG FOOTinnovate Webinar Nov. 12, 9 P.M. EST
Webinar: Monday, November 12, 2018 9:00 PM EST CLICK HERE TO REGISTER TODAY! Dr. Alexej Barg,…
Barry University School of Podiatric Medicine Acquires the pedCAT for Research
Weight-bearing CT imaging systems are quickly approaching the standard of podiatry care. The pedCAT bilateral…
Kent State Faculty and Students Research the Use of Weight Bearing CT to Effectively Assess Magnitude of Hallux Valgus Deformity
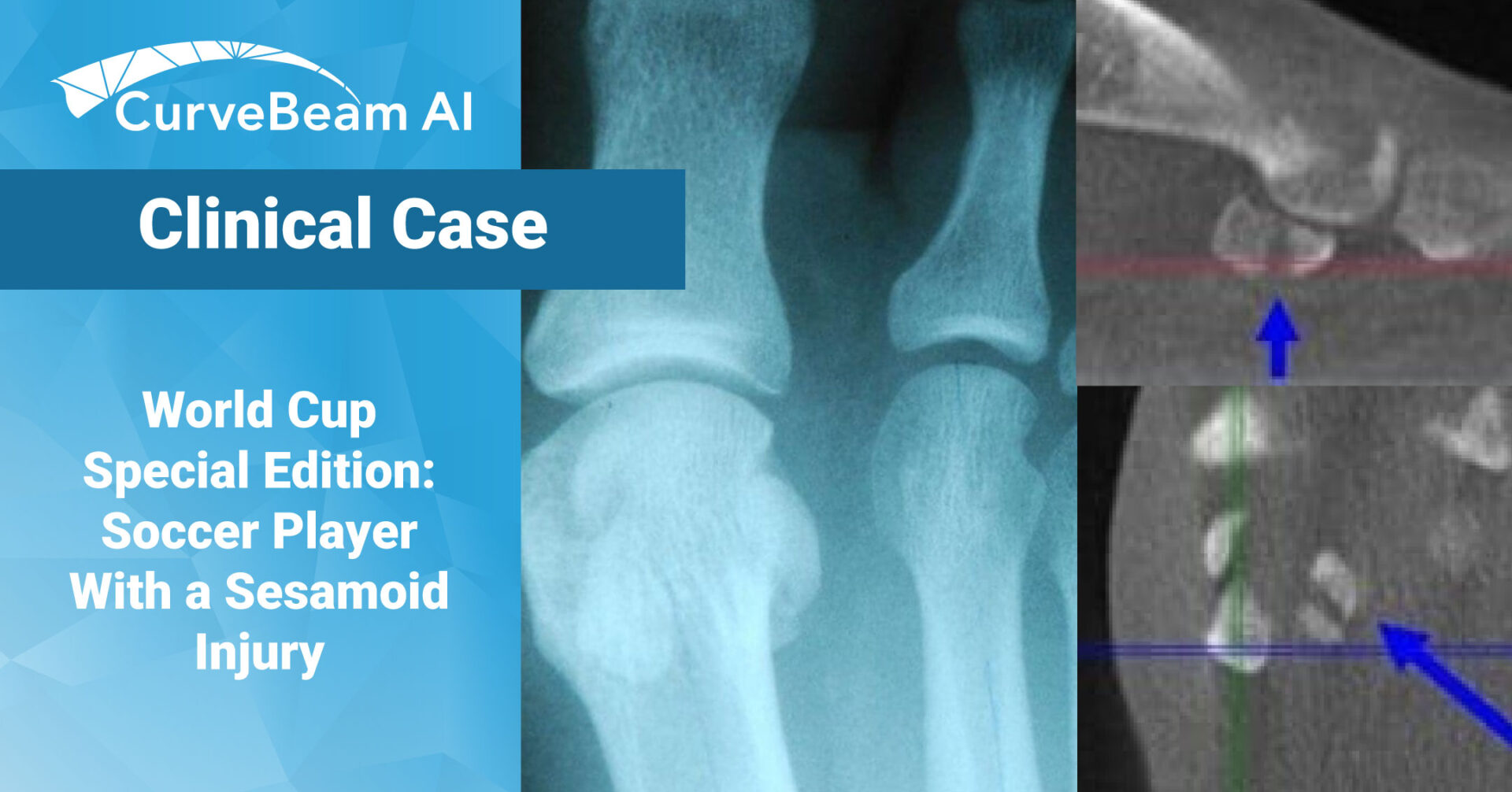
Hallux valgus is a triplane deformity. In patients with this deformity, the sesamoids displace from their…
Researchers propose reproducible method to quantify syndesmosis displacement based on spatial WBCT data
The syndesmosis is located just above the ankle where the tibia and fibula meet, providing…
Cone Beam Computed Tomography with Load Technique (WBCT) Allows Correct Evaluation of Foot and Ankle Anatomy
Imaging studies are important tools in diagnosis, therapeutic decision-making, and evaluation of functional results in…
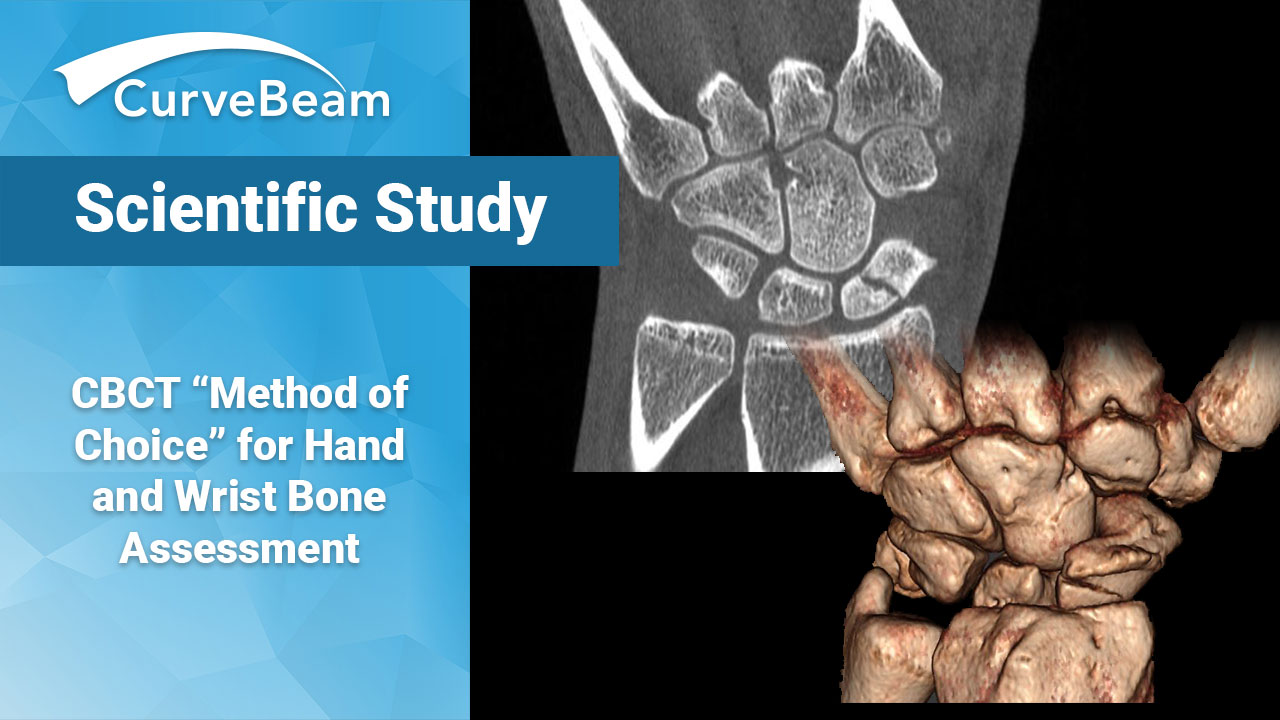
Study Calls CBCT “Method of Choice” for Hand and Wrist Bone Assessment
A study published in the International Journal of Biomedicine concludes Cone Beam CT (CBCT) imaging…
New Algorithm Uses CT Data to Create 3D Map and Better Assess Osteoarthritis
Engineers, radiologists and physicians based in the UK have developed a new technique for improving…
CurveBeam Relocates Headquarters for Expanding Team
In 2009, a group of professionals with a passion and proficiency in extremity CT imaging…
The University of Kansas Announces Grant Funding for Knee Imaging Biomarkers Acquired from Weight Bearing CT
The University of Kansas Medical Center Research Institute Department of Rehabilitation Medicine has received a…
CurveBeam Teams Up with the Paley Foundation to Change the Lives of Children
On June 3, the Paley Foundation hosted the Bill Bones Saving Limbs Ride to benefit…
Curvebeam Achieves ISO 13485:2016 & MDSAP Certification
Curvebeam announced the company's ISO 13485 certification has been updated to the latest revision of…
CurveBeam Unveils New Logo
On the heels of a recent FDA approval for its LineUP system, CurveBeam unveiled a…
Weight-Bearing CT May Improve Diagnoses in Hallux Valgus Patients
A recent analysis of studies titled Imaging of Hallux Valgus by James Welck and Naji…
i-FAB Awarded to Hindfoot Misalignment Research Study
Hindfoot malalignment is a major cause of pathology in the foot and ankle and impacts…
Studies Point to Many Benefits of Weightbearing CT Scanning Technology
For patients whose lives are impacted by medical technology breakthroughs, there is a significant improvement…
Weight Bearing CT Finds Important Displacement in Patients with Hallux Valgus
It’s difficult to understand how much a bunion can affect your life until you experience…
CurveBeam Throws A Curve at Conventional CT Scans for Orthopedic Practices
Often used for orthopedic and podiatric use when plain x-rays do not provide the visualization…
Health Canada Approves CurveBeam’s InReach Cone Beam CT
March 26, 2018 – Warrington, Penn. – CurveBeam announced the InReach cone beam CT system…
Weight-Bearing CT Can Help Doctors Zoom in on the Problem
Sometimes the simplest solution to a difficult challenge is a change of perspective. That’s exactly…
Joan Oloff is Making Fashion More Functional
All too often, our choice of wardrobe comes down to function or fashion. We sacrifice…
Why CurveBeam Technology is Even Changing the Way We Talk
Languages are constantly shifting and changing. As our experiences change, new terminology and ways to…
CurveBeam: Moving Orthopedics Forward
Scroll to the bottom of this post to access a technical comparison chart of the…
When Billing CT Scans, a Little Effort Goes a Long Way
It should come as no surprise that providing a patient’s clinical history can help…
Takeaways from the 2018 OrthoForum Annual Meeting
The 2018 OrthoForum Annual Meeting concluded this past weekend in Orlando, and the agenda was…
Curvebeam Corporate Webcast: Standing CT—Zooming in on the Problem with Dr. Selene Parekh
Mark your calendars for an upcoming FOOTInnovate lecture with Dr. Selene Parekh. Titled, “Standing CT—Zooming…
Orthopedic Cone Beam CT: A Primer for Radiologists
Over the last decade, cone beam CT (CBCT) imaging is gaining ground as an imaging…
Weight Bearing CT: Visualizing True Alignment of the Lower Limbs
Traditional CT scans require patients to lie down, limiting doctors' ability to assess how weight…
Avoiding False Negatives by Dodging Potential Pitfalls
It would seem radiography reigns supreme when it comes to initial fracture detection. And yet,…
In Lecture, Dr. De Cesar Netto Calls WBCT an Essential Diagnostic Tool
The field of foot and ankle pathology has seen recent technological advancements that allow specialists…
Examining the Added Value of Preoperative CT for Determining Cartilage Degeneration in Patients with Osteochondral Lesions of the Talar Dome
Osteochondral lesions of the talar dome (OLTs) involve the articular cartilage and subchondral bone. These…
A Mobile Solution for Weight-bearing CT in the UK
Britain’s national healthcare system can make getting a CT scan difficult, and weight-bearing scanners are…
Study Finds Low Levels of Radiation May Actually Help Fight Off Cancer
It has long been accepted that there is no such thing as a safe dose…
A Recent Study Demonstrates the Need for CT Scans in Complex Ankle Fractures
CT scans were shown to be better at detecting fractures over plain radiographs, according to…
CurveBeam Systems Offered on the Stratum Med GPO to Save Practices Money
CurveBeam, the designer and manufacturer of Cone Beam CT imaging equipment for orthopedic and podiatric…
RSNA Daily Dispatch
The Radiological Society of North America (RSNA) commenced its 103rd Annual Meeting in Chicago on…
Department of Energy Considers Radiation Research Funding
Radiation has a bad reputation, especially among lay people, but even in the medical community…
Pedography Plus PedCAT Clarifies the Relationship Between COG and FC
For those needing to analyze the relationship between morphology-based foot center (FC) and force/pressure-based center…
Webinar: Clinical Insights from Dr. Cesar de Cesar Netto
Current research in orthopedics and podiatry have revealed many advantages of weight-bearing CT procedures over…
CT Has Numerous Advantages over X-Ray in the Taylor Spatial Frame Treatment
Winner of the Medical Device Excellence Award, the Taylor Spatial Frame (TSF), is regularly employed…
Exposure to Low-Level Radiation is Less of a Risk than Obesity, Smoking, and Air Pollution
While the risks to health from medium- and high-level radiation have been studied for years…
A Sure Fit for the Mountains
Anyone who has been skiing knows that the difference between a fantastic day of carving…
Measuring Flexible Adult Acquired Flatfoot Deformity with Weight-Bearing High-Resolution 3D Cone-Beam CT Scanners
Adult acquired flatfoot deformity is a common, complex disorder associated with the failure of various…
Skeletomuscular Imaging Use Increased Dramatically, 1994-2013
Use of radiography, ultrasound, MR, and CT—the four most common imaging tests used for orthopedic…
One Step at a Time? Knee Replacements Are Advancing by Leaps and Bounds!
An increasing number of U.S. residents over the age of 65 has expanded the need…
In-Office CT Imaging Equipment Helps Urgent Care Centers Better Address Orthopedic Care
According to a 2015 survey by the Urgent Care Association of America, there are more…
New Findings on the Impact of Weight on Arthroscopic Osteochondral Talar Reconstruction; MRI Over CT for Detecting Edema
A recent study published by the American Orthopaedic Foot and Ankle Society on the impact…
Advanced CT Technology Increases Presence in Sports Medicine with Decreasing Footprint
Football season is about to begin and many teams are already in full practice mode…
New, Improved Tool for Measuring Hindfoot Alignment Made Possible Using 3D Biometric TALAS Software
A groundbreaking study that outlines a new semi-automatic tool that can be used to quantify…
CurveBeam Celebrates the History of Independence Day
Independence Day is one of CurveBeam’s favorite holidays. While every American loves celebrating the Fourth…
Weightbearing CT Helps Orthopedists More Effectively Treat Hindfoot and Ankle Deformities
Dr. Arne Burssens, orthopedic resident at Ghent University Hospital in Flanders, Belgium, held a webinar…
CT Scans: Customer Experience and Perception Drive Value
While doctors and radiologists understand the value generated from a CT scan, the same may…
Webinar: Pitfalls In Hindfoot and Ankle deformities tackled by weightbearing CT
You are invited to join FOOTinnovateTM and Dr. Arne Burrssens for the webinar “Pitfalls In…
CurveBeam and Carestream Health Announce Collaboration to Promote Weight-Bearing CT Awareness and Research
Warrington, Penn. – June 6, 2017 – Medical device companies CurveBeam and Carestream Health announced…
Keep Your Orthopedic Practice Alive Through Diversification
Medical providers are always looking for ways to create optimal patient outcomes while lowering costs…
A pedCAT Scan: Less Radiation Than A Serving of Brazil Nuts
Most people may not realize it, but humans are exposed to natural background radiation all…
The pedCAT Weight-Bearing CT Scan Put CurveBeam a Step Ahead of the Competition
It’s hard to forget about your feet—socks and shoes, not to mention pain at the…
CurveBeam Announces FDA 510(k) Clearance for InReach Cone Beam CT Imaging System for the Upper Extremities
May 8, 2017 – Warrington, Penn. – CurveBeam announced it has received FDA 510(k) clearance…
Discussion Looks to Provide Blueprint for Foot and Ankle Deformity Correction
Foot and Ankle Specialist (FAS), a bi-monthly journal for orthopedic surgeons and podiatrists, recently published…
Weightbearing CT Revolutionizing Hindfoot Alignment Measurements
A new study, led by famous Belgian M.D. A. Burssens, emerged in the February issue…
CurveBeam Is the Innovator in 3D Orthopedic Imaging Technology
Cone beam computed topography, or CBCT, has become the standard of care in orthopedic imaging…
Jon-David Deeson Testimonial on pedCAT
During OrthoForum’s Annual Conference in early March, OrthoTennessee CEO Jon-David Deeson discussed his experiences with…
Literature Review and Case Studies from CurveBeam’s “Weight-Bearing CT of the Foot and Ankle” Webinar
On February 23, Dr. Alexej Barg, an orthopedic surgeon at University of Utah Health Care,…
CBCT Technology is Increasingly Used by Orthopedists and Podiatrists
Cone beam computed topography (CBCT) systems are becoming popular with orthopedists and podiatrists because they…
The Hospital for Special Surgery Utilization of pedCAT
The Hospital for Special Surgery (HSS) located in Manhattan is ranked as the #1 hospital…
CurveBeam Selected to Join The OrthoForum
2017 is in full swing, and we at CurveBeam are excited about the prospects the…
Announcing a CurveBeam Webcast: “Weight-Bearing CT of the Foot & Ankle – Literature Review and Indications for Use in the Clinic”
To delve into the effect Weight-Bearing CT scans can have on modern orthopedic medicine, we…
Lower Limb Influence on Hindfoot Alignment
A recent study headed by L. Baverel, a distinguished French M.D., came out of the…
The Opportunity for Private Office Imaging
Changes in healthcare insurance, regulations, and other factors have shifted the bulk of imaging services…
CurveBeam’s Volunteer Day Warms Community
The holiday season is an excellent time for any company to celebrate and share its…
CurveBeam’s Top 5 Moments of 2016
Founded in 2009, the news of CurveBeam and its advanced, compact 3D imaging devices spread…
Comparison of Three Hindfoot Alignment Measurements: Radiographic Hindfoot Moment Arm, Radiographic Hindfoot Alignment Angle, and TALAS
Dr. Matthew Welck presented findings from a study overseen by Dr. Mark Myerson at the…
Value of 3D Reconstructions of CT Scans for Calcaneal Fracture Assessment
Operative fracture treatment of irregularly shaped bones, such as the calcaneus, scapula and scaphoid, demands…
Using The InReach to Detect Scaphoid Fractures
Scaphoid fractures are often difficult to diagnose. The scaphoid may be difficult to visualize on…
From RADAR to pedCAT, Amrita Kar is Making Her Mark
The strength of our company originates with engaged and driven employees. The staff at CurveBeam…
Join The TALAS Revolution in Foot and Ankle Diagnosis
For years, Dr. François Lintz, an orthopedic foot & ankle surgeon at Clinique de l’Union…
Weightbearing CT Imaging Methodology with pedCAT by CurveBeam
Three-dimensional weight-bearing computed tomography (CT) can be a powerful diagnostic tool, typically used when more…
Healthcare costs have skyrocketed in recent years
This trend has bewildered patients, who see their medical bills ballooning with no commensurate improvement…
The Cream of the Crop
A company is only as good as its team members, and CurveBeam is no exception…
CurveBeam CT Imaging Technology is Right On Point
Ballet is an art of extremes. As such, the risk factors are high among dancers…
Webinar Summary: Advanced CT Imaging in Foot and Ankle Surgical Considerations
We all know that X-rays and MRIs aren’t the be-all, end-all of diagnostic imagery. Try…
CurveBeam Announces First pedCAT Installation at an Accredited College of Podiatric Medicine
CurveBeam is proud to announce the installation of a pedCAT cone beam CT imaging system…
Weight Bearing CT Sheds Light on Understanding of Rotational Dynamics in Syndesmosis
As the orthopaedic and podiatric specialties continue to advance, there is great potential for technology…
pedCAT Shown to be Most Effective Imaging Option for Mülller-Weiss Disease
The CurveBeam pedCAT imaging system was recently recommended as an incredibly beneficial option for the…
Weight Bearing CT Advances Planning for Forefoot Reconstructive Surgery
Think of a surgical procedure like a construction project. No contractor would build a building…
What Is Cone Beam CT? The Science Behind the Image. Part 1: Overview
Within the last two decades, Cone beam Computed Tomography (CBCT) imaging applications have diversified in…
Need Bunion Surgery? A Weight Bearing CT Scan Could Help You Decide
When a patient appears to have a bunion, a physician typically orders traditional foot X-Rays…
Orientation of the Subtalar Joint: Measurement and Reliability Using Weight Bearing CT Scans
Is there a reliable method to predict the type and perhaps the extent of osteoarthritis…
Panel Recap: Dr. Michael Chin, DPM, Speaks about pedCAT at FABI
His decade old X-Ray system was failing, and feeling like a warrior without his weapon,…
Webinar Recap: “Using Weight Bearing CT to Guide Clinical Decisions”
Dr. Steven K Neufeld, MD, presented “Using Weight Bearing CT to Guide Clinical Decisions” on…
Weight Bearing CT Imaging for Cuboid Subluxations
Dr. Michael Chin, DPM, presented how weight bearing CT imaging has changed how he evaluates cuboid subluxations at the American Academy of Podiatric Sports Medicine meeting held near the West Point Military Academy campus in early September, 2015. Dr. Chin began using the pedCAT in his office in February of this year. Not much research is out there on how to use plain radiographs to measure cuboid subluxations, Dr. Chin said in his lecture, titled, “Cuboid Syndrome…The Other Side of Heel Pain.” Dr. Chin has tested using a bilateral oblique projection to understand the cuboid/ metatarsal relationship, and has been able to observe a slide between the head of the fourth metatarsal and the head of the cuboid. An MRI could be ordered to see the condition of the peroneal tendon, but the study would be limited because the scan would not be weight bearing, he said.. A traditional CT scan would provide great visualization of the bone, but would provide no information on anatomic alignment. The pedCAT weight bearing CT imaging system is excellent for evaluating stress fractures, sesamoids, periosteal changes, or anything medullar, Dr. Chin said. Another benefit is he can measure the exact degree of subluxation between the cuboid and the fourth metatarsal head. Dr. Chin displayed pedCAT images depicting pre and post-reduction views of a cuboid subluxation. pedCAT scan of a pre-surgical patient with cuboid subluxation. Dr. Chin was able to reduce the subluxation to 2.18 mm. Dr. Chin practices at The Running Institute in Chicago.
AOFAS Annual Meeting – CurveBeam Symposium Recap
Weight bearing CT is a vital tool for determining the cause of inexplicable pain, and also for avoiding painful surgical complications. That was the takeaway message from a talk by Dr. Phinit Phisitkul, a clinical associate professor of orthopaedics at the University of Iowa. He shared some of his most interesting cases at a CurveBeam sponsored symposium held during the AOFAS Annual Meeting in Long Beach during the evening session. We’ve selected three of his cases to share on this blog: 18-year-old male with Noonan Syndrome & severe flat foot: The patient presented with an unusual amount of pain that was difficult to diagnose on plain X-Ray. A weight bearing CT scan revealed he had a severe deformity – a congenital vertical talus. He also had severe impingement. Vertical Talus - Weight Bearing CT Vertical Talus - Weight Bearing CT Impingement - weight bearing CT Impingement - weight bearing CT 58 year-old male with ankle arthritis: The patient presented with a lot of pain in the ankle joint. A weight bearing CT scan showed a subluxation of the ankle joint and dramatic impingement of the calcaneal fibula. Interestingly, the subtalar joint was in pristine condition. Dr. Phisitkul determined the patient was a good candidate for ankle replacement and hindfoot realignment, and that his subtalar joint could be spared. Calcaneal-fibular impingement and arthritis - Weight Bearing CT Calcaneal-fibular impingement and arthritis - Weight Bearing CT 41-year-old female with Hallux Valgus: A weight bearing CT scan revealed a bone spur on the patient’s first metatarsal head. If the doctor had done a normal release, the spur may have ended up pinching the sesamoid. Instead, he performed a lateral release and excised the bone spur.
pedCAT: A Positive User Experience
“After using the pedCAT for a year, I am very impressed with how much I need it, and how many different things I use it for,” Dr. Bob Baravarian said at the 2015 Western Foot and Ankle Conference held in Anaheim, Cali. The California Podiatric Medical Association hosted the meeting at the end of June. Dr. Baravarian cited Hallux Rigidus as one example where pedCAT imaging has been helpful because he is able to properly assess the sesamoids. Dr. Baravarian confirmed his practice is realizing positive revenue streams as a result of the device.
Diagnostic Options for Freiberg’s Avascular Necrosis
The underlying causes of avascular necrosis of the second metatarsal head are not totally understood, but early diagnosis is essential. Delayed treatment can result in a collapse of the articular surface, making treatment more difficult. Dr. Bob Baravarian, DPM, explained his preferred methods for diagnosing the condition in the May 2015 issue of Podiatry Today. An X-Ray will show the overall contour and alignment of the metatarsal head. MRI is one option for a secondary study, but edema and swelling can limit visualization of the surrounding bone region, Dr. Baravarian explained. Weight bearing CT scans, meanwhile, clearly show the bones and joints and how they are aligned. Weight bearing CT is "our go-to imaging study," Dr. Baravarian said in the article. "With adequate diagnostic testing and proper patient and procedure selection, one can treat avascular necrosis of the metatarsal head with good to excellent outcomes," Dr. Baravarian said. Dr. Baravarian is the director of University Foot and Ankle Institute in Los Angeles. His practice offers weight bearing CT imaging services.
Weight Bearing CT in Everyday Practice
“In my opinion, this will be the standard of care in the next couple of years. We are switching from two-dimensional technology to three-dimensional technology and this is really the future of not just foot and ankle, but the future of medicine,” Dr. Alex Tievsky, DPM, said in a lecture he gave at the Graham International Implant Institute 8th Annual Symposium in Miami, on April 17. 2D is beginning to be phased out,” Tievsky said. “Now we’re beginning to see the problem from all angles and all planes, so this is super helpful from that respect.” Dr. Tievsky presented a number of cases where he benefited from access to weight bearing 3D technology in his office. Clinical Case #1 A 50-year-old female presented with bilateral flat feet for 20 years. She had heard about the HyProCure procedure, which corrects hindfoot misalignment through a minimally invasive procedure. She was eager to have the procedure done, no matter the cost. Dr. Tievsky took a pedCAT scan, and found she had a severe talar coalition. talar calc coalition coronal left “How many times do you catch a coalition on an X-Ray? It’s hard,” Dr. Tievsky said. “Sometimes you can see a halo sign, but it’s often missed. On the first visit, I was able to tell her, we either have to resect this coalition or we have to do a fusion. It’s impossible to get this level of information on an X-Ray.” Clinical Case #2 A 16-year old girl came in with first metatarsal head pain. She had already been to two other podiatrists in the last six months, and they had prescribed steroid injections. The pedCAT revealed a fracture on her fibular sesamoid that is extremely easy to miss on X-Ray. fractured sesamoid “Within her first 10 minutes in the office, we had a diagnosis,” Dr. Tievsky said. “We treated her appropriately. We immobilized her for eight weeks and gave her a bone stimulator. And she was pain free, three podiatrists later. She was happy, her mom was happy, and she never came back.” Clinical Case #3 A patient presented with a lateral plantar fasciitis, a talo-tarsal dislocation, back pain for five years, knee pain, and hip pain. A pedCAT revealed she had a tumor in her bone and it was eroding it. “There was no way we would have caught it on X-Ray,” Dr. Tievsky said. “We sent her out for oncology. It could have a malignant tumor, and we could have saved her life.” Cyst Coronal The scan is quick to take and you have a full work-up in about three minutes, Dr. Tievsky said as a closing statement. “This is a super important tool,” he said. “I’m kind of biased. I love this now. I can’t practice without it.”
Weight Bearing CT Scans for the Evaluation of Implant Arthroplasty Candidates
Weight bearing CT scans can be critical to a proper diagnosis, even for routine procedures. In the following case, for example, a patient’s X-Rays indicated that he would be a good candidate for a metatarsal head hemi-implant arthroplasty. However, when the patient sought a second opinion, a weight bearing CT (pedCAT) scan revealed the true condition of the metatarsal head, and the surgical plan was considerably altered as a result. Click on the blog post title to read the case and view images.
Dr. Gary Briskin: The pedCAT is the Future
"It's the future - embrace it. It's your future - control it." Dr. Gary Briskin delivered this motivating message at the Foot & Ankle Business Innovations conference in Chicago on Jan. 23. Dr. Briskin, of University Foot & Ankle Institute in Santa Monica, Cali, discussed how the pedCAT weight bearing CT system has improved his practice since his group acquired one last year. His main take-away points were: - The pedCAT gives you the advantage when it comes to diagnosing pathology. "You can solve that Lisfranc case no one else can," Dr. Briskin said. "It opens up the midfoot, which historically has been a problem for us." - The pedCAT allows him to clearly determine if bones and joints are completely fused after surgery. "I do a lot of lapidus procedures," Dr. Briskin said. "My concern; is this patient fused adequately to start bearing weight?" The exact level of fusion is visible with a pedCAT scan 5 - 6 weeks later, and with minimal scatter. - Radiologists like the pedCAT images "I think I'm getting a better CT, because it's weight bearing and I think the quality is far superior. We get all of our CTs read by a radiologist. We have everything sent digitally. And the feedback we get is they are also impressed with the quality of the images."
pedCAT: Optimal Visualization of Metal Implants and Hardware
One advantage of CT imaging over MRI imaging is the ability to scan patients…
pedCAT: Visualizing the Sesamoids in a Hallux Valgus Deformity
Dr. Robert Weinstein, DPM, recently lectured at The International Foot & Ankle Foundation for Education…
Case Study: pedCAT vs. Radiographs
A patient sought a second opinion for the cause of her medial foot and ankle pain. The treating physician used plain radiographs to diagnose her with posterior tibial tendon dysfunction/partial tear and degenerative joint disease of the 1st, 2nd, and 3rd tarsometatarsal joints. The treating physician also noted a “chip” of bone on the inside of the ankle. The treating physician planned on performing a flat foot reconstruction, a posterior tibial tendon repair, and a tarsometatarsal joint fusion. The physician performing the second opinion noted the patient’s discomfort over the medial ankle gutter was much more significant than over the posterior tibial tendon and the spring ligament. The patient had minimal discomfort through the tarsometatarsal joints. The physician performing the second opinion ordered a weight bearing pedCAT study to assess the midfoot DJD and to better evaluate the midtarsal joint and ankle joint. The pedCAT study clearly documented a degenerative process in the medial ankle gutter with a bony impingement. On secondary exam, the majority of the symptoms arose from the medial ankle gutter. If the flat foot reconstruction was performed as planned, the talus would have been dorsiflexed and the tibio-talar impingement would have been worsened. The pedCAT images helped prevent an un-necessary surgery, while directing the physician to the appropriate pathology.
pedCAT CubeVue: Automatic X-Ray Views
It takes less than a minute to scan a patient in the pedCAT, but that’s enough time for the pedCAT system to collect enough data to create a 3D reconstruction of the foot and ankle, as well as .3 mm slices in all three planes.
Distinguishing Categories of Medial Column Ligamentous Fracture
Weight bearing CT scans offer new, three-dimensional perspectives that reveal a better understanding of the biomechanics of flat foot. That was a finding on an academic poster on display at The American Orthotic and Prosthetic Association's National Assembly in Las Vegas (Sept. 4 - 9). "IRD, PTTD, Posterior Tib, Flat Foot, Pronation...these are all terms that various professions use to describe some sort of medial column ligamentous breakdown," according to the poster. "The purpose of this study is to better distinguish some types of pathomechanics of the foot. By categorizing and redefining them, it makes the description of pathology more useful for Orthotic Treatment. This was possible through the use of 3D rendering of the pedCAT machine and CurveBeam software." The study was conducted by Ian Engelman, M.S. CPO, and Harold Chamberlin, DPM. They looked at 3D renderings of the bony structures in flat feet, and came up with some new descriptions, as well as their possible ligamentous causes. The study concluded that while the exterior contours of the foot suggest certain bony pathologies, the 3D renderings provided by the pedCAT would allow for orthotists to better distinguish the pathomechanics of the foot and ankle. An unexpected result showed a moderately strong correlation (-0.68) between the vertical position of the navicular and medial displacement in a patient population with ranging degrees of medial column breakdown.
pedCAT: Weight Bearing CT Views of Lisfranc conditions
Lisfranc injuries can easily be missed on traditional radiographs. One research study tried to determine the level of accuracy when using plain radiographs to assess midfoot conditions. Their results confirmed a high incidence of missed diagnosis even by experienced observers.¹ “Radiographic evaluation is crucial in the diagnosis…Weight bearing views are more sensitive since tarso-metatarsal instability may be revealed…The threshold for cross sectional imaging such as multi-detector CT should be low,” according to the study. With the pedCAT, it is possible to get a weight bearing, three dimensional scan at the point of care. The tarso-metatarsal joint can be viewed without any superimposition from surrounding anatomy. Delaying a 3D scan can be detrimental to recovery. “The identification of Lisfranc joint injury can be difficult and is often not detected on initial presentation to the accident department. This is important because of the correlation between delay in treatment, particularly more than six months, and poor functional outcome,” the study reported. Click on the blog post title to see examples of weight bearing CT scans of Lisfranc injuries.
Webinar: Dr. Erik Nilssen – A New Paradigm in Foot & Ankle Diagnosis
“Patients come to my practice with an expectation of a higher level of care, that they’re getting the best. With the pedCAT, I am able to give them information immediately, and they leave feeling this was well worth the trip.” – Dr. Erik Nilssen Learn how the pedCAT has changed Dr. Nilssen’s clinical practice in a webinar hosted by Foot & Ankle International and Foot & Ankle Specialist. We hope you can join us! Click on the title of the blog post for the link to sign up.
pedCAT: Fracture & Fusion Assessments
X-rays of the foot and ankle may not always provide conclusive assessments for post-operative fusions such as of the tarso-metatarsal joint or hind foot joints. Similarly, the physician is often left guessing if a fracture has properly healed. “The pedCAT takes all the variability out, all the guesswork out of it,” Dr. Martin O’Malley, MD, an Associate Attending Orthopedic Surgeon at the Hospital for Special Surgery in New York, New York, said. Dr. O’Malley said pedCAT scans allow him to clearly determine if a fusion has healed more than 50 percent, and he decides when to ambulate his patient accordingly. “I let people walk on it earlier than before, and I keep them off longer than before,” Dr. O’Malley said. The pedCAT provides a three-dimensional view of fractures that changes the way O’Malley sees this common diagnosis. “These posterior pieces are often bigger than we thought were based on plain X-ray and they often travel all the way around the medial side as well, which we never thought they did,” Dr. O’Malley said. “You know, we thought it was an infrequent fracture, but now we see it routinely. Now most of my ankle fracture work, I’d say more than half the time, is through a posterior approach. For the first 15 to 18 years of my practice I would do medial/ lateral incisions. Now I’m going to the back of the ankle. And a lot of it is driven by the pedCAT.” pedCAT Stress Fracture Cross Section Click on the blog post title to see an example of a navicular stress fracture in a collegiate runner that had not healed at all after six weeks of casting. Were it not for the conclusive pedCAT scan, this patient would have been allowed to ambulate. Watch Dr. O’Malley talk more about the pedCAT here . To offer your patients state-of-the-art fracture & fusion assessment, consider a pedCAT for your practice.
pedCAT: Will it Fit?
The pedCAT has been deployed to a range of medical facilities – from large hospitals to single physician practices. Each site has its own unique requirements, and CurveBeam works with each customer to ensure the first weight bearing CT scanner dedicated to the foot & ankle meets all safety requirements. Because the pedCAT is an ultra low dose device, the shielding infrastructure required is similar to that of a plain X-Ray device. The pedCAT does not need to be in a lead lined room. Because of its compact size and relatively minimal required shielding, the pedCAT can easily fit into a practice with limited space. Click on the title of this post to see some examples.
World Cup Special Edition: Soccer Player with a Sesamoid Injury
The 2014 FIFA World Cup officially begins Thursday, June 12. The world’s top soccer players will all be in Rio, Brazil for one of the biggest sporting events of the decade. The next two weeks will be filled with cheering fans, tears of joy, and national pride. But inevitably, there will also be injuries. In fact, before the games even begin, a number of players already know they will have to sit them out. For professional athletes and weekend warriors alike, an accurate diagnosis from the onset can make a huge impact on the speed of recovery. When it comes to bony injuries of the foot and ankle, the pedCAT is the only tool in the world that can provide a weight bearing, three dimensional image of the entire foot. Here’s a case of a 30-year-old soccer player, who was treated by a California podiatrist. The athlete injured his right great toe and sesamoids while playing soccer. The doctor observed that the patient’s first metatarsophalangeal joint was swollen and ecchymotic, and that he was tender upon palpation of the great toe joint and sesamoid bones. The doctor took standard non-weight bearing X-Ray images of the patient. The AP and lateral X-Rays showed an obvious fracture with displacement in the medial sesamoid. The lateral appeared to have a fracture, but the doctor could not confirm his suspicion with the X-Ray. The doctor also took an axial X-Ray, but it had no diagnostic benefit since the patient could not extend his great toe. double sesamoid fracture x-ray The doctor then took a pedCAT scan of the same patient. The CT images clearly showed a severely displaced right medial sesamoid oblique fracture and a minimally displaced lateral sesamoid fracture. The oblique lateral sesamoid fracture demonstrated 2 mm of plantar displacement with excellent bony contact/ apposition in the dorsal 4 mm of the sesamoid. pedCAT double sesamoid fracturepedCAT lateral sesamoid fracture displacement Although the diagnostic information provided by the pedCAT did not alter the treatment plan for the medial sesamoid, it did confirm the injury to the lateral sesamoid. More importantly, the pedCAT was able to provide diagnostic information regarding the geometric nature of the lateral sesamoid fracture. “I could determine that there was enough bony contact to allow for bone healing with appropriate conservative care management,” the doctor said.
New Study: pedCAT Allows for More Accurate Bone Angle Measurements than Radiographs or CT
A study published in Foot and Ankle Surgery compared three common angle measurements of the foot taken from non-weight bearing CT, weight bearing digital radiographs, and weight bearing pedCAT scans. The study concludes, "The angles differed between radiographs, CT and pedCAT, indicating that only pedCAT is able to detect the correct angles... pedCAT prevents inaccuracies of projection and foot orientation in contrast to radiographs due to the 3D dataset which is principally independent from projection and foot orientation." The study also found the average image acquisition time for the pedCAT (average 270 seconds) was 70 percent faster than with radiographs and 35 percent faster than with CT. The study was authored by Martinus Richter, Bernd Seidl, Zech Stefan, and Sarah Hahn.
High Resolution Imaging
pedCAT scans give you one of the highest resolution views of the foot and ankle available. And those high resolution views are infinite - physicians can view the foot and ankle from the axial, coronal and sagittal planes by scrolling through .5 mm slices. The average medical CT slice, in comparison, is about 1 mm – 5 mm thick.
pedCAT: Improved Outcomes
The message at RSNA Annual Meeting this past November was clear: new US healthcare laws mean the practice of radiology is no longer about volume, but value. “We are focusing on quality metrics. It’s becoming important for us to become champions of quality in our institutions,” said Dr. Vijay Rao, MD, in a course at the meeting. How could a pedCAT add value to your practice? We might get an idea by looking at a comparable new technology. Breast tomosynthesis mammography provides 3D imaging for breast cancer screening, similar to the way the pedCAT provides 3D imaging for the foot and ankle. Breast tomosynthesis technology can detect breast cancers earlier than traditional 2D mammography, and can more accurately pinpoint the size, shape and location of abnormalities, according to the Massachusetts General Hospital Imaging Department. Dr. Liane Philpotts, professor of diagnostic radiology at the Yale School of Medicine, called tomosynthesis a “game changer” and a “win-win.” In the same way, the pedCAT eliminates variability, helps lead to better diagnoses, and makes both you and your patients more confident that treatment will result in better outcomes. Dr. Erik Nilssen, MD, said the pedCAT helps him determine exactly when to allow patients to ambulate, “based on our ability to monitor fracture healing and fusion rates.” Also, pedCAT scans can take the guesswork out of hindfoot alignment, said Dr. Martin O’Malley, MD, because they allow for reproducible measurements. “We’ve never had a reproducible measurement,” until now, he said. To see an example of a pedCAT scan that led to a more accurate diagnosis, click on the blog post title.
pedCAT: Easy to Incorporate into Your Medical Facility
The pedCAT system and your iPhone charger have something in common…you can plug them both in anywhere! That’s right. The pedCAT plugs into any standard 120 volt power outlet (or 220 volt outlet in certain countries). We suggest the pedCAT be connected to a dedicated line to prevent interference from other equipment as a precaution, although it is not required. The pedCAT’s simple power requirements are one of the factors that make it easy to integrate into any office workflow – even offices that have limited free space. Click on the blog post title to see some examples.
pedCAT: What’s the Dose?
At CurveBeam, patient safety is the top priority. When it comes to CT imaging, it’s important to know the radiation dose a patient can expect during the procedure. So what is the radiation dose of a typical weight bearing CT scan taken with the pedCAT? But before we get to that, here’s a little quiz. Can you rank the below items from greatest to least in terms of radiation exposure? - A pedCAT scan of both feet - A serving of Brazil nuts - A roundtrip flight from New York to Tokyo - An X-Ray of the chest - Working in an Australian Uranium mine for a year (Click the blog post title to see the answer.)
Made in the USA
At CurveBeam, we are proud that the pedCAT is manufactured entirely in the United States. Our factory headquarters are located in Warrington, Penn., just north of Philadelphia. The factory floor is housed in the same building as our administrative, sales, and research & development offices. We are committed to creating opportunities in our community. Most of the pedCAT’s components are sourced from local manufacturers as well. CurveBeam’s manufacturing process is certified by ISO, a developer of voluntary industry standards. Each pedCAT goes through a rigorous testing process to ensure it produces high quality images. An engineer surveys inventory at CurveBeam headquarters. An engineer surveys inventory at CurveBeam headquarters. Engineers on the factory floor follow the principles of lean manufacturing, developed by Toyota. The process is easily scalable, meaning CurveBeam can react quickly to large orders or orders that require a fast turnaround time. We would like to extend an open invitation to all foot and ankle specialists to visit us. We’d be happy to give you a tour and show you the pedCAT “behind the scenes.” Tweet: Check out this example of high tech medical device manufacturing on American soil: http://ctt.ec/HPlnc+
Welcome to the CurveBeam Blog!
Welcome to the CurveBeam Blog! It’s an exciting time at CurveBeam. Almost two years ago…